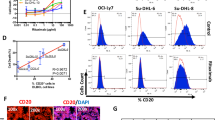
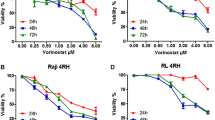
Abstract
Diffuse large B-cell lymphoma (DLBCL) is an aggressive type of non-Hodgkin’s lymphoma. A total of 10%–15% of DLBCL cases are associated with myelocytomatosis viral oncogene homolog (MYC) and/or B-cell lymphoma-2 (BCL2) translocation or amplification. BCL2 inhibitors have potent anti-tumor effects in DLBCL; however, resistance can be acquired through up-regulation of alternative anti-apoptotic proteins. The histone deacetylase (HDAC) inhibitor chidamide can induce BIM expression leading to apoptosis of lymphoma cells with good efficacy in refractory recurrent DLBCL. In this study the synergistic mechanism of chidamide and venetoclax in DLBCL was determined through in vitro and in vivo models. We found that combination therapy significantly reduced the protein levels of MYC TP53 and BCL2 in activated apoptotic-related pathways in DLBCL cells by increasing BIM levels and inducing cell apoptosis. Moreover combination therapy regulated expression of multiple transcriptomes in DLBCL cells involving apoptosis cell cycle phosphorylation and other biological processes and significantly inhibited tumor growth in DLBCL-bearing xenograft mice. Taken together these findings verify the in vivo therapeutic potential of chidamide and venetoclax combination therapy in DLBCL warranting pre-clinical trials for patients with DLBCL.
摘要
目的
探讨表观遗传学组蛋白去乙酰化酶(HDAC)抑制剂西达本胺与BCL2抑制剂维奈托克的联合对弥漫性大B细胞淋巴瘤(DLBCL)生长的影响及相关机制。
创新点
本研究首次探索了将西达本胺和维奈托克联合作用于MYC+/BCL2+DLBCL, 使用二代测序(NGS)开创性地从表观遗传学和基因蛋白层面探讨这种联合用药的效果及作用机制。
方法
利用生物信息学技术分析表观遗传学HDAC基因与BCL2基因之间的相关性; 体外应用DHL细胞株DB(MYC/BCL2重排)和DEL细胞株SUDHL-4(MYC、BCL2表达)分别进行单药和联合用药处理, 通过CCK-8法检测细胞活力, 流式细胞术检测细胞凋亡和周期, RNA测序和蛋白质印迹(western blot)检测MYC、BCL2、TP53等相关基因的mRNA水平及蛋白的表达; 体内建立DLBCL异种移植小鼠模型进行单药和联合用药治疗, 分析并评价药物治疗后皮下瘤的大小和病理切片。
结论
西达本胺与维奈托克协同抑制DLBCL的生长, 通过调控表观遗传的改变, 沉默MYC、TP53的表达, 降低抗凋亡蛋白BCL2表达以及增加促凋亡蛋白BIM表达, 诱导肿瘤细胞的凋亡和细胞周期阻滞。
Similar content being viewed by others
References
Adams CM, Hiebert SW, Eischen CM, 2016. Myc induces miRNA-mediated apoptosis in response to HDAC inhibition in hematologic malignancies. Cancer Res, 76(3): 736–748. https://doi.org/10.1158/0008-5472.CAN-15-1751
Baluapuri A, Wolf E, Eilers M, 2020. Target gene-independent functions of MYC oncoproteins. Nat Rev Mol Cell Biol, 21(5):255–267. https://doi.org/10.1038/s41580-020-0215-2
Berendsen MR, Stevens WBC, van den Brand M, et al., 2020. Molecular genetics of relapsed diffuse large B-cell lymphoma: insight into mechanisms of therapy resistance. Cancers, 12(12):3553. https://doi.org/10.3390/cancers12123553
Bhola PD, Letai A, 2016. Mitochondria—judges and executioners of cell death sentences. Mol Cell, 61(5):695–704. https://doi.org/10.1016/j.molcel.2016.02.019
Bobrowicz M, Dwojak M, Pyrzynska B, et al., 2017. HDAC6 inhibition upregulates CD20 levels and increases the efficacy of anti-CD20 monoclonal antibodies. Blood, 130(14): 1628–1638. https://doi.org/10.1182/blood-2016-08-736066
Burotto M, Berkovits A, Dunleavy K, 2016. Double hit lymphoma: from biology to therapeutic implications. Exp Rev Hematol, 9(7):669–678. https://doi.org/10.1080/17474086.2016.1182858
Chan TS, Tse E, Kwong YL, 2017. Chidamide in the treatment of peripheral T-cell lymphoma. OncoTargets Ther, 10:347–352. https://doi.org/10.2147/ott.S93528
Chen K, Yang QY, Zha J, et al., 2020. Preclinical evaluation of a regimen combining chidamide and ABT-199 in acute myeloid leukemia. Cell Death Dis, 11(9):778. https://doi.org/10.1038/s41419-020-02972-2
Chou TC, 2010. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res, 70(2):440–446. https://doi.org/10.1158/0008-5472.CAN-09-1947
Croce CM, Reed JC, 2016. Finally, an apoptosis-targeting therapeutic for cancer. Cancer Res, 76(20):5914–5920. https://doi.org/10.1158/0008-5472.CAN-16-1248
Duffy MJ, O’Grady S, Tang MH, et al., 2021. MYC as a target for cancer treatment. Cancer Treat Rev, 94:102154. https://doi.org/10.1016/j.ctrv.2021.102154
Ecker J, Thatikonda V, Sigismondo G, et al., 2021. Reduced chromatin binding of MYC is a key effect of HDAC inhibition in MYC amplified medulloblastoma. Neuro Oncol, 23(2):226–239. https://doi.org/10.1093/neuonc/noaa191
Friedberg JW, 2017. How I treat double-hit lymphoma. Blood, 130(5):590–596. https://doi.org/10.1182/blood-2017-04-737320
Gong P, Wang YT, Jing YK, 2019. Apoptosis induction by histone deacetylase inhibitors in cancer cells: role of Ku70. Int J Mol Sci, 20(7):1601. https://doi.org/10.3390/ijms20071601
Guan XW, Wang HQ, Ban WW, et al., 2020. Novel HDAC inhibitor Chidamide synergizes with Rituximab to inhibit diffuse large B-cell lymphoma tumour growth by upregulating CD20. Cell Death Dis, 11(1):20. https://doi.org/10.1038/s41419-019-2210-0
Hafezi S, Rahmani M, 2021. Targeting BCL-2 in cancer: advances, challenges, and perspectives. Cancers, 13(6): 1292. https://doi.org/10.3390/cancers13061292
Hata AN, Engelman JA, Faber AC, 2015. The BCL2 family: key mediators of the apoptotic response to targeted anticancer therapeutics. Cancer Discov, 5(5):475–487. https://doi.org/10.1158/2159-8290.CD-15-0011
Heideman MR, Wilting RH, Yanover E, et al., 2013. Dosage-dependent tumor suppression by histone deacetylases 1 and 2 through regulation of c-Myc collaborating genes and p53 function. Blood, 121(11):2038–2050. https://doi.org/10.1182/blood-2012-08-450916
Huang H, Wu HW, Hu YX, 2020. Current advances in chimeric antigen receptor T-cell therapy for refractory/relapsed multiple myeloma. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(1):29–41. https://doi.org/10.1631/jzus.B1900351
Kapoor I, Bodo J, Hill BT, et al., 2020. Targeting BCL-2 in B-cell malignancies and overcoming therapeutic resistance. Cell Death Dis, 11(11):941. https://doi.org/10.1038/s41419-020-03144-y
Laszig S, Boedicker C, Weiser T, et al., 2020. The novel dual BET/HDAC inhibitor TW09 mediates cell death by mitochondrial apoptosis in rhabdomyosarcoma cells. Cancer Lett, 486:46–57. https://doi.org/10.1016/j.canlet.2020.05.008
Li SY, Young KH, Medeiros LJ, 2018. Diffuse large B-cell lymphoma. Pathology, 50(1):74–87. https://doi.org/10.1016/j.pathol.2017.09.006
Li WP, Gupta SK, Han WG, et al., 2019. Targeting MYC activity in double-hit lymphoma with MYC and BCL2 and/or BCL6 rearrangements with epigenetic bromodomain inhibitors. J Hematol Oncol, 12:73. https://doi.org/10.1186/s13045-019-0761-2
Li X, Yan X, Guo WJ, et al., 2017. Chidamide in FLT3-ITD positive acute myeloid leukemia and the synergistic effect in combination with cytarabine. Biomed Pharmacother, 90:699–704. https://doi.org/10.1016/j.biopha.2017.04.037
Matthews GM, Newbold A, Johnstone RW, 2012. Intrinsic and extrinsic apoptotic pathway signaling as determinants of histone deacetylase inhibitor antitumor activity. Adv Cancer Res, 116:165–197. https://doi.org/10.1016/B978-0-12-394387-3.00005-7
Morin RD, Assouline S, Alcaide M, et al., 2016. Genetic landscapes of relapsed and refractory diffuse large B-cell lymphomas. Clin Cancer Res, 22(9):2290–2300. https://doi.org/10.1158/1078-0432.CCR-15-2123
Mrakovcic M, Kleinheinz J, Fröhlich LF, 2019. p53 at the crossroads between different types of HDAC inhibitormediated cancer cell death. Int J Mol Sci, 20(10):2415. https://doi.org/10.3390/ijms20102415
Muthalagu N, Junttila MR, Wiese KE, et al., 2014. BIM is the primary mediator of MYC-induced apoptosis in multiple solid tissues. Cell Rep, 8(5):1347–1353. https://doi.org/10.1016/j.celrep.2014.07.057
Nebbioso A, Carafa V, Conte M, et al., 2017. c-Myc modulation and acetylation is a key HDAC inhibitor target in cancer. Clin Cancer Res, 23(10):2542–2555. https://doi.org/10.1158/1078-0432.CCR-15-2388
Ning ZQ, Li ZB, Newman MJ, et al., 2012. Chidamide (CS055/HBI-8000): a new histone deacetylase inhibitor of the benzamide class with antitumor activity and the ability to enhance immune cell-mediated tumor cell cytotoxicity. Cancer Chemother Pharmacol, 69(4):901–909. https://doi.org/10.1007/s00280-011-1766-x
Niu XJ, Zhao JY, Ma J, et al., 2016. Binding of released Bim to Mcl-1 is a mechanism of intrinsic resistance to ABT-199 which can be overcome by combination with daunorubicin or cytarabine in AML cells. Clin Cancer Res, 22(17):4440–4451. https://doi.org/10.1158/1078-0432.CCR-15-3057
Nowakowski GS, Blum KA, Kahl BS, et al., 2016. Beyond RCHOP: a blueprint for diffuse large B cell lymphoma research. JNCI, 108(12):djw257. https://doi.org/10.1093/jnci/djw257
Pan H, Jiang YW, Boi M, et al., 2015. Epigenomic evolution in diffuse large B-cell lymphomas. Nat Commun, 6:6921. https://doi.org/10.1038/ncomms7921
Patriarca A, Gaidano G, 2021. Investigational drugs for the treatment of diffuse large B-cell lymphoma. Exp Opin Invest Drugs, 30(1):25–38. https://doi.org/10.1080/13543784.2021.1855140
Perini GF, Ribeiro GN, Neto JVP, et al., 2018. BCL-2 as therapeutic target for hematological malignancies. J Hematol Oncol, 11:65. https://doi.org/10.1186/s13045-018-0608-2
Riedell PA, Smith SM, 2018. Double hit and double expressors in lymphoma: definition and treatment. Cancer, 124(24): 4622–4632. https://doi.org/10.1002/cncr.31646
Rodríguez-Paredes M, Esteller M, 2011. Cancer epigenetics reaches mainstream oncology. Nat Med, 17(3):330–339. https://doi.org/10.1038/nm.2305
Rosenthal A, Younes A, 2017. High grade B-cell lymphoma with rearrangements of MYC and BCL2 and/or BCL6: double hit and triple hit lymphomas and double expressing lymphoma. Blood Rev, 31(2):37–42. https://doi.org/10.1016/j.blre.2016.09.004
Santoro F, Botrugno OA, Dal Zuffo R, et al., 2013. A dual role for Hdac1: oncosuppressor in tumorigenesis, oncogene in tumor maintenance. Blood, 121(17):3459–3468. https://doi.org/10.1182/blood-2012-10-461988
Sarkozy C, Traverse-Glehen A, Coiffier B, 2015. Double-hit and double-protein-expression lymphomas: aggressive and refractory lymphomas. Lancet Oncol, 16(15):E555–E567. https://doi.org/10.1016/s1470-2045(15)00005-4
Sermer D, Pasqualucci L, Wendel HG, et al., 2019. Emerging epigenetic-modulating therapies in lymphoma. Nat Rev Clin Oncol, 16(8):494–507. https://doi.org/10.1038/s41571-019-0190-8
Shi YK, Jia B, Xu W, et al., 2017. Chidamide in relapsed or refractory peripheral T cell lymphoma: a multicenter real-world study in China. J Hematol Oncol, 10:69. https://doi.org/10.1186/s13045-017-0439-6
Shimizu R, Kikuchi J, Wada T, et al., 2010. HDAC inhibitors augment cytotoxic activity of rituximab by upregulating CD20 expression on lymphoma cells. Leukemia, 24(10): 1760–1768. https://doi.org/10.1038/leu.2010.157
Souers AJ, Leverson JD, Boghaert ER, et al., 2013. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med, 19(2): 202–208. https://doi.org/10.1038/nm.3048
Stazi G, Fioravanti R, Mai A, et al., 2019. Histone deacetylases as an epigenetic pillar for the development of hybrid inhibitors in cancer. Curr Opin Chem Biol, 50:89–100. https://doi.org/10.1016/j.cbpa.2019.03.002
Swerdlow SH, Campo E, Pileri SA, et al., 2016. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood, 127(20):2375–2390. https://doi.org/10.1182/blood-2016-01-643569
Vandenberg CJ, Cory S, 2013. ABT-199, a new Bcl-2-specific BH3 mimetic, has in vivo efficacy against aggressive Mycdriven mouse lymphomas without provoking thrombocytopenia. Blood, 121(12):2285–2288. https://doi.org/10.1182/blood-2013-01-475855
Wang P, Wang Z, Liu J, 2020. Role of HDACs in normal and malignant hematopoiesis. Mol Cancer, 19:5. https://doi.org/10.1186/s12943-019-1127-7
Wang XG, Waschke BC, Woolaver RA, et al., 2020. HDAC inhibitors overcome immunotherapy resistance in B-cell lymphoma. Protein Cell, 11(7):472–482. https://doi.org/10.1007/s13238-020-00694-x
Xu Y, Zhang P, Liu Y, 2017. Chidamide tablets: HDAC inhibition to treat lymphoma. Drugs Today, 53(3): 167–176. https://doi.org/10.1358/dot.2017.53.3.2595452
Yuan XG, Huang YR, Yu T, et al., 2019. Chidamide, a histone deacetylase inhibitor, induces growth arrest and apoptosis in multiple myeloma cells in a caspase-dependent manner. Oncol Lett, 18(1):411–419. https://doi.org/10.3892/ol.2019.10301
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 81460030 and 81770221).
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Cancan LUO performed research design, the experimental research, and data analysis, wrote and edited the manuscript. Tiantian YU performed the experimental research and data analysis. Ken H. YOUNG performed the research design and data analysist. Li YU performed research design and data analysis, wrote and edited the manuscript. All authors have read and approved the final manuscript, and therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Cancan LUO, Tiantian YU, Ken H. YOUNG, and Li YU declare that they have no conflict of interest.
This study was carried out in accordance with the recommendations of the Ethics Committee of the Second Affiliated Hospital of Nanchang University. The protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Nanchang University. All subjects gave written informed consent in accordance with the Declaration of Helsinki.
Rights and permissions
About this article
Cite this article
Luo, C., Yu, T., Young, K.H. et al. HDAC inhibitor chidamide synergizes with venetoclax to inhibit the growth of diffuse large B-cell lymphoma via down-regulation of MYC, BCL2, and TP53 expression. J. Zhejiang Univ. Sci. B 23, 666–681 (2022). https://doi.org/10.1631/jzus.B2200016
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200016