Abstract
Introduction
This study aimed to compare the accuracy of selected laboratory markers in assessing disease activity in patients with ulcerative colitis (UC). The analysis included serum IL-2, IL-4, IL-6, IL-10, IL-17, TNF-α, IFN-γ, hsCRP, peripheral regulatory T cells, as well as fecal calprotectin and lactoferrin.
Patients and methods
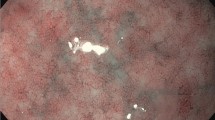
A group of 45 adults with UC was enrolled in the study. Disease activity was assessed using the Mayo endoscopic index, while for clinical activity scoring, the Clinical Activity Index (CAI) was used. Concentrations of markers investigated were estimated by means of flow cytometry and enzyme-linked immunosorbent assays: the results were correlated with both indices.
Results
The study demonstrated that both fecal markers, i.e. calprotectin (r = 0.880, P<0.001) and lactoferrin (r = 0.799, P<0.001) correlated closely with the Mayo endoscopic score, and might be used to evaluate the severity of UC in the clinical setting. The correlation of these markers with CAI was also significant, with r = 0.831 for calprotectin (P<0.001) and r = 0.672 for lactoferrin (P<0.05). As for the other markers investigated, only IL-6 (r = 0.598, P<0.001), IL-17A (r = 0.587, P<0.005), and TNF-α (r = 0.701, P<0.001) correlated closely with the Mayo endoscopic index. The correlation of the markers with CAI was also significant, though weaker, with r = 0.525 for IL-6 (P<0.001), r = 0.587 for IL-17A (P<0.05), and r = 0.624 for TNF-α (P<0.001).
Discussion
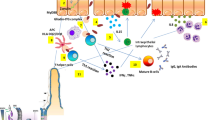
Despite the fact, that UC is generally considered to be an IL-13-driven, Th2-like type of disease, markers of inflammation such as serum interleukin (IL)-6, IL-17, TNF-α, fecal calprotectin and lactoferrin might be useful in assessing disease activity.
Similar content being viewed by others
References
Schoepfer AM, Vavricka S, Zahnd-Straumann N, Straumann A, Beglinger C. Monitoring inflammatory bowel disease activity: clinical activity is judged to be more relevant than endoscopic severity or biomarkers. Journal of Crohn’s and Colitis 2012; 6: 412–8.
Cooney RM, Warren BF, Altman DG, Abreu MT, Travis SP. Outcome measurement in clinical trials for Ulcerative Colitis: towards standardisation. Trials 2007; 8: 17.
Satsangi J, Silverberg MS, Vermeire S, Colombel JF. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut 2006; 55: 749–53.
Sartor RB. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nature Clinical Practice Gastronetrology and Hepatology 2006: 390–407.
Fuss IJ, Strober W. The role of IL-13 and NK T cells in experimental and human ulcerative colitis. Mucosal Immunology 2008; 1(Suppl 1): s31–3.
Waldenr MJ, Neurath MF. Master regulator of intestinal disease: IL-6 in chronic inflammation and cancer development. Semminars in Immunology 2014. doi: 10.1016/j.smim.2013.12.003.
Naugler WE, Karin M. The wolf in sheep’s clothing: the role of interleukin-6 in immunity, inflammation and cancer. Trends Iin Molecular Medicine 2008; 14: 109–19.
Neurath MF, Finotto S. IL-6 signaling in autoimmunity, chronic inflammation and inflammation-associated cancer. Cytokine & Growth Factor Reviews 2011; 22: 83–9.
Hyams JS, Fitzgerald JE, Treem WR, Wyzga N, Kreutzer DL. Relationship of functional and antigenic interleukin 6 to disease activity in inflammatory bowel disease. Gastroenterology 1993; 103: 1285–92.
Mahida YR, Kurlac L, Gallagher A, Hawkey CJ. High circulating concentrations of interleukin-6 in active Crohn’s disease but not ulcerative colitis. Gut 1991; 32: 1531–4.
Abraham C, Cho J. Interleukin-23/Th17 pathways and inflammatory bowel disease. Inflammatory Bowel Disease 2009; 15: 1090–100.
Brand S. Crohn’s disease: Th1, Th17 or both? The change of a paradigm: new immunological and genetic insights implicate Th17 cells in the pathogenesis of Crohn’s disease. Gut 2009; 58: 1152–67.
Fujino S, Andoh A, Bamba S, Ogawa A, Hata K, Araki Y, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut 2003; 52: 75–80.
Ohman L, Dahlén R, Isaksson S, Sjöling A, Wick MJ, Sjövall H, et al. Serum IL-17A in newly diagnosed treatment-naive patients with ulcerative colitis reflects clinical disease severity and predicts the course of disease. Inflammatory Bowel Disease 2013; 19: 2433–9.
Jiang S. Th17 cells in health and disease. 1 ed: Springer Science+ Business Media, LLC; 2011.
Karczewski J, Mazur M, Karczewski M. Dual role of Th17 cells in Crohn’s disease. Central European Journal of Immunology. 2011.
Sanchez-Munoz F, Dominguez-Lopez A, Yamamoto-Furusho JK. Role of cytokines in inflammatory bowel disease. World Journal of Gastroenetrology 2008; 14: 4280–8.
Eastaff-Leung N, Mabarrack N, Barbour A, Cummins A, Barry S. Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. Journal of Clinical Immunology 2010; 30: 80–9.
Karczewski J, Karczewski M. Possible defect of regulatory T cells in patients with ulcerative colitis. Central European Journal of Immunology 2011; 36: 254–5.
Willrich MA, Murray DL, Snyder MR. Tumor necrosis factor inhibitors: clinical utility in autoimmune diseases. Translational Research 2015; 165: 270–82.
Colombel JF, Sandborn WJ, Ghosh S, Wolf DC, Panaccione R, Feagan B, et al. Four-year maintenance treatment with adalimumab in patients with moderately to severely active ulcerative colitis: Data from ULTRA 1, 2, and 3. American Journal of Gastroenterology 2013; 109: 1771–80.
Bradley JR. TNF-mediated inflammatory disease. Journal of Pathology 2008; 214: 149–60.
Targan SR, Hanauer SB, van Deventer SJ, Mayer L, Present DH, Braakman T, et al. A short term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn’s disease. Crohn’s Disease cA2 study Group. NewEngland Journal of Medicine 1997; 337: 1029–35.
Papadakis KA, Targan SR. Role of cytokines in the pathogenesis of inflammatory bowel disease. Annual Review of Medicine 2000; 51: 289–98.
Braegger CP, Nicholls S, Murch SH, Stephens S, MacDonald TT. Tumour necrosis factor alpha in stool as a marker of intestinal inflammation. Lancet 1992; 339: 89–91.
Maeda M, Watanabe N, Neda H, Yamauchi N, Okamoto T, Sasaki H, et al. Serum tumor necrosis factor activity in inflammatory bowel disease. Immunopharmacology and Immunotoxicology 1992; 14: 451–61.
Ford ES, Giles WH, Myers GL, Rifai N, Ridker PM, Mannino DM. C-reactive protein concentration distribution among US children and young adults: findings from the National Health and Nutrition Examination Survey, 1999-2000. Clinical Chemistry and Laboratory Medicine 2003; 49: 1353–7.
Tall AR. C-reactive protein reassessed. New England Journal of Medicine 2004; 350: 1450–2.
Du Clos TW. Function of C-reactive protein. Annals of Medicine 2000; 32: 274–8.
Vermeire S, Van Assche G, Rutgeerts P. C-reactive protein as a marker for inflammatory bowel disease. Inflammatory Bowel Disease 2004; 10: 661–5.
Henriksen M, Jahnsen J, Lygren I, Stray N, Sauar J, Vatn MH, et al. C-reactive protein: a predictive factor and marker of inflammation in inflammatory bowel disease. Results from a prospective populationbased study. Gut 2008; 57: 1518–23.
Bjerke K, Halstensen TS, Jahnsen F, Pulford K, Brandtzaeg P. Distribution of macrophages and granulocytes expressing L1 protein (calprotectin) in human Peyer’s patches compared with normal ileal lamina propria and mesenteric lymph nodes. Gut 1993; 34: 1357–63.
Konikoff MR, Denson LA. Role of fecal calprotectin as a biomarker of intestinal inflammation in inflammatory bowel disease. Inflammatory Bowel Disease 2006; 12: 524–34.
Kayazawa M, Saitoh O, Kojima K, Nakagawa K, Tanaka S, Tabata K, et al. Lactoferrin in whole gut lavage fluid as a marker for disease activity in inflammatory bowel disease: comparison with other neutrophil-derived proteins. American Journal of Gastroenterology 2002; 97: 360–9.
