Abstract
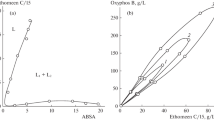
Color-changeable extradants for acids, acidophores, were synthesized and examined; they were 4-(N,N-dimethylaminophenylazo)-pyridines and quinolines. The fundamental physicochemical constants for the acidophores and their ion associates were determined by spectrophotometry. The liquid-liquid distribution was carried out between an aqueous phase and a chloroform phase. A parameter of the extractability of the protonated cations of the acidophores and of several inorganic and organic anions as ion associates was determined on the basis of the assumption that the extraction constant of an ion association complex, log Kex, is divided into a cationic and an anionic contributions, C and A, respectively. The order of the extractability of the anions was: Br<I−<ClO4−<SCN-<Pic−<(Ethyl Orange)−<C12H25-OSO3−. Anionic surfactants could be quantitatively extracted into chloroform with 4-(4-N,N-dimethylaminophenylazo)-2-methylquinoline: the molar absorptivity was 4.5×104 l mol−1 cm−1 at 560 nm. The acidophores once used could be recycled and be used repeatedly.
Similar content being viewed by others
References
A. S. Kertes, Y. Marcus and E. Yanir, “Equilibrium Constants of Liquid-Liquid Distribution Reactions”, Butterworths, London, 1974.
A. S. Kertes, “Critical Evaluation of Some Equilibrium Constants Involving Alkylammonium Extradants”, Pergamon Press, Oxford, 1977.
T. Sekine and Y. Hasegawa, “Solvent Extraction Chemistry”, p. 218, Marcel Dekker, Inc., New York and Basel, 1977.
M. Puttemans, L. Dryon and D. L. Massart, Anal. Chim. Acta, 161, 221 (1984).
M. Puttemans, L. Dryon and D. L. Massart, Anal Chim. Acta, 161, 381 (1984).
S. Motomizu and H. Kubota, Proceedings of Symposium on Solvent Extraction 1989, Sendai, November 1989, p. 45.
R. W. Faessinger and E. V. Brown, J. Am. Chem. Soc, 73, 4606 (1951).
R. W. Faessinger and E. V. Brown, Trans. Kentucky Acad. Sci., 24, 106 (1963). Chem. Abstr., 60, 14465 (1964).
E. V. Brown, R. M. Novack and A. A. Hamdan, J. Org. Chem., 26, 2831 (1961).
S. Motomizu, Y. Hazaki, M. Oshima and K. Toei, Anal Set, 3, 265 (1987).
C. Hansch and A. Leo, “Substituent Constants for Correlation Analysis in Chemistry and Biology”, p. 49, Wiley-Interscience Publ, New York, 1979.
S. Motomizu, Bunseki Kagaku, 33, 31 (1984).
S. Motomizu and K. Tôei, Anal Chim. Acta, 120, 267 (1980).
S. Motomizu, Bunseki Kagaku, 38, 147 (1989).
I. M. Klotz, H. A. Fiess, J. Y. Chen Ho and M. Mellody, J. Am. Chem. Soc, 76, 5136 (1954).
E. V. Brown and W. H. Kipp, Cancer Res., 30, 2089 (1970).
K. Gustavü, Acta Pharm. Suecica, 4, 233 (1967).
K. O. Borg and D. Westerland, Fresenius’ Z. Anal Chem., 252, 275 (1970).
G. Schill, “Ion Exchange and Solvent Extraction”, ed. J. A. Marinsky and Y. Marcus, Vol. 6, Chap. 1, p. 17, Marcel Dekker, New York, 1974.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kubota, H., Motomizu, S. Liquid-Liquid Extraction Behavior of Inorganic and Organic Anions with Color-Changeable Neutral Lxtractants. ANAL. SCI. 6, 737–745 (1990). https://doi.org/10.2116/analsci.6.737
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.6.737