Abstract
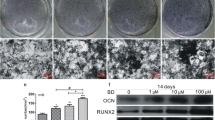
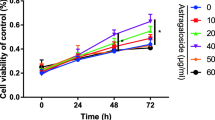
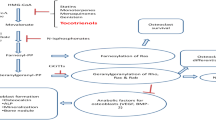
Hypertension is a prevalent systemic disease in the elderly, who can suffer from several pathological skeletal conditions simultaneously, including osteoporosis. Benidipine (BD), which is widely used to treat hypertension, has been proved to have a beneficial effect on bone metabolism. In order to confirm the osteogenic effects of BD, we investigated its osteogenic function using mouse MC3T3-E1 preosteoblast cells in vitro. The proliferative ability of MC3T3-E1 cells was significantly associated with the concentration of BD, as measured by methylthiazolyldiphenyl-tetrazolium bromide (MTT) assay and cell cycle assay. With BD treatment, the osteogenic differentiation and maturation of MC3T3-E1 cells were increased, as established by the alkaline phosphatase (ALP) activity test, matrix mineralized nodules formation, osteogenic genetic test, and protein expression analyses. Moreover, our data showed that the BMP2/Smad pathway could be the partial mechanism for the promotion of osteogenesis by BD, while BD might suppress the possible function of osteoclasts through the OPG/RANKL/RANK (receptor activator of nuclear factor- κB (NF-κB)) pathway. The hypothesis that BD bears a considerable potential in further research on its dual therapeutic effect on hypertensive patients with poor skeletal conditions was proved within the limitations of the present study.
概要
高血压在老年人群中多发, 而不良骨质情况例如骨质疏松症也多见于老年患者。贝尼地平(Benidipine, BD)作为广泛使用的抗高血压药, 被发现具有一定的改善骨代谢的作用。为探究贝尼地平的促成骨作用, 本实验选用了小鼠MC3T3-E1前成骨细胞进行体外实验。MTT及细胞周期试验显示M3CT3-E1细胞增殖能力受BD影响显著提升且具有浓度依赖性。碱性磷酸酶活力试验、基质矿化试验、成骨相关基因和蛋白表达的测定发现, BD可促进MC3T3-E1细胞的成骨向分化和成熟。此外, 本项研究结果显示, BMP2/Smad通路在BD的促成骨效应中发挥作用, 而OPG/RANKL/RANK通路的改变提示BD可能通过抑制破骨细胞的活动间接改善骨代谢。综上, 在进一步研发治疗伴有不良骨质情况的高血压患者的双效药物方面, BD具有广阔的研究前景和应用潜力。
Similar content being viewed by others
References
Afzal F, Pratap J, Ito K, et al., 2005. Smad function and intranuclear targeting share a Runx2 motif required for osteogenic lineage induction and BMP2 responsive transcription. J Cell Physiol, 204(1):63–72. https://doi.org/10.1002/jcp.20258
Boyle WJ, Simonet WS, Lacey DL, 2003. Osteoclast differentiation and activation. Nature, 423(6937):337–342. https://doi.org/10.1038/nature01658
Cao X, Chen D, 2005. The BMP signaling and in vivo bone formation. Gene, 357(1): 1–8. https://doi.org/10.1016/j.gene.2005.06.017
Chen MM, Zhang YH, Du YP, et al., 2019. Epidemiological and clinical study of hip fracture in hospitalized elderly patients in Shanghai, China. Arch Osteoporos, 14:37. https://doi.org/10.1007/s11657-019-0580-7
Chen P, Li ZZ, Hu YH, 2016. Prevalence of osteoporosis in China: a meta-analysis and systematic review. BMC Public Health, 16:1039. https://doi.org/10.1186/s12889-016-3712-7
Chen X, Wang ZQ, Duan N, et al., 2018. Osteoblast-osteoclast interactions. Connect Tissue Res, 59(2):99–107. https://doi.org/10.1080/03008207.2017.1290085
Cui ZY, Meng XY, Feng H, et al., 2019. Estimation and projection about the standardized prevalence of osteoporosis in mainland China. Arch Osteoporos, 15:2. https://doi.org/10.1007/s11657-019-0670-6
Ducy P, Starbuck M, Priemel M, et al., 1999. A Cbfa1-dependent genetic pathway controls bone formation beyond embryonic development. Genes Dev, 13(8):1025–1036. https://doi.org/10.1101/gad.13.8.1025
Gao SW, Liu F, 2019. Novel insights into cell cycle regulation of cell fate determination. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 20(6):467–475. https://doi.org/10.1631/jzus.B1900197
Ghosh M, Majumdar SR, 2014. Antihypertensive medications, bone mineral density, and fractures: a review of old cardiac drugs that provides new insights into osteoporosis. Endocrine, 46(3):397–405. https://doi.org/10.1007/s12020-014-0167-4
Higuchi A, Ling QD, Hsu ST, et al., 2012. Biomimetic cell culture proteins as extracellular matrices for stem cell differentiation. Chem Rev, 112(8):4507–4540. https://doi.org/10.1021/cr3000169
Huang ZN, Ren PG, Ma T, et al., 2010. Modulating osteogenesis of mesenchymal stem cells by modifying growth factor availability. Cytokine, 51(3):305–310. https://doi.org/10.1016/j.cyto.2010.06.002
Ilić K, Obradović N, Vujasinović-Stupar N, 2013. The relationship among hypertension, antihypertensive medications, and osteoporosis: a narrative review. Calcif Tissue Int, 92(3):217–227. https://doi.org/10.1007/s00223-012-9671-9
Inayoshi A, Sugimoto Y, Funahashi J, et al., 2011. Mechanism underlying the block of human Cav3.2 T-type Ca2+ channels by benidipine, a dihydropyridine Ca2+ channel blocker. Life Sci, 88(19–20):898–907. https://doi.org/10.1016/j.lfs.2011.03.019
Kosaka H, Hirayama K, Yoda N, et al., 2010. The L-, N-, and T-type triple calcium channel blocker benidipine acts as an antagonist of mineralocorticoid receptor, a member of nuclear receptor family. Eur J Pharmacol, 635(1–3):49–55. https://doi.org/10.1016/j.ejphar.2010.03.018
Larijani B, Bekheirnia MR, Soltani A, et al., 2004. Bone mineral density is related to blood pressure in men. Am J Hum Biol, 16(2):168–171. https://doi.org/10.1002/ajhb.20005
Lee KS, Hong SH, Bae SC, 2002. Both the Smad and p38 MAPK pathways play a crucial role in Runx2 expression following induction by transforming growth factor- β and bone morphogenetic protein. Oncogene, 21(47):7156–7163. https://doi.org/10.1038/sj.onc.1205937
Lim LS, Fink HA, Blackwell T, et al., 2009. Loop diuretic use and rates of hip bone loss and risk of falls and fractures in older women. J Am Geriatr Soc, 57(5): 855–862. https://doi.org/10.1111/j.1532-5415.2009.02195.x
Liu L, Wang D, Qin Y, et al., 2019. Astragalin promotes osteoblastic differentiation in MC3T3-E1 cells and bone formation in vivo. Front Endocrinol, 10:228. https://doi.org/10.3389/fendo.2019.00228
Lynn H, Kwok T, Wong SYS, et al., 2006. Angiotensin converting enzyme inhibitor use is associated with higher bone mineral density in elderly Chinese. Bone, 38(4):584–588. https://doi.org/10.1016/j.bone.2005.09.011
Ma J, Wang Z, Zhao JQ, et al., 2018. Resveratrol attenuates lipopolysaccharides (LPS)-induced inhibition of osteoblast differentiation in MC3T3-E1 cells. Med Sci Monit, 24: 2045–2052. https://doi.org/10.12659/msm.905703
Ma JY, You D, Li WY, et al., 2019. Bone morphogenetic proteins and inner ear development. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 20(2):131–145. https://doi.org/10.1631/jzus.B1800084
Ma ZP, Liao JC, Zhao C, et al., 2015. Effects of the 1,4-dihydropyridine L-type calcium channel blocker benidipine on bone marrow stromal cells. Cell Tissue Res, 361(2): 467–476. https://doi.org/10.1007/s00441-015-2115-x
Metz JA, Morris CD, Roberts LA, et al., 1999. Blood pressure and calcium intake are related to bone density in adult males. Br J Nutr, 81(5):383–388. https://doi.org/10.1017/S0007114599000665
Mundy GR, Chen D, Zhao M, et al., 2001. Growth regulatory factors and bone. Rev Endocr Metab Disord, 2(1):105–115. https://doi.org/10.1023/a:1010015309973
Neve A, Corrado A, Cantatore FP, 2013. Osteocalcin: skeletal and extra-skeletal effects. J Cell Physiol, 228(6):1149–1153. https://doi.org/10.1002/jcp.24278
Nishiya Y, Sugimoto S, 2001. Effects of various antihypertensive drugs on the function of osteoblast. Biol Pharm Bull, 24(6):628–633. https://doi.org/10.1248/bpb.24.628
Nishiya Y, Kosaka N, Uchii M, et al., 2002. A potent 1,4-dihydropyridine L-type calcium channel blocker, benidipine, promotes osteoblast differentiation. Calcif Tissue Int, 70(1):30–39. https://doi.org/10.1007/s00223-001-1010-5
Noël D, Gazit D, Bouquet C, et al., 2004. Short-term BMP-2 expression is sufficient for in vivo osteochondral differentiation of mesenchymal stem cells. Stem Cells, 22(1):74–85. https://doi.org/10.1634/stemcells.22-1-74
Shimizu H, Nakagami H, Yasumasa N, et al., 2012. Links between hypertension and osteoporosis: benidipine ameliorates osteoporosis in ovariectomized hypertensive rats through promotion of osteoblast proliferation and inhibition of osteoclast differentiation. Curr Cardiovasc Risk Rep, 6(4):274–280. https://doi.org/10.1007/s12170-012-0248-y
Sims NA, Gooi JH, 2008. Bone remodeling: multiple cellular interactions required for coupling of bone formation and resorption. Semin Cell Dev Biol, 19(5):444–451. https://doi.org/10.1016/j.semcdb.2008.07.016
Sudo H, Kodama HA, Amagai Y, et al., 1983. In vitro differentiation and calcification in a new clonal osteogenic cell line derived from newborn mouse calvaria. J Cell Biol, 96(1):191–198. https://doi.org/10.1083/jcb.96.1.191
Suzuki H, Yokoyama K, Akimoto Y, et al., 2007. Clinical efficacy of benidipine for vasospastic angina pectoris. Arzneimittelforschung, 57(1):20–25. https://doi.org/10.1055/s-0031-1296581
Teti A, 2011. Bone development: overview of bone cells and signaling. Curr Osteoporos Rep, 9(4):264–273. https://doi.org/10.1007/s11914-011-0078-8
Wang BX, Bi M, Zhu Z, et al., 2014. Effects of the antihypertensive drug benidipine on osteoblast function in vitro. Exp Ther Med, 7(3):649–653. https://doi.org/10.3892/etm.2014.1475
Wang ZW, Chen Z, Zhang LF, et al., 2018. Status of hypertension in China: results from the China hypertension survey, 2012–2015. Circulation, 137(22):2344–2356. https://doi.org/10.1161/CIRCULATIONAHA.117.032380
Wu MR, Chen GQ, Li YP, 2016. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res, 4:16009. https://doi.org/10.1038/boneres.2016.9
Xue C, Zhou CC, Yang B, et al., 2017. Comparison of efficacy and safety between benidipine and hydrochlorothiazide in fosinopril-treated hypertensive patients with chronic kidney disease: protocol for a randomised controlled trial. BMJ Open, 7(2):e013672. https://doi.org/10.1136/bmjopen-2016-013672
Yang SM, Nguyen ND, Eisman JA, et al., 2012. Association between beta-blockers and fracture risk: a Bayesian meta-analysis. Bone, 51(5):969–974. https://doi.org/10.1016/j.bone.2012.07.013
Yao K, Nagashima K, Miki H, 2006. Pharmacological, pharmacokinetic, and clinical properties of benidipine hydrochloride, a novel, long-acting calcium channel blocker. J Pharmacol Sci, 100(4):243–261. https://doi.org/10.1254/jphs.dtj05001x
Yu D, Wang J, Qian KJ, et al., 2020. Effects of nanofibers on mesenchymal stem cells: environmental factors affecting cell adhesion and osteogenic differentiation and their mechanisms. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(11):871–884. https://doi.org/10.1631/jzus.B2000355
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 81600909 and 81800934) and the Zhejiang Provincial Medical Science and Technology Project of China (Nos. 2017RC009, 2018RC012, and 2021KY773).
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Baixiang WANG, Jiakang YANG, and Yu WANG performed the experimental research and data analysis, wrote and edited the manuscript. Chenqiu ZHANG created the figures. Lijie FAN and Huiming WANG contributed to the study design, data analysis, and discussion. All authors have read and approved the final manuscript and, therefore, have full access to all data relevant to the study and take responsibility for the integrity and security of such data.
Compliance with ethics guidelines
Baixiang WANG, Jiakang YANG, Lijie FAN, Yu WANG, Chenqiu ZHANG, and Huming WANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Wang, B., Yang, J., Fan, L. et al. Osteogenic effects of antihypertensive drug benidipine on mouse MC3T3-E1 cells in vitro. J. Zhejiang Univ. Sci. B 22, 410–420 (2021). https://doi.org/10.1631/jzus.B2000628
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000628