Abstract
Background
Current parameters for assessing the efficacy of actinic keratosis (AK) treatments compare clinical lesions at the start and end of a study. However, the sun-exposed field also contains subclinical lesions which may become detectable during treatment. Lmax, the maximum lesion count during treatment, is a new concept to better assess the efficacy of field-directed AK therapies. Measuring efficacy using the reduction in lesions from Lmax includes for the first time the clearance of both subclinical and clinical lesions.
Objectives
To evaluate the reduction of lesions from Lmax to study end and compare the results with traditional efficacy endpoints using imiquimod 3.75% (IQ3.75%) as an example of field-directed AK therapy.
Materials & Methods
Pooled analysis of data from two 14-week, vehicle-controlled, double-blind studies of IQ3.75%.
Results
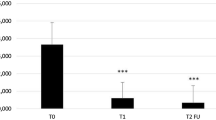
With IQ3.75%, the median number of lesions increased from 10 at baseline to an Lmax of 22. The median absolute reduction in lesions to study end was 18 from Lmax versus 7 from baseline. The median percentage reduction in AK lesions to study end was 92.2% from Lmax compared with 81.8% from baseline.
Conclusions
The reduction in lesion count from Lmax is a novel efficacy parameter that should become the new way of evaluating field-directed AK therapies since it enables their efficacy against both clinical and subclinical lesions to be accurately determined. Together, the Lmax concept and IQ3.75% represent a newapproach for the management ofAKacross a large sun-exposed field.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Stockfleth E, Ortonne JP, Alomar A. Actinic keratosis and field cancerisation. Eur J Dermatol 2011; 21(Suppl 1): 3–12.
Ortonne JP, Gupta G, Ortonne N, Duteil L, Queille C, Mallefet P. Effectiveness of cross polarized light and fluorescence diagnosis for detection of sub-clinical and clinical actinic keratosis during imiquimod treatment. Exp Dermatol 2010; 19: 641–647.
Ulrich M, Krueger-Corcoran D, Roewert-Huber J, Sterry W, Stockfleth E, Astner S. Reflectance confocal microscopy for noninvasive monitoring of therapy and detection of subclinical actinic keratoses. Dermatology 2010; 220: 15–24.
Rowert-Huber J, Patel MJ, Forschner T, et al. Actinic keratosis is an early in situ squamous cell carcinoma: a proposal for reclassification. Br J Dermatol 2007; 156(Suppl 3): 8–12.
Goldberg LH, Mamelak AJ. Review of actinic keratosis. Part I: etiology, epidemiology and clinical presentation. J Drugs Dermatol 2010; 9: 1125–1132.
Cockerell CJ. Pathology and pathobiology of the actinic (solar) keratosis. Br J Dermatol 2003; 149(Suppl 66): 34–36.
Feldman SR, Fleischer AB Jr. Progression of actinic keratosis to squamous cell carcinoma revisited: clinical and treatment implications. Cutis 2011; 87: 201–207.
Stockfleth E, Ferrandiz C, Grob JJ, Leigh I, Pehamberger H, Kerl H. Development of a treatment algorithm for actinic keratoses: a European Consensus. Eur J Dermatol 2008; 18: 651–659.
Berman B, Amini S. Pharmacotherapy of actinic keratosis: an update. Expert Opin Pharmacother 2012; 13: 1847–1871.
Wolf JE Jr., Taylor JR, Tschen E, Kang S. Topical 3.0% diclofenac in 2.5% hyaluronan gel in the treatment of actinic keratoses. Int J Dermatol 2001; 40: 709–713.
Rivers JK, Arlette J, Shear N, Guenther L, Carey W, Poulin Y. Topical treatment of actinic keratoses with 3.0% diclofenac in 2.5% hyaluronan gel. Br J Dermatol 2002; 146: 94–100.
Krawtchenko N, Roewert-Huber J, Ulrich M, Mann I, Sterry W, Stockfleth E. A randomised study of topical 5% imiquimod vs. topical 5-fluorouracil vs. cryosurgery in immunocompetent patients with actinic keratoses: a comparison of clinical and histological outcomes including 1-year follow-up. Br J Dermatol 2007; 157(Suppl 2): 34–40.
Gupta AK, Davey V, Mcphail H. Evaluation of the effectiveness of imiquimod and 5-fluorouracil for the treatment of actinic keratosis: Critical review and meta-analysis of efficacy studies. J Cutan Med Surg 2005; 9: 209–214.
Szeimies RM, Karrer S, Radakovic-Fijan S, et al. Photodynamic therapy using topical methyl 5-aminolevulinate compared with cryotherapy for actinic keratosis: A prospective, randomized study. J Am Acad Dermatol 2002; 47: 258–262.
Piacquadio DJ, Chen DM, Farber HF, et al. Photodynamic therapy with aminolevulinic acid topical solution and visible blue light in the treatment of multiple actinic keratoses of the face and scalp: investigator-blinded, phase 3, multicenter trials. Arch Dermatol 2004; 140: 41–46.
Lebwohl M, Swanson N, Anderson LL, Melgaard A, Xu Z, Berman B. Ingenol mebutate gel for actinic keratosis. N Engl J Med 2012; 366: 1010–1019.
Swanson N, Abramovits W, Berman B, Kulp J, Rigel DS, Levy S. Imiquimod 2.5% and 3.75% for the treatment of actinic keratoses: results of two placebo-controlled studies of daily application to the face and balding scalp for two 2-week cycles. J Am Acad Dermatol 2010; 62: 582–590.
Gupta AK, Cooper EA, Abramovits W. Zyclara (imiquimod) cream, 3.75%. Skinmed 2010; 8: 227–229.
Quist SR, Gollnick HP. Imiquimod 3.75% cream (Zyclara) for the treatment of actinic keratoses. Expert Opin Pharmacother 2011; 12: 451–461.
Gaspari AA. Mechanism of action and other potential roles of an immune response modifier. Cutis 2007; 79: 36–45.
Sauder DN. Immunomodulatory and pharmacologic properties of imiquimod. J Am Acad Dermatol 2000; 43: S6–11.
Schon M, Bong AB, Drewniok C, et al. Tumor-selective induction of apoptosis and the small-molecule immune response modifier imiquimod. J Natl Cancer Inst 2003; 95: 1138–1149.
Dodson JM, DeSpain J, Hewett JE, Clark DP. Malignant potential of actinic keratoses and the controversy over treatment. A patientoriented perspective. Arch Dermatol 1991; 127: 1029–1031.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Stockfleth, E., Gupta, G., Peris, K. et al. Reduction in lesions from Lmax: a new concept for assessing efficacy of field-directed therapy for actinic keratosis. Results with imiquimod 3.75%. Eur J Dermatol 24, 23–27 (2014). https://doi.org/10.1684/ejd.2014.2265
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2014.2265