Abstract
Background
Combination of a photosensitizer, 5-aminolevulinic acid (ALA), with photodynamic therapy (PDT) has been widely used to treat skin squamous cell carcinoma (SCC). However, a portion of SCC patients do not respond well to PDT. The molecular reason for this resistance is not clear. We hypothesize that mitogen-activated phosphorylation kinase (MAPK) plays a key role in mediating SCC resistance to PDT.
Objectives
To determine whether inhibition of MAPK signaling enhances the anti-tumor effect of ALA-PDT in SCC.
Material and methods
The human squamous carcinoma cell line, SCL-1, was either untreated or treated with various combinations of ALA, PDT light source and inhibitors of MAPK signaling components.
Results
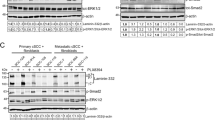
ALA-PDT treatment significantly decreased cell viability, increased the percentage of annexin-V positive cells and resulted in formation of apoptotic bodies. ALA-PDT treated cells showed increased levels of p-MEK, p-ERK1/2, p-p38, p-Elk-1, p-JNK and p-c-Jun. Addition of inhibitors for ERK1/2 (PD98059), p38 (SB203580) and JNK (SP60125) reversed the changes and led to a more dramatic decrease in SCL-1 cell viability than seen with ALA-PDT alone.
Conclusion
Inhibition of the MAPK pathway enhances the cytotoxic effect of ALA–PDT on SCL-1.
Similar content being viewed by others
References
Linares MA, Zakaria A, Nizran P. Skin Cancer. Prim Care 2015; 42: 645–59.
Jiao YN, Xia L, Ge XH. The incidence analysis of skin common tumor from five hospitals in Ningxia from 2002 to 2011. Chi J Dermatovenereol 2013; 27: 36–8.
Celli JP, Spring BQ, Rizvi I, et al. Imaging and photodynamic therapy: mechanisms, monitoring, and optimization. Chem Rev 2010; 110: 2795–838.
Gilbert DJ. Incorporating photodynamic therapy into a medical and cosmetic dermatology practice. Dermatol Clin 2007; 25: 111–8.
Dijkstra AT, Majoie IM, van Dongen JW, van WH, van Vloten WA. Photodynamic therapy with violet light and topical 6-aminolaevulinic acid in the treatment of actinic keratosis, Bowen’s disease and basal cell carcinoma. J Eur Acad Dermatol Venereol 2001; 15: 550–4.
Morton C, Szeimies RM, Sidoroff A, et al. European Dermatology Forum Guidelines on topical photodynamic therapy. Eur J Dermatol 2015; 25: 296–311.
Calzavara-Pinton PG, Rossi MT, Sala R. A retrospective analysis of real-life practice of off-label photodynamic therapy using methyl aminolevulinate (MAL-PDT) in 20 Italian dermatology departments. Part 2: oncologic and infectious indications. Photochem Photobiol Sci 2013; 12: 158–65.
Wu RW, Yow CM,Wong CK, Lam YH. Photodynamic therapy (PDT) -Initiation of apoptosis via activation of stress-activated p38 MAPK and JNK signal pathway in H460 cell lines. Photodiagnosis Photodyn Ther 2011; 8: 254–63.
Qiu JW, Guo W, Shen LJ. Advances In research on p38 MAPK-related hepatocellular carcinoma. World Chin J Digestol 2009; 16: 503–9.
Umbrasaite J, Schweighofer A, Kazanaviciute V, et al. MAPK Phosphatase AP2C3 Induces Ectopic Proliferation of Epidermal Cells Leading to Stomata Development in Arabidopsis. PLoS One 2010; 5: 1–18.
Nix P, Hisamoto N, Matsumoto K, Bastiani M. Axon regeneration requires coordinate activation of p38 and JNK MAPK pathways. Proc Natl Acad Sci USA 2011; 108: 10738–43.
Shen SJ, Guo JC. The role of MAPK signaling pathway in tumor drug resistance based on chemotherapy. Foreign Medical Sciences. Oncologyr Section 2005; 32: 579–82.
Ge X, Tang Z, Jiao YE. The expression of EGFR and MAPK in actinic keratosis and skin squamous cell carcinoma. Chin J Dermatol 2015; 48: 8–11.
Rubio N, Verrax J, Dewaele M, et al. p38(MAPK)-regulated induction of p62 and NBR1 after photodynamic therapy promotes autophagic clearance of ubiquitin aggregates and reduces reactive oxygen species levels by supporting Nrf2-antioxidant signaling. Free Radic Biol Med 2014; 67: 292–303.
Peng Q, Warloe T, Moan J, et al. Antitumor effect of 5-aminolevulinic acid-mediated photodynamic therapy can be enhanced by the use of a low dose of photofrin in human tumor xenografts. Cancer Res 2001; 61: 5824–32.
Niedre M, Patterson MS, Wilson BC. Direct nearinfrared luminescence detection of singlet oxygen generated by photodynamic therapy in cells in vitro and tissues in vivo. Photochem Photobiol 2002; 75: 382–91.
Haddad R, Kaplan O, Greenberg R, Siegal A, Skornick Y, Kashtan H. Photodynamic therapy of murine colon cancer and melanoma using systemic aminolevulinic acid as a photosensitizer. Int J Surg Investig 2000; 2: 171–8.
Lioni M, Noma K, Snyder A, et al. Bortezomib induces apoptosis in esophageal squamous cell carcinoma cells through activation of the p38 mitogen-activated protein kinase pathway. Mol Cancer Ther 2008; 7: 2866–75.
Tong Z, Singh G, Rainbow AJ. Sustained activation of the extracellular signal-regulated kinase pathway protects cells from photofrin-mediated photodynamic therapy. Cancer Res 2002; 62: 5528–35.
Dodeller F, Schulze KH. The p38 mitogen-activated protein kinase signaling cascade in CD4 T cells. Arthritis Res Ther 2006; 8: 205.
Li ZH. JNK signal pathway. Int J Pathol Clin Med 2010; 30: 273–6.
Chen YR, Meyer CF, Tan TH. Persistent activation of c-Jun Nterminal kinase 1 (JNK1) in gamma radiation-induced apoptosis. J Biol Chem 1996; 271: 631–4.
Gilaberte Y, Milla L, Salazar N, et al. Cellular intrinsic factors involved in the resistance of squamous cell carcinoma to photodynamic therapy. J Invest Dermatol 2014; 134: 2428–37.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ge, X., Liu, J., Shi, Z. et al. Inhibition of MAPK signaling pathways enhances cell death induced by 5-Aminolevulinic acid-photodynamic therapy in skin squamous carcinoma cells. Eur J Dermatol 26, 164–172 (2016). https://doi.org/10.1684/ejd.2015.2725
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2015.2725