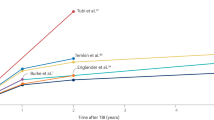
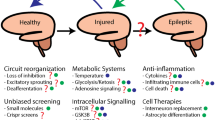
Abstract
Any attempt tomake antiepileptogenesis arealisticgoal in clinical trials should be based on the experience of failures of the past. A wide variety of experimental studies and clinical trials using chronic antiseizure drug therapy during the extended post-injury period have had minimal success. The disappointing results of these studies may be due to several factors including the possibility that antiseizure drugs, despite the fact they suppress seizure activity, do not interfere in any substantial way with the “epileptogenic” process of focal epilepsies. Although the reasons for the failure are not entirely clear, it may be that the antiseizure drugs may have been tested at the wrong doses, for the wrong duration, or at the wrong time after brain injury. Surprisingly, the anti-absence drug ethosuximide has also been shown to be antiepileptogenic in several experimental models of absence epilepsy. In addition, clinical trials aimed at preventing focal post-injury epilepsy have suffered from poor enrolment and other issues related to the comorbidity of severe epilepsies that follow overt brain injury. Testing specific anti-inflammatory and immunological antiepileptogenic agents to prevent focal epilepsies, as well as prevention trials for genetic epilepsies, possibly with anti-absence drugs, may be a way to resolve the dilemma. Although more evidence is needed, there is hope on the horizon for antiepileptogenic therapy that works.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baulac M, Pitkänen A. Research priorities in epilepsy for the next decade. A representative view of the European scientific community: summary of the ILAE Epilepsy Research Workshop, Brussels, 17–18 January 2008. Epilepsia 2009; 50: 571–578.
Beghi E. Overview of studies to prevent posttraumatic epilepsy. Epilepsia 2003; 44: 21–26.
Blumenfeld H, Klein JP, Schridde U, et al. Early treatment suppresses the development of spike-wave epilepsy in a rat model. Epilepsia 2008; 49: 400–409.
Blumenfeld H. New strategies for preventing epileptogenesis: perspective and overview. Neurosci Lett 2011; 497: 153–154.
Brodie M, Covanis T, Gil-Nagel A, et al. Antiepileptic drug therapy: does mechanism of action matter? Epilepsy Behav 2011; 21: 490.
Buckmaster PS, Lew FH. Rapamycin suppresses mossy fiber sprouting but not seizure frequency in a mouse model of temporal lobe epilepsy. J Neurosci 2011; 31: 2337–2347.
Choi J, Koh S. Role of brain inflammation in epileptogenesis. Yonsei Med J 2008; 49: 1–18.
Christensen J, Pedersen MG, Pedersen CB, Sidenius P, Olsen J, Vestergaard M. Long-term risk of epilepsy after traumatic brain injury in children and young adults: a population-based cohort study. Lancet 2009; 373: 1105–1110.
Fisher M, Feuerstein G, Howells DW, et al. Update of the stroke therapy academic industry roundtable preclinical recommendations. Stroke 2009; 40: 2244–2250.
Galanopoulou AS, Buckmaster P, Staley K, et al. Identification of new treatments for epilepsy: issues in preclinical methodology. Epilepsia 2012; 53: 571–582.
Hauser WA, Hesdorffer DC. Epilepsy: frequency, causes and consequences. Landover (MD); NewYork (NY): Epilepsy Foundation of America, 1990.
Herman ST. Clinical trials for prevention of epileptogenesis. Epilepsy Res 2006; 68: 35–38.
Ji-qun C, Ishihara K, Nagayama T, Serikawa T, Sasa M. Longlasting antiepileptic effects of levetiracetam against epileptic seizures in the spontaneously epileptic rat (SER): differentiation of levetiracetam from conventional antiepileptic drugs. Epilepsia 2005; 46: 1362–1370.
Jozwiak S, Kotulska K, Domanska-Pakiela D, et al. Antiepileptic treatment before the onset of seizures reduces epilepsy severity and risk of mental retardation in infants with tuberous sclerosis complex. Eur J Paediatr Neurol 2011; 15: 424–431.
Kelley MS, Jacobs MP, Lowenstein DH; NINDS Epilepsy Benchmark Stewards. The NINDS epilepsy research benchmarks. Epilepsia 2009; 50: 579–82.
Kharatishvili I, Pitkänen A. Posttraumatic epilepsy. Curr Opin Neurol 2010; 23: 183–188.
Langer M, Brandt C, Zellinger C, Läncher W. Therapeutic window of opportunity for the neuroprotective effect of valproate versus the competitive AMPA receptor antagonist NS1209 following status epilepticus in rats. Neuropharmacology 2011; 61: 1033–1047.
