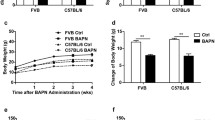
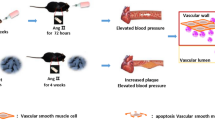
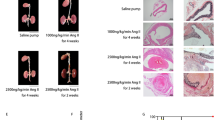
Abstract
Thoracic aortic dissection (TAD) is one of the most lethal aortic diseases due to its acute onset, rapid progress, and high rate of aortic rupture. The pathogenesis of TAD is not completely understood. In this mini-review, we introduce three emerging experimental mouse TAD models using β-aminopropionitrile (BAPN) alone, BAPN for a prolonged duration (four weeks) and then with added infusion of angiotensin II (AngII), or co-administration of BAPN and AngII chronically. We aim to provide insights into appropriate application of these three mouse models, thereby enhancing the understanding of the molecular mechanisms of TAD.
概 要
胸主动脉夹层具有发病急、 发展迅速、 主动脉破裂率高的特点, 是最致命的大动脉疾病之一. 但是胸主动脉夹层的发病机制目前还没有被完全了解. 本综述介绍了三种新兴的β-氨基丙腈相关的小鼠胸主动脉夹层模型, 分别是: 单用β-氨基丙腈; 先用β-氨基丙腈处理 (四周) 再用血管紧张素 2 (AngII) 处理; β-氨基丙腈和 AngII 同时进行处理. 希望通过更好地运用这三种β-氨基丙腈相关的小鼠胸主动脉夹层模型, 从而对胸主动脉夹层的分子机制有更深入的了解.
Similar content being viewed by others
References
Anzai A, Shimoda M, Endo J, et al., 2015. Adventitial CXCL1/G-CSF expression in response to acute aortic dissection triggers local neutrophil recruitment and activation leading to aortic rupture. Circ Res, 116(4):612–623. https://doi.org/10.1161/CIRCRESAHA.116.304918
Behmoaras J, Slove S, Seve S, et al., 2008. Differential expression of lysyl oxidases LOXL1 and LOX during growth and aging suggests specific roles in elastin and collagen fiber remodeling in rat aorta. Rejuvenation Res, 11(5): 883–889. https://doi.org/10.1089/rej.2008.0760
Elefteriades JA, Farkas EA, 2010. Thoracic aortic aneurysm: clinically pertinent controversies and uncertainties. J Am Coll Cardiol, 55(9):841–857. https://doi.org/10.1016/j.jacc.2009.08.084
Elsayed RS, Cohen RG, Fleischman F, et al., 2017. Acute type A aortic dissection. Cardiol Clin, 35(3):331–345. https://doi.org/10.1016/j.ccl.2017.03.004
English SJ, Piert MR, Diaz JA, et al., 2015. Increased 18F-FDG uptake is predictive of rupture in a novel rat abdominal aortic aneurysm rupture model. Ann Surg, 261(2): 395–404. https://doi.org/10.1097/SLA.0000000000000602
Fhayli W, Boëté Q, Kihal N, et al., 2020. Dill extract induces elastic fiber neosynthesis and functional improvement in the ascending aorta of aged mice with reversal of age-dependent cardiac hypertrophy and involvement of lysyl oxidase-like-1. Biomolecules, 10(2):173. https://doi.org/10.3390/biom10020173
Gao YX, Wang ZZ, Zhao JQ, et al., 2019. Involvement of B cells in the pathophysiology of β-aminopropionitrile-induced thoracic aortic dissection in mice. Exp Anim, 68(3):331–339. https://doi.org/10.1538/expanim.18-0170
Gawinecka J, Schönrath F, von Eckardstein A, 2017. Acute aortic dissection: pathogenesis, risk factors and diagnosis. Swiss Med Wkly, 147:w14489. https://doi.org/10.4414/smw.2017.14489
Guo DC, Papke CL, He RM, et al., 2006. Pathogenesis of thoracic and abdominal aortic aneurysms. Ann N Y Acad Sci, 1085(1):339–352. https://doi.org/10.1196/annals.1383.013
Han L, Dai L, Zhao YF, et al., 2018. CD40L promotes development of acute aortic dissection via induction of inflammation and impairment of endothelial cell function. Aging (Albany NY), 10(3):371–385. https://doi.org/10.18632/aging.101394
Howard DP, Banerjee A, Fairhead JF, et al., 2013. Population-based study of incidence and outcome of acute aortic dissection and premorbid risk factor control: 10-year results from the Oxford Vascular Study. Circulation, 127(20): 2031–2037. https://doi.org/10.1161/CIRCULATIONAHA.112.000483
Hu CK, Tan H, Lin QY, et al., 2019. SPECT/CT imaging of apoptosis in aortic aneurysm with radiolabeled duramycin. Apoptosis, 24(9–10):745–755. https://doi.org/10.1007/s10495-019-01554-8
