Abstract
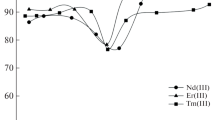
Solvent extraction of lanthanoid and yttrium ions(M3+) was investigated in a water-dichloroethane system using poly(oxyethylene)-type nonionic surfactants such as Triton X-100 and Triton X-405(L) and picrate ion(A-) as extraction agent and pairing anion, respectively. The result of extraction studies at various extraction agent concentrations and at various picrate ion concentrations, suggested that the composition of the extracted species was MLA3. Among the lanthanoid ions investigated, the ions such as Nd3+, Sm3+, Eu3+, and Gd3+ gave relatively larger distribution ratios than the other lanthanoid ions, while the yttrium ion was the least extractable among the metal ions investigated. The distribution ratio with extraction agent Triton X-405 was about ten times larger than those with Triton X-100.
Similar content being viewed by others
References
T. Sotobayashi, T. Suzuki and S. Tonouchi, Chem. Lett., 1976, 585.
T. Sotobayashi, T. Suzuki and K. Yamada, Chem. Lett., 1976, 77.
T. Sotobayashi, T. Suzuki and H. Kudo, J. Radioanal. Chem., 36, 145 (1977).
M. Otomo and Y. Wakamatsu, Bunseki Kagaku, 17, 764 (1968).
L. M. Tsay, J. S. Shih and S. C. Wu, Analyst [London], 108, 1108 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, I., Takeshita, R., Ueno, K. et al. Solvent Extraction of Lanthanoid and Yttrium Ions with Poly(oxyethylene)alkylphenylether. ANAL. SCI. 2, 53–56 (1986). https://doi.org/10.2116/analsci.2.53
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.2.53