Abstract
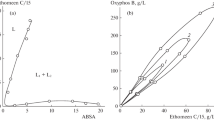
Several metal chelates of 2-(2-pyridylazo)-5-methylphenol (PAP-5-Me) were extracted from an aqueous phase into a surfactant phase which was separated from a micellar solution of poly(oxyethylene)=4-nonylphenyl=ether with oxyethylene units of 7.5 (PONPE-7.5). On the basis of the distribution curves for the respective chelates, the partition constants of the neutral chelates were determined at 293 K at an ionic strength of 0.1 (NaCl). The partition constants were considerably smaller than those expected from the regular solution theory. The partition constants were also dependent upon the kind of metal ions. This fact is probably due to such an ordered structure as liquid crystal in the surfactant phase which can differentiate the size of the metal chelates.
Similar content being viewed by others
References
M. J. Rosen, “Surfactants and Interfacial phenomena”, p. 139, Wiley-Interscience, New York (1978).
H. Ishii, J. Miura and H. Watanabe, Bunseki Kagaku, 26, 252 (1977).
H. Watanabe and H. Tanaka, Talanta, 25, 585 (1978).
S. Kawamorita, H. Watanabe and K. Haraguchi, Anal Sci., 1, 41 (1985).
H. Irving and H. S. Rossotti, J. Chem. Soc., 1953, 339.
K. Kiyokawa and A. Kawase, Bunseki Kagaku, 21, 244 (1972).
T. Sekine and Y. Hasegawa, “Solvent Extraction Chemistry”, p. 88, Marcel Dekker, New York (1977).
M. Tanaka, “Chemistry of Solvent Extraction”, p. 153, Kyoritsu Pub. Co., Tokyo (1977).
K. Kenjyo, Bull. Chem. Soc. Jpn., 39, 685 (1966)
H. Toh, Nippon Kagaku Kaishi, 1981, 1691.
Author information
Authors and Affiliations
Additional information
This study was supported by the Grant-in-Aid for Scientific Research (No. 58550481) from the Ministry of Education, Science and Culture, for which the authors are very grateful.
Rights and permissions
About this article
Cite this article
Watanabe, H., Kamidate, T., Kawamorita, S. et al. Distribution of Nickel(II), Cadmium(II) and Copper(II) Chelates of 2-(2-Pyridylazo)-5-methylphenol in Two Phases Separated from Micellar Solution of Nonionic Surfactant. ANAL. SCI. 3, 433–436 (1987). https://doi.org/10.2116/analsci.3.433
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.3.433