Abstract
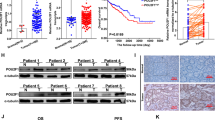
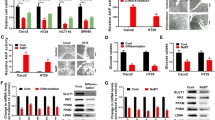
Metabolic reprogramming is a common phenomenon in cancer, with aerobic glycolysis being one of its important characteristics. Hypoxia-inducible factor-1α (HIF1A) is thought to play an important role in aerobic glycolysis. Meanwhile, naringin is a natural flavanone glycoside derived from grapefruits and many other citrus fruits. In this work, we identified glycolytic genes related to HIF1A by analyzing the colon cancer database. The analysis of extracellular acidification rate and cell function verified the regulatory effects of HIF1A overexpression on glycolysis, and the proliferation and migration of colon cancer cells. Moreover, naringin was used as an inhibitor of colon cancer cells to illustrate its effect on HIF1A function. The results showed that the HIF1A and enolase 2 (ENO2) levels in colon cancer tissues were highly correlated, and their high expression indicated a poor prognosis for colon cancer patients. Mechanistically, HIF1A directly binds to the DNA promoter region and upregulates the transcription of ENO2; ectopic expression of ENO2 increased aerobic glycolysis in colon cancer cells. Most importantly, we found that the appropriate concentration of naringin inhibited the transcriptional activity of HIF1A, which in turn decreased aerobic glycolysis in colon cancer cells. Generally, naringin reduces glycolysis in colon cancer cells by reducing the transcriptional activity of HIF1A and the proliferation and invasion of colon cancer cells. This study helps to elucidate the relationship between colon cancer progression and glucose metabolism, and demonstrates the efficacy of naringin in the treatment of colon cancer.
概要
代谢重编程是癌症中一种常见的现象,而有氧糖酵解是其重要特征之一。缺氧诱导因子1亚基(HIF1Α)被认为在有氧糖酵解中发挥重要作用。柚皮苷是一种从葡萄柚和柑橘类水果中提取的天然的黄酮糖苷。在本研究中,我们通过分析结肠癌数据库,确定了与HIF1Α相关的糖酵解基因。通过进行细胞外酸化率和细胞功能的实验,证实了过表达HIF1Α对糖酵解以及结肠癌细胞增殖和迁移的调控作用。此外,我们用柚皮苷作为结肠癌细胞的抑制剂来阐明其对HIF1Α功能的影响。结果显示,结肠癌组织中HIF1Α和烯醇酶-2(ENO2)水平高度相关,其高表达提示结肠癌患者预后较差。HIF1Α直接与DNA启动子区域结合,上调ENO2的转录;而ENO2的异常表达增加了结肠癌细胞的有氧糖酵解水平。最重要的是,一定浓度的柚皮苷抑制了HIF1Α的转录活性,从而降低了结肠癌细胞的有氧糖酵解水平。总之,柚皮苷通过降低HIF1Α的转录活性来减少结肠癌细胞的糖酵解以及结肠癌细胞的增殖和侵袭。本研究有助于阐明结肠癌的进展与糖代谢之间的关系,并证明柚皮苷对结肠癌的治疗效果。
Similar content being viewed by others
References
Ademosun AO, Oboh G, Passamonti S, et al., 2015. Inhibition of metalloproteinase and proteasome activities in colon cancer cells by citrus peel extracts. J Basic Clin Physiol Pharmacol, 26(5):471–477. https://doi.org/10.1515/jbcpp-2013-0127
Ahmad SS, Glatzle J, Bajaeifer K, et al., 2013. Phosphoglycerate kinase 1 as a promoter of metastasis in colon cancer. Int J Oncol, 43(2):586–590. https://doi.org/10.3892/ijo.2013.1971
André T, de Gramont A, Vernerey D, et al., 2015. Adjuvant fluorouracil, leucovorin, and oxaliplatin in stage II to III colon cancer: updated 10-year survival and outcomes according to BRAF mutation and mismatch repair status of the MOSAIC study. J Clin Oncol, 33(35):4176–4187. https://doi.org/10.1200/JCO.2015.63.4238
Andreou D, Steen NE, Jørgensen KN, et al., 2021. Lower circulating neuron-specific enolase concentrations in adults and adolescents with severe mental illness. Psychol Med, published online. https://doi.org/10.1017/S0033291721003056
Ansari B, Aschner M, Hussain Y, et al., 2022. Suppression of colorectal carcinogenesis by naringin. Phytomedicine, 96:153897. https://doi.org/10.1016/j.phymed.2021.153897
Cartwright TH, 2012. Treatment decisions after diagnosis of metastatic colorectal cancer. Clin Colorectal Cancer, 11(3): 155–166. https://doi.org/10.1016/j.clcc.2011.11.001
Chen R, Qi QL, Wang MT, et al., 2016. Therapeutic potential of naringin: an overview. Pharm Biol, 54(12):3203–3210. https://doi.org/10.1080/13880209.2016.1216131
Denlinger CS, Barsevick AM, 2009. The challenges of colorectal cancer survivorship. J Natl Compr Canc Netw, 7(8): 883–894. https://doi.org/10.6004/jnccn.2009.0058