Imanishi M, Chiba Y, Tomita N, et al., 2016. Hypoxia-inducible factor-1α in smooth muscle cells protects against aortic aneurysms—brief report. Arterioscler Thromb Vasc Biol, 36(11):2158–2162. https://doi.org/10.1161/ATVBAHA.116.307784
Imanishi M, Izawa-Ishizawa Y, Sakurada T, et al., 2018. Nitrosonifedipine, a photodegradation product of nifedipine, suppresses pharmacologically induced aortic aneurysm formation. Pharmacology, 102(5–6):287–299. https://doi.org/10.1159/000492577
Iwatsuki K, Cardinale GJ, Spector S, et al., 1977. Reduction of blood pressure and vascular collagen in hypertensive rats by β-aminopropionitrile. Proc Natl Acad Sci USA, 74(1): 360–362. https://doi.org/10.1073/pnas.74.1.360
Izawa-Ishizawa Y, Imanishi M, Zamami Y, et al., 2019. Development of a novel aortic dissection mouse model and evaluation of drug efficacy using in-vivo assays and database analyses. J Hypertens, 37(1):73–83. https://doi.org/10.1097/HJH.0000000000001898
Jia LX, Zhang WM, Zhang HJ, et al., 2015. Mechanical stretch-induced endoplasmic reticulum stress, apoptosis and inflammation contribute to thoracic aortic aneurysm and dissection. J Pathol, 236(3):373–383. https://doi.org/10.1002/path.4534
Jia LX, Zhang WM, Li TT, et al., 2017. ER stress dependent microparticles derived from smooth muscle cells promote endothelial dysfunction during thoracic aortic aneurysm and dissection. Clin Sci (Lond), 131(12):1287–1299. https://doi.org/10.1042/CS20170252
Johnston WF, Salmon M, Pope NH, et al., 2014. Inhibition of interleukin-1 β decreases aneurysm formation and progression in a novel model of thoracic aortic aneurysms. Circulation, 130(11_Suppl_1):S51–S59. https://doi.org/10.1161/CIRCULATIONAHA.113.006800
Jones JA, Ruddy JM, Bouges S, et al., 2010. Alterations in membrane type-1 matrix metalloproteinase abundance after the induction of thoracic aortic aneurysm in a murine model. Am J Physiol Heart Circ Physiol, 299(1):H114–H124. https://doi.org/10.1152/ajpheart.00028.2010
Kanematsu Y, Kanematsu M, Kurihara C, et al., 2010. Pharmacologically induced thoracic and abdominal aortic aneurysms in mice. Hypertension, 55(5):1267–1274. https://doi.org/10.1161/HYPERTENSIONAHA.109.140558
Kawai T, Takayanagi T, Forrester SJ, et al., 2017. Vascular ADAM17 (a disintegrin and metalloproteinase domain 17) is required for angiotensin II/β-aminopropionitrile-induced abdominal aortic aneurysm. Hypertension, 70(5):959–963. https://doi.org/10.1161/HYPERTENSIONAHA.117.09822
Kurihara T, Shimizu-Hirota R, Shimoda M, et al., 2012. Neutrophil-derived matrix metalloproteinase 9 triggers acute aortic dissection. Circulation, 126(25):3070–3080. https://doi.org/10.1161/CIRCULATIONAHA.112.097097
Kurobe H, Matsuoka Y, Hirata Y, et al., 2013a. Azelnidipine suppresses the progression of aortic aneurysm in wild mice model through anti-inflammatory effects. J Thorac Cardiovasc Surg, 146(6):1501–1508. https://doi.org/10.1016/j.jtcvs.2013.02.073
Kurobe H, Hirata Y, Matsuoka Y, et al., 2013b. Protective effects of selective mineralocorticoid receptor antagonist against aortic aneurysm progression in a novel murine model. J Surg Res, 185(1):455–462. https://doi.org/10.1016/joss.2013.05.002
Landenhed M, Engström G, Gottsäter A, et al., 2015. Risk profiles for aortic dissection and ruptured or surgically treated aneurysms: a prospective cohort study. J Am Heart Assoc, 4(1):e001513. https://doi.org/10.1161/JAHA.114.001513
Lee VS, Halabi CM, Hoffman EP, et al., 2016. Loss of function mutation in LOX causes thoracic aortic aneurysm and dissection in humans. Proc Natl Acad Sci USA, 113(31): 8759–8764. https://doi.org/10.1073/pnas.1601442113
