Abstract
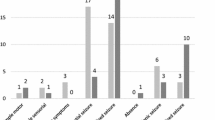
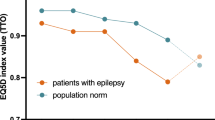
This prospective, multicenter, non-interventional surveillance study (ZADE study) explored seizure outcome and tolerability of adjunctive treatment with zonisamide (ZNS) in a non-selected sample of patients with partial-onset seizures in everyday clinical practice. Changes in quality of life (QOL) and health status were also recorded. Clinical status was assessed before and 4 months after introduction of ZNS. The herein reported evaluation of QOL and health status was based on a representative subsample of 207 patients. In this subgroup, a reduced QOL had been apparent in 68% of patients at baseline. After introduction of ZNS, all measures improved, with ameliorations in QOL in up to 35% of patients. Major determinants for a better QOL outcome were (1) a better score at baseline, (2) a higher degree of seizure reduction, and (3) a lower number of concomitant AEDs. Tolerability was subjectively rated as good by 89% of patients. With a ZNS dose of 244.8±108 mg/d at study end, seizure frequency had dropped from 8.8±19.2 within 8 weeks before baseline to 3.6±9.1 seizures within the period of 8 weeks before study end. A total of 79% of patients responded to ZNS treatment with a≥50% reduction in seizure frequency; 34% became seizure free. In conclusion, adjunctive treatment with ZNS seems to be efficacious and well tolerated. QOL improvement was predicted by baseline score, seizure outcome, and overall drug load, and is thus more likely a result of enhanced seizure control, rather than an intrinsic psychotropic effect of zonisamide.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aldenkamp AP, van Meel HF, Baker GA, Brooks J, Hendriks MP. The A-B neuropsychological assessment schedule (ABNAS): the relationship between patient-perceived drug related cognitive impairment and results of neuropsychological tests. Seizure 2002; 11: 231–237.
Arif H, Buchsbaum R, Weintraub D, Pierro J, Resor SR Jr, Hirsch LJ. Patient-reported cognitive side effects of antiepileptic drugs: predictors and comparison of all commonly used antiepileptic drugs. Epilepsy Behav 2009; 14: 202–209.
Baker G, Frances P, Middleton E. Initial development, reliability, and validity of a patient-based adverse events scale. Epilepsia 1994; 35: 80.
Biton V. Clinical pharmacology and mechanism of action of zonisamide. Clin Neuropharmacol 2007; 30: 230–240.
Brodie MJ, Duncan R, Vespignani H, Solyom A, Bitenskyy V, Lucas C. Dose-dependent safety and efficacy of zonisamide: a randomized, double-blind, placebo-controlled study in patients with refractory partial seizures. Epilepsia 2005; 46: 31–41.
Brooks J, Baker GA, Aldenkamp AP. The A-B neuropsychological assessment schedule (ABNAS): the further refinement of a patient-based scale of patient-perceived cognitive functioning. Epilepsy Res 2001; 43: 227–237.
Commission on Classification and Terminology of the International League Against Epilepsy. Proposal for Revised Classification of Epilepsies and Epileptic Syndromes. Epilepsia 1989; 30: 389–399.
Cramer JA, Perrine K, Devinsky O, Meador K. Abrief questionnaire to screen for quality of life in epilepsy: the QOLIE-10. Epilepsia 1996; 37: 577–582.
Dupont S, Striano S, Trinka E, et al. Flexible dosing of adjunctive zonisamide in the treatment of adult partial-onset seizures: a non-comparative, open-label study (ZEUS). Acta Neurol Scand 2010; 121: 141–148.
Faught E, Ayala R, Montouris GG, Leppik IE. Randomized controlled trial of zonisamide for the treatment of refractory partial-onset seizures. Neurology 2001; 57: 1774–1779.
Gillham R, Baker G, Thompson P, et al. Standardisation of a self-report questionnaire for use in evaluating cognitive, affective and behavioural side-effects of anti-epileptic drug treatments. Epilepsy Res 1996; 24: 47–55.
Gillham R, Bryant-Comstock L, Kane K. Validation of the side effect and life satisfaction (SEALS) inventory. Seizure 2000; 9: 458–463.
Helmstaedter C, Witt JA. Cognitive outcome of antiepileptic treatment with levetiracetam versus carbamazepine monotherapy: a non-interventional surveillance trial. Epilepsy Behav 2010; 18: 74–80.
Helmstaedter C, Witt JA. The effects of levetiracetam on cognition: a non-interventional surveillance study. Epilepsy Behav 2008; 13: 642–649.
Hoppe C, Elger CE, Helmstaedter C. Long-term memory impairment in patients with focal epilepsy. Epilepsia 2007; 48: 26–29.
Marino SE, Meador KJ, Loring DW, et al. Subjective perception of cognition is related to mood and not performance. Epilepsy Behav 2009; 14: 459–464.
Park SP, Hwang YH, Lee HW, Suh CK, Kwon SH, Lee BI. Longterm cognitive andmoodeffects of zonisamide monotherapy in epilepsy patients. Epilepsy Behav 2008; 12: 102–108.
Sackellares JC, Ramsay RE, Wilder BJ, Browne TR 3rd, Shellenberger MK. Randomized, controlled clinical trial of zonisamide as adjunctive treatment for refractory partial seizures. Epilepsia 2004; 45: 610–617.
Salinsky MC, Storzbach D. The Portland Neurotoxicity Scale: validation of a brief self-report measure of antiepileptic-drugrelated neurotoxicity. Assessment 2005; 12: 107–117.
Schmidt D, Jacob R, Loiseau P, et al. Zonisamide for add-on treatment of refractory partial epilepsy: a European doubleblind trial. Epilepsy Res 1993; 15: 67–73.
Schulze-Bonhage A. Zonisamide in the treatment of epilepsy. Expert Opin Pharmacother 2010; 11: 115–126.
Sills G, Brodie M. Pharmacokinetics and drug interactions with zonisamide. Epilepsia 2007; 48: 435–441.
Stefan H, Bauer B, Kockelmann E, Helmstaedter C. Zonisamide in everyday clinical practice: Results from the non-interventional ZADE study. Poster presented at Neurowoche 2010, Mannheim, Germany.
White JR, Walczak TS, Marino SE, Beniak TE, Leppik IE, Birnbaum AK. Zonisamide discontinuation due to psychiatric and cognitive adverse events: a case-control study. Neurology 2010; 75: 513–518.
Zonegran® Summary of Product Characteristics, March 2010. Eisai Ltd., Hatfield, UK.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Helmstaedter, C., Stefan, H. & Witt, JA. Quality of life in patients with partial-onset seizures under adjunctive therapy with zonisamide: results from a prospective non-interventional surveillance study. Epileptic Disord 13, 263–276 (2011). https://doi.org/10.1684/epd.2011.0459
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/epd.2011.0459