Dienstmann R, Salazar R, Tabernero J, 2015. Personalizing colon cancer adjuvant therapy: selecting optimal treatments for individual patients. J Clin Oncol, 33(16):1787–1796. https://doi.org/10.1200/JCO.2014.60.0213
Du X, Zhang J, Liu L, et al., 2022. A novel anticancer property of Lycium barbarum polysaccharide in triggering ferroptosis of breast cancer cells. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 23(4):286–299. https://doi.org/10.1631/jzus.B2100748
Gopinath K, Sudhandiran G, 2012. Naringin modulates oxidative stress and inflammation in 3-nitropropionic acid-induced neurodegeneration through the activation of nuclear factor-erythroid 2-related factor-2 signalling pathway. Neuroscience, 227:134–143. https://doi.org/10.1016/j.neuroscience.2012.07.060
Habauzit V, Sacco SM, Gil-Izquierdo A, et al., 2011. Differential effects of two citrus flavanones on bone quality in senescent male rats in relation to their bioavailability and metabolism. Bone, 49(5): 1108–1116. https://doi.org/10.1016/j.bone.2011.07.030
Hsu PP, Sabatini DM, 2008. Cancer cell metabolism: Warburg and beyond. Cell, 134(5):703–707. https://doi.org/10.1016/j.cell.2008.08.021
Hu HL, Zhu WW, Qin J, et al., 2017. Acetylation of PGK1 promotes liver cancer cell proliferation and tumorigenesis. Hepatology, 65(2):515–528. https://doi.org/10.1002/hep.28887
Jiang XY, Chen Z, Zhu JP, et al., 2022. E2F1 promotes Warburg effect and cancer progression via upregulating ENO2 expression in Ewing sarcoma. Mol Med Rep, 26(1):237. https://doi.org/10.3892/mmr.2022.12753
Kim DI, Lee SJ, Lee SB, et al., 2008. Requirement for Ras/Raf/ ERK pathway in naringin-induced G1-cell-cycle arrest via p21WAF1 expression. Carcinogenesis, 29(9):1701–1709. https://doi.org/10.1093/carcin/bgn055
Kim J, Jin H, Zhao JC, et al., 2017. FOXA1 inhibits prostate cancer neuroendocrine differentiation. Oncogene, 36(28): 4072–4080. https://doi.org/10.1038/onc.2017.50
Kim JW, Dang CV, 2005. Multifaceted roles of glycolytic enzymes. Trends Biochem Sci, 30(3):142–150. https://doi.org/10.1016/j.tibs.2005.01.005
Koppenol WH, Bounds PL, Dang CV, 2011. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat Rev Cancer, 11(5):325–337. https://doi.org/10.1038/nrc3038
Li HZ, Yang B, Huang J, et al., 2013. Naringin inhibits growth potential of human triple-negative breast cancer cells by targeting β-catenin signaling pathway. Toxicol Lett, 220(3):219–228. https://doi.org/10.1016/j.toxlet.2013.05.006
Liu DS, Mao YM, Chen C, et al., 2020. Expression patterns and clinical significances of ENO2 in lung cancer: an analysis based on Oncomine database. Ann Transl Med, 8(10):639. https://doi.org/10.21037/atm-20-3354
Luo YL, Zhang CC, Li PB, et al., 2012. Naringin attenuates enhanced cough, airway hyperresponsiveness and airway inflammation in a guinea pig model of chronic bronchitis induced by cigarette smoke. Int Immunopharmacol, 13(3): 301–307. https://doi.org/10.1016/j.intimp.2012.04.019
Miller KD, Nogueira L, Mariotto AB, et al., 2019. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin, 69(5):363–385. https://doi.org/10.3322/caac.21565
Rajedadram A, Pin KY, Ling SK, et al., 2021. Hydroxychavicol, a polyphenol from Piper betle leaf extract, induces cell cycle arrest and apoptosis in TP53-resistant HT-29 colon cancer cells. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 22(2):112–122. https://doi.org/10.1631/jzus.B2000446
Ramzy A, ElSafy S, Elshoky HA, et al., 2023. Drugless nanoparticles tune-up an array of intertwined pathways contributing to immune checkpoint signaling and metabolic reprogramming in triple-negative breast cancer. Biomed Mater, 18:015023. https://doi.org/10.1088/1748-605X/aca85d
Schwartz L, Seyfried T, Alfarouk KO, et al., 2017. Out of Warburg effect: an effective cancer treatment targeting the tumor specific metabolism and dysregulated pH. Semin Cancer Biol, 43:134–138. https://doi.org/10.1016/j.semcancer.2017.01.005