Levene CI, 1962. Studies on the mode of action of lathyrogenic compounds. J Exp Med, 116(2):119–130. https://doi.org/10.1084/jem.116.2.119
Li JS, Li HY, Wang L, et al., 2013. Comparison of β-aminopropionitrile-induced aortic dissection model in rats by different administration and dosage. Vascular, 21(5): 287–292. https://doi.org/10.1177/1708538113478741
Li ZQ, Zhao ZQ, Cai ZY, et al., 2020. Runx2 (runt-related transcription factor 2)-mediated microcalcification is a novel pathological characteristic and potential mediator of abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol, 40(5):1352–1369. https://doi.org/10.1161/ATVBAHA.119.314113
MacFarlane EG, Parker SJ, Shin JY, et al., 2019. Lineage-specific events underlie aortic root aneurysm pathogenesis in Loeys-Dietz syndrome. J Clin Invest, 129(2):659–675. https://doi.org/10.1172/JCI123547
McClure RS, Brogly SB, Lajkosz K, et al., 2018. Epidemiology and management of thoracic aortic dissections and thoracic aortic aneurysms in Ontario, Canada: a population-based study. J Thorac Cardiovasc Surg, 155(6):2254–2264.e4. https://doi.org/10.1016/j.jtcvs.2017.11.105
Milewicz DM, Prakash SK, Ramirez F, 2017. Therapeutics targeting drivers of thoracic aortic aneurysms and acute aortic dissections: insights from predisposing genes and mouse models. Annu Rev Med, 68:51–67. https://doi.org/10.1146/annurev-med-100415-022956
Narayanan AS, Siegel RC, Martin GR, 1972. On the inhibition of lysyl oxidase by β-aminopropionitrile. Biochem Biophys Res Commun, 46(2):745–751. https://doi.org/10.1016/s0006-291x(72)80203-1
Nienaber CA, Clough RE, 2015. Management of acute aortic dissection. Lancet, 385(9970):800–811. https://doi.org/10.1016/S0140-6736(14)61005-9
Obama T, Tsuji T, Kobayashi T, et al., 2015. Epidermal growth factor receptor inhibitor protects against abdominal aortic aneurysm in a mouse model. Clin Sci (Lond), 128(9): 559–565. https://doi.org/10.1042/CS20140696
O’Dell BL, Elsden DF, Thomas J, et al., 1966. Inhibition of the biosynthesis of the crosslinks in elastin by a lathyrogen. Nature, 209(5021):401–402. https://doi.org/10.1038/209401a0
Oller J, Méndez-Barbero N, Ruiz EJ, et al., 2017. Nitric oxide mediates aortic disease in mice deficient in the metalloprotease Adamts1 and in a mouse model of Marfan syndrome. Nat Med, 23(2):200–212. https://doi.org/10.1038/nm.4266
Péterszegi G, Andrès E, Molinari J, et al., 2008. Effect of cellular aging on collagen biosynthesis: I. Methodological considerations and pharmacological applications. Arch Gerontol Geriatr, 47(3):356–367. https://doi.org/10.1016/j.archger.2007.08.019
Pinnell SR, Martin GR, 1968. The cross-linking of collagen and elastin: enzymatic conversion of lysine in peptide linkage to α-aminoadipic-δ-semialdehyde (allysine) by an extract from bone. Proc Natl Acad Sci USA, 61(2):708–716. https://doi.org/10.1073/pnas.61.2.708
Ponseti IV, Baird WA, 1952. Scoliosis and dissecting aneurysm of the aorta in rats fed with Lathyrus odoratus seeds. Am J Pathol, 28(6):1059–1077.
Ren WH, Liu Y, Wang XR, et al., 2016. β-Aminopropionitrile monofumarate induces thoracic aortic dissection in C57BL/6 mice. Sci Rep, 6:28149. https://doi.org/10.1038/srep28149
Ren WH, Liu Y, Wang XR, et al., 2018. The complement C3a-C3aR axis promotes development of thoracic aortic dissection via regulation of MMP2 expression. J Immunol, 200(5):1829–1838. https://doi.org/10.4049/jimmunol.1601386
Sakai LY, Keene DR, Renard M, et al., 2016. FBN1: the disease-causing gene for Marfan syndrome and other genetic disorders. Gene, 591(1):279–291. https://doi.org/10.1016/j.gene.2016.07.033
Saraff K, Babamusta F, Cassis LA, et al., 2003. Aortic dissection precedes formation of aneurysