Abstract
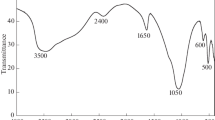
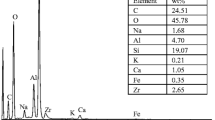
Zirconium phosphate, HZr2(PO4)3 (NZP), in a powder form was synthesized from zirconium chloride oxide and sodium dihydrogenphosphate, and its ion exchange adsorption properties to alkali metal ions were studied. The ion-exchange adsorption partition coefficients of NZP to alkali metal ions at pH=3.4 were found to be 3.2×103 (Li*), 4.5×105 (Na+), 2.6 (K+), 12 (Rb+), and 380 (Cs+) cm3 g−1. The results indicate that NZP has a high adsorbability to sodium ions. Mixed granules of NZP and silica sol (NZP-P) were then prepared and applied for the selective removal of sodium ions by a column adsorption method.
Similar content being viewed by others
References
J. Lehto, A. Paajanen, R. Harjula and H. Leinonen, React. Polym., 23, 135 (1994).
T. Nabarawy and S. E. Samra, Adsorption Sci. Technol., 11, 51 (1994).
N. Z. Misak, H. B. Maghrawy and I. M. Elnaggar, Solid State Ionics, 37, 1 (1989).
I. Tomita, K. Magami and H. Watanabe, K. Suzuki and T. Nakamura, Bull. Chem. Soc. Jpn., 56, 3183 (1983).
R. Llavona, J. R. Garcia, C. Aluarez, M. Suarez and J. Rodriguez, Solvent Extr. Ion. Exch., 4, 567 (1986).
S. Komarneni and R. Roy, Adv. Ceram., 20, 207 (1986).
E. Torracca, J. Inorg. Nucl. Chem., 31, 1189 (1969).
L. Kullberg and A. Clearfield, J. Inorg, Nucl Chem., 43, 2543 (1981).
N. Z. Misak and E. M. Mikhail, Z. Phys. Chem., 264, 995 (1983).
K. Ito, T. Suzuki, M. Kawaguchi and M. Oda, Adv. Ceram., 24, 1007 (1988).
A. Clearfield, Solid State Ionics, 46, 35 (1991).
H. A. Benesi, J. Phys. Chem., 61, 970 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakajima, Y., Yoshida, I. Sodium Selective Ion-Exchange Properties of Zirconium Phosphate, HZr2(PO4)3, and Its Application for the Removal of Sodium Ions. ANAL. SCI. 12, 935–940 (1996). https://doi.org/10.2116/analsci.12.935
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.12.935