Annese V, Daperno M, Rutter MD, Amiot A, Bossuyt P, East J, et al. European evidence based consensus for endoscopy in inflammatory bowel diseas. Journal of Crohn’s and Colitis 2013; 7: 982–1018.
Rachmilewitz D. Coated mesalazine (5-aminosalicylic acid) versus sulphasalazine in the treatment of active ulcerative colitis: a randomised trial. BMJ 1989; 298: 82–6.
Røseth AG, Aadland E, Grzyb K. Normalization of faecal calprotectin: a predictor of mucosal healing in patients with inflammatory bowel disease. Scandinavian Journal of Gastroenterology 2004; 39: 1017–20.
D’Haens G, Ferrante M, Vermeire S, Baert F, Noman M, Moortgat L, et al. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflammatory Bowel Disease 2012; 18: 2218–24.
Sipponen T, Savilahti E, Kolho KL, Nuutinen H, Turunen U, Färkkilä M. Crohn’s disease activity assessed by fecal calprotectin and lactoferrin: correlation with Crohn’s disease activity index and endoscopic findings. Inflammatory Bowel Disease 2008; 14: 40–6.
Karczewski J, Swora-Cwynar E, Rzymski P, Poniedzialek B, Adamski Z. Selected biologic markers of inflammation and activity of Crohn’s disease. Autoimmunity 2015. doi: 10.3109/08916934.2015.1016221 (Epub ahead of print).
Otten CM, Kok L, Witteman BJ, Baumgarten R, Kampman E, Moons KG, et al. Diagnostic performance of rapid tests for detection of fecal calprotectin and lactoferrin and their ability to discriminate inflammatory from irritable bowel syndrome. Clinical Chemistry and Laboratory Medicine 2008; 46: 1275–80.
Schoepfer AM, Trummler M, Seeholzer P, Seibold-Schmid B, Seibold F. Discriminating IBD from IBS: comparison of the test performance of fecal markers, blood leukocytes, CRP, and IBD antibodies. Inflammatory Bowel Disease 2008; 14: 32–9.
van Rheenen PF, Van de Vijver E, Fidler V. Faecal calprotectin for screening of patients with suspected inflammatory bowel disease: diagnostic meta-analysis. BMJ 2010; 341: c3369.
Gisbert JP, McNicholl AG, Gomollon F. Questions and answers on the role of fecal lactoferrin as a biological marker in inflammatory bowel disease. Inflammatory Bowel Disease 2009; 15: 1746–54.
Silberer H, Küppers B, Mickisch O, Baniewicz W, Drescher M, Traber L, et al. Fecal leukocyte proteins in inflammatory bowel disease and irritable bowel syndrome. Clinical Laboratory 2005; 51: 117–26.
Schröder O, Naumann M, Shastri Y, Povse N, Stein J. Prospective evaluation of faecal neutrophil-derived proteins in identifying intestinal inflammation: combination of parameters does not improve diagnostic accuracy of calprotectin. Alimentary Pharmacology & Therapeutics 2007; 26: 1035–42.
Kallen KJ. The role of transsignalling via the agonistic soluble IL-6 receptor in human diseases. Biochimica at Biophysica Acta 2002; 1592: 323–43.
Jones SA, Horiuchi S, Topley N, Yamamoto N, Fuller GM. The soluble interleukin 6 receptor: mechanisms of production and implications in disease. FASEB Journal 2001; 15: 43–58.
Heinrich PC, Behrmann I, Müller-Newen G, Schaper F, Graeve L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochemical Journal 1998; 334: 297–314.
Hirano T, Taga T, Nakano N, Yasukawa K, Kashiwamura S, Shimizu K, et al. Purification to homogeneity and characterization of human B-cell differentiation factor (BCDF or BSFp-2). Proceedings of the National Academy of Sciences of the United States of America 1985; 82: 5490–4.
Baumann H, Gauldie J. Regulation of hepatic acute phase plasma protein genes by hepatocyte stimulating factors and other mediators of inflammation. Molecular Biology and Medicine 1990; 7: 147–59.
Mudter J, Neurath MF. Il-6 signaling in inflammatory bowel disease: pathophysiological role and clinical relevance. Inflammatory Bowel Disease 2007; 13: 1016–23.
Umehara Y, Kudo M, Nakaoka R, Kawasaki T, Shiomi M. Serum proinflammatory cytokines and adhesion molecules in ulcerative colitis. Hepatogastroenterology 2006; 53: 879–82.
Wine E, Mack DR, Hyams J, Otley AR, Markowitz J, Crandall WV, et al. Interleukin-6 is associated with steroid resistance and reflects disease activity in severe pediatric ulcerative colitis. Journal of Crohn’s and Colitis 2013; 7: 916–22.
Bernardo D, Vallejo-Díez S, Mann ER, Al-Hassi HO, Martínez-Abad B, Montalvillo E, et al. IL-6 promotes immune responses in human ulcerative colitis and induces a skin-homing phenotype in the dendritic cells and Tcells they stimulate. European Journal of Immunology 2012; 42: 1337–53.
Atreya R, Mudter J, Finotto S, Müllberg J, Jostock T, Wirtz S, et al. Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: evidence in crohn disease and experimental colitis in vivo. Nature Medicine 2000; 6: 583–8.
Mudter J, Neurath MF. Apoptosis of T cells and the control of inflammatory bowel disease: therapeutic implications. Gut 2007; 56: 293–303.
Yen D, Cheung J, Scheerens H, Poulet F, McClanahan T, McKenzie B, et al. IL-23 is essential for T cell-mediated colitis and promotes inflammation via IL-17 and IL-6. Journal of Clinical Investigation 2006; 116: 1310–6.
Yamamoto M, Yoshizaki K, Kishimoto T, Ito H. IL-6 is required for the development of Th1 cell-mediated murine colitis. Journal of Immunology 2000; 164: 4878–82.
Fujimoto M, Nakano M, Terabe F, Kawahata H, Ohkawara T, Han Y, et al. The influence of excessive IL-6 production in vivo on the development and function of Foxp3+ regulatory T cells. Journal of Immunology 2011; 186: 32–40.
Kimura A, Kishimoto T. IL-6: regulator of Treg/Th17 balance. European Journal of Immunology 2010; 40: 1830–5.
Kirkham BW, Kavanaugh A, Reich K. Interleukin-17A: a unique pathway in immune-mediated diseases: psoriasis, psoriatic arthritis and rheumatoid arthritis. Immunology 2014; 141: 133–42.
Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annual Review of Immunology 2007; 25: 821–52.
Zhu S, Qian Y. IL-17/IL-17 receptor system in autoimmune disease: mechanisms and therapeutic potential. Clinical Science 2012; 122: 487–511.
Langley RG, Elewski BE, Lebwohl M, Reich K, Griffiths CE, Papp K, et al. Secukinumab in Plaque Psoriasis-Results of Two Phase 3 Trials. New England Journal of Medicine 2014 [Epub ahead of print].
McInnes IB, Sieper J, Braun J, Emery P, van der Heijde D, Isaacs JD, et al. Efficacy and safety of secukinumab, a fully human anti-interleukin-17A monoclonal antibody, in patients with moderate-to-severe psoriatic arthritis: a 24-week, randomised, double-blind, placebo-controlled, phase II proof-of-concept trial. Annals of the Rheumatic Diseases 2014; 73: 349–56.
Baeten D, Baraliakos X, Braun J, Sieper J, Emery P, van der Heijde D, et al. Anti-interleukin-17A monoclonal antibody secukinumab in treatment of ankylosing spondylitis: a randomised, double-blind, placebo-controlled trial. Lancet 2013; 382: 1705–13.
Genovese MC, Durez P, Richards HB, Supronik J, Dokoupilova E, Mazurov V, et al. Efficacy and safety of secukinumab in patients with rheumatoid arthritis: a phase II, dose-finding, double-blind, randomised, placebo controlled study. Annals of the Rheumatic Diseases 2013; 72: 863–9.
Nielsen OH, Kirman I, Rüdiger N, Hendel J, Vainer B. Upregulation of interleukin-12 and-17 in active inflammatory bowel disease. Scandinavian Journal of Gastroenterology 2003; 38: 180–5.
Elson CO, Cong Y, Weaver CT, Schoeb TR, McClanahan TK, Fick RB, et al. Monoclonal anti-interleukin 23 reverses active colitis in a T cell-mediated model in mice. Gastroenterology 2007; 132: 2359–70.
Khor B, Gardet A, Xavier RJ. Genetics and pathogenesis of inflammatory bowel disease. Nature 2011; 474: 307–17.
Li J, Tian H, Jiang HJ, Han B. Interleukin-17 SNPs and serum levels increase ulcerative colitis risk: a meta-analysis. World Journal of Gastroenetrology 2014; 20: 15899–909.
O’Connor W, Kamanaka M, Booth CJ, Town T, Nakae S, Iwakura Y, et al. A protective function for interleukin 17A in T cell-mediated intestinal inflammation. Nature Immunology 2009; 10: 603–9.
Ogawa A, Andoh A, Araki Y, Bamba T, Fujiyama Y. Neutralization of interleukin-17 aggravates dextran sulfate sodium-induced colitis in mice. Clinical Immunology 2004; 110: 55–62.
Strober W, Fuss IJ. Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology 2011; 140: 1756–67.
Murch SH, Braegger CP, Walker-Smith JA, MacDonald TT. Location of tumour necrosis factor a by immunohistochemistry in chronic inflammatory bowel disease. Gut 1993; 34: 1705–9.
Pallone F, Blanco Gdel V, Vavassori P, Monteleone I, Fina D, Monteleone G. Genetic and pathogenetic insights into inflammatory bowel disease. Current Gastroenterology Reprots 2003; 5: 487–92.
van Heel DA, Udalova IA, De Silva AP, McGovern DP, Kinouchi Y, Hull J, et al. Inflammatory bowel disease is associated with a TNF polymorphism that affects an interaction between the OCT1 and NF(-kappa)B transcription factors. Human Molecular Genetics 2002; 11: 1281–9.
Rossetti S, Actis GC, Fadda M, Rizzetto M, A. P. M. The use of the anti-tumour necrosis factor monoclonal antibody–infliximab–to treat ulcerative colitis: implications and trends beyond the available data. Digestive and Liver Disease 2004; 36: 426–31.
Ye D, Ma I, Ma TY. Molecular mechanism of tumor necrosis factor-alpha modulation of intestinal epithelial tight junction barrier. American Jornal of Physiology Gastrointestinal and Liver Physiology 2006; 290: G496–504.
Muro M, López-Hernández R, Mrowiec A. Immunogenetic biomarkers in inflammatory bowel diseases: role of the IBD3 region. World Journal of Gastroenetrology 2014; 20: 15037–24048.
Olsen T, Goll R, Cui G, Husebekk A, Vonen B, Birketvedt GS, et al. Tissue levels of tumor necrosis factor-alpha correlates with grade of inflammation in untreated ulcerative colitis. Scandinavian Journal of Gastroenterology 2007; 42: 1312–20.
Spoettl T, Hausmann M, Klebl F, Dirmeier A, Klump B, Hoffmann J, et al. Serum soluble TNF receptor I and II levels correlate with disease activity in IBD patients. Inflammatory Bowel Disease 2007; 13: 727–32.
Lawson MM, Thomas AG, Akobeng AK. Tumour necrosis factor alpha blocking agents for induction of remission in ulcerative colitis. The Cochrane Database of Systematic Reviews 2006; 19: CD005112.
Sipponen SE, Kärkkäinen P, Kolho KL, Nuutinen H, Turunen U, Färkkilä M. Fecal calprotectin, lactoferrin, and endoscopic disease activity in monitoring anti-TNF-alpha therapy for Crohn’s disease. Inflammatory Bowel Disease 2008; 14: 1392–8.
Hölttä V, Sipponen T, Westerholm-Ormio M, Salo HM, Kolho KL, Färkkilä M, et al. In Crohn’s disease, anti-TNF-α treatment changes the balance between mucosal IL-17, FOXP3, and CD4 Cells. ISNR Gastroenterology 2012; 2012: 505432.
Gisbert JP, Bermejo F, Pérez-Calle JL, Taxonera C, Vera I, McNicholl AG, et al. Fecal calprotectin and lactoferrin for the prediction of inflammatory bowel disease relapse. Inflammatory Bowel Disease 2009; 15: 1190–8.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Mańkowska-Wierzbicka, D., Swora-Cwynar, E., Poniedziałek, B. et al. Usefulness of selected laboratory markers in ulcerative colitis. Eur Cytokine Netw 26, 26–37 (2015). https://doi.org/10.1684/ecn.2015.0363
Published:
Issue Date:
DOI: https://doi.org/10.1684/ecn.2015.0363