Abstract
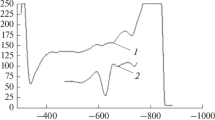
The (FeNO)2+ complex can be formed in a solution that contains ferrous ion (ammonium ferrous sulfate), nitrite (sodium nitrite) and ascorbic acid. With a dropping mercury electrode, this complex has a catalytic wave at —0.58 V which can be used for the determination of trace nitrite in the range of 1×10-7 to 4×10-4 M with the detection limit of 5×10-8 M. The function of ascorbic acid is to reduce NO2- to NO quantitatively for complete formation of the electroactive complex. The mechanism of the electrode process has been studied by linear sweep polarography, direct current polarography, cyclic voltammetry and spectrophotometry.
Similar content being viewed by others
References
B. Keilin and J. W. Otvos, J. Am. Ckem. Soc, 68, 2665 (1946).
J. Koryta, Collect. Czech. Chem. Commun., 20, 667 (1959).
D. T. Chow and R. J. Robinson, Anal. Chem., 25, 1493 (1953).
F. Baumann, J. Electroanal. Chem. Interfacial Electrochem., 6, 245 (1963).
S. W. Boese, V. S. Archer and J. W. CLaughlin, Anal Chem., 49, 479 (1977).
E. Wang and X. Lin, J. Electroanal Chem. Interfacial Electrochem., 136, 311 (1982).
R. E. Hamm and C. D. Withrow, Anal. Chem., 27, 1913 (1955).
A. Saito and S. Himeno, J. Electroanal. Chem. Interfacial Electrochem., 98, 181 (1979).
N. Kato, K. Yoshikiyo, K. Nakano and K. Tanaka, Bunseki Kagaku, 32, 139 (1983).
S. T. Sulaiman, Microchem. J., 29, 201 (1984).
Z. Zhao and X. Cai, J. Electroanal. Chem. Interfacial Ekctrochem., 252, 361 (1988).
R. Greef, R. Peat, L. M. Peter, D. Pletcher and J. Robinson, “Instrumental Methods in Electrochemistry”, p. 199, Ellis Horwood, Chichester, 1985.
F. Feigle and M. Steinhauser, Mikrochem. Mikrochim. Acta, 35, 553 (1950).
J. P. Yuan, Q. S. Tian and X. H. Cai, Fenxi Huaxue, 16, 788 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mo, J., Jin, D., Pei, J. et al. Single Sweep Polarographic Determination of Trace Nitrite in Ferrous and Ascorbic Acid Medium. ANAL. SCI. 6, 251–255 (1990). https://doi.org/10.2116/analsci.6.251
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.6.251