Soh MA, Garrett SH, Somji S, et al., 2011. Arsenic, cadmium and neuron specific enolase (ENO2, γ-enolase) expression in breast cancer. Cancer Cell Int, 11:41. https://doi.org/10.1186/1475-2867-11-41
Sun BY, Liu YQ, He DH, et al., 2021. Traditional Chinese medicines and their active ingredients sensitize cancer cells to TRAIL-induced apoptosis. J Zhejiang Univ-Sci B (Biomed& Biotechnol), 22(3):190–203. https://doi.org/10.1631/jzus.B2000497
Taieb J, le Malicot K, Shi Q, et al., 2017. Prognostic value of BRAF and KRAS mutations in MSI and MSS stage III colon cancer. J Natl Cancer Inst, 109(5):djw272. https://doi.org/10.1093/jnci/djw272
Vanamala J, Leonardi T, Patil BS, et al., 2006. Suppression of colon carcinogenesis by bioactive compounds in grapefruit. Carcinogenesis, 27(6):1257–1265. https://doi.org/10.1093/carcin/bgi318
vander Heiden MG, Cantley LC, Thompson CB, 2009. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science, 324(5930):1029–1033. https://doi.org/10.1126/science.1160809
Wang DM, Yan JQ, Chen J, et al., 2015. Naringin improves neuronal insulin signaling, brain mitochondrial function, and cognitive function in high-fat diet-induced obese mice. Cell Mol Neurobiol, 35(7):1061–1071. https://doi.org/10.1007/s10571-015-0201-y
Warburg O, Wind F, Negelein E, 1927. The metabolism of tumors in the body. J Gen Physiol, 8(6):519–530. https://doi.org/10.1085/jgp.8.6.519
Yu LL, Lu MY, Jia DY, et al., 2017. Modeling the genetic regulation of cancer metabolism: interplay between glycolysis and oxidative phosphorylation. Cancer Res, 77(7): 1564–1574. https://doi.org/10.1158/0008-5472.CAN-16-2074
Yukimoto R, Nishida N, Hata T, et al., 2021. Specific activation of glycolytic enzyme enolase 2 in BRAF V600E-mutated colorectal cancer. Cancer Sci, 112(7):2884–2894. https://doi.org/10.1111/cas.14929
Zhang JZ, Gao WY, Liu Z, et al., 2014. Systematic analysis of main constituents in rat biological samples after oral administration of the methanol extract of fructus aurantii by HPLC-ESI-MS/MS. Iran J Pharm Res, 13(2): 493–503.
Zheng Y, Wu C, Yang JM, et al., 2020. Insulin-like growth factor 1-induced enolase 2 deacetylation by HDAC3 promotes metastasis of pancreatic cancer. Sig Transduct Target Ther, 5:53. https://doi.org/10.1038/s41392-020-0146-6
Acknowledgments
This work was supported by the Fund of Hubei Provincial Health Commission (No. ZY2021M080), the Medical Research Project of Jiangsu Provincial Health Commission (No. Z2021068), the Yancheng Medical Science and Technology Development Plan Project (No. YK2021004), and the Young Scientific and Technological Talents Support Project by Jiangsu Association for Science and Technology (No. TJ-2022-097), China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author contributions
Guangtao PAN and Ping ZHANG performed the experiments and prepared the manuscript. Aiying CHEN, Yu DENG, Zhen ZHANG, Han LU, Aoxun ZHU, and Cong ZHOU performed the experiments and statistical analyses. Sen LI and Yanran WU designed the draft of the research process and provided funding for the experiment. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Guangtao PAN, Ping ZHANG, Aiying CHEN, Yu DENG, Zhen ZHANG, Han LU, Aoxun ZHU, Cong ZHOU, Yanran WU, and Sen LI declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Supplementary information
Figs. S1-S4; Materials and methods
Supplementary information
Rights and permissions
About this article
Cite this article
Pan, G., Zhang, P., Chen, A. et al. Aerobic glycolysis in colon cancer is repressed by naringin via the HIF1A pathway. J. Zhejiang Univ. Sci. B 24, 221–231 (2023). https://doi.org/10.1631/jzus.B2200221
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200221
Key words
- Colon cancer
- Naringin
- Hypoxia inducible factor-1α (HIF1A)
- Enolase 2 (ENO2)
- Glycolysis
- Metabolic reprogramming