Löscher W, Brandt C. Prevention or modification of epileptogenesis after brain insults: experimental approaches and translational research. Pharmacol Rev 2010; 62: 668–700.
Löscher W, Hönack D, Rundfeldt C. Antiepileptogenic effects of the novel anticonvulsant levetiracetam (ucb L059) in the kindling model of temporal lobe epilepsy. J Pharmacol Exp Ther 1998; 284: 474–479.
Luat AF, Chugani HT. Molecular and diffusion tensor imaging of epileptic networks. Epilepsia 2008; 49: 15–22.
Lux AL, Edwards SW, Hancock E, et al. The United Kingdom Infantile Spasms Study (UKISS) comparing hormone treatment with vigabatrin on developmental and epilepsy outcomes to age 14 months: a multicentre randomised trial. Lancet Neurol 2005; 4: 712–717.
Mani R, Pollard J, Dichter MA. Human clinical trials in antiepileptogenesis. Neurosci Lett 2011; 497: 251–256.
Pitkänen A. Therapeutic approaches to epileptogenesis-hope on the horizon. Epilepsia 2010; 51: 2–17.
Pitkänen A, Lukasiuk K. Mechanisms of epileptogenesis and potential treatment targets. Lancet Neurol 2010; 10: 173–186.
Russo E, Citraro R, Scicchitano F, et al. Comparison of the antiepileptogenic effects of an early long-term treatment with ethosuximide or levetiracetam in a genetic animal model of absence epilepsy. Epilepsia 2010; 51: 1560–1569.
Schierhout G, Roberts I. Antiepileptic drugs for preventing seizures following acute traumatic brain injury. Cochrane Database Syst Rev 2001; (4): CD000173. doi: 10.1002/14651858.CD000173
Schmidt D, Löscher W. Uncontrolled epilepsy following discontinuation of antiepileptic drugs in seizure-free patients: a review of current clinical experience. Acta Neurol Scand 2005; 111: 291–300.
Sillanpää M, Schmidt D. Prognosis of seizure recurrence after stopping antiepileptic drugs in seizure-free patients: A long-term population-based study of childhood-onset epilepsy. Epilepsy Behav 2006a; 8: 713–719.
Sillanpää M, Schmidt D. Natural history of treated childhood-onset epilepsy: prospective, long-term population-based study. Brain 2006b; 129: 617–624.
Sirven JI, Wingerchuk DM, Drazkowski JF, Lyons MK, Zimmerman RS. Seizure prophylaxis in patients with brain tumors: a meta-analysis. Mayo Clin Proc 2004; 79: 1489–1494.
Sloviter RS. Progress on the issue of excitotoxic injury modification vs. real neuroprotection; implications for posttraumatic epilepsy. Neuropharmacology 2011; 61: 1048–1050.
Temkin NR. Preventing and treating posttraumatic seizures: the human experience. Epilepsia 2009; 50: 10–13.
van Tuijl JH, van Raak EP, de Krom MC, Lodder J, Aldenkamp AP. Early treatment after stroke for the prevention of late epileptic seizures: a report on the problems performing a randomised placebo-controlled double-blind trial aimed at anti-epileptogenesis. Seizure 2011; 20: 285–291.
Vezzani A, French J, Bartfai T, Baram TZ. The role of inflammation in epilepsy. Nat Rev Neurol 2011; 7: 31–40.
Walker MC, White HS, Sander JW. Disease modification in partial epilepsy. Brain 2002; 125: 1937–1950.
Wong M. Rapamycin for treatment of epilepsy: antiseizure, antiepileptogenic, both, or neither? Epilepsy Curr 2011; 11: 66–68.
Yan HD, Ji-qun C, Ishihara K, Nagayama T, Serikawa T, Sasa M. Separation of antiepileptogenic and antiseizure effects of levetiracetam in the spontaneously epileptic rat (SER). Epilepsia 2005; 46: 1170–1177.
Author information
Authors and Affiliations
Corresponding author
Additional information
Updated following presentation and discussion at the 2011 Progress in Epileptic Disorders Workshop on “Antiepileptic Drug Trials: will the future challenge the past” held at the Chaeauform’ La Maison des Contes, Dareizé, 69490, France. The workshop was partly supported by an educational grant from UCB. The program was under the exclusive responsibility of a Scientific Committee composed by Prs. Philippe Ryvlin (France), Emilio Perucca (Italy), Jackie French (USA), Steve White (USA), Graeme Sills (UK) and Alexis Arzimanoglou (France).
About this article
Cite this article
Schmidt, D. Is antiepileptogenesis a realistic goal in clinical trials? Concerns and new horizons. Epileptic Disord. 14, 105–113 (2012). https://doi.org/10.1684/epd.2012.0512
Published:
Issue Date:
DOI: https://doi.org/10.1684/epd.2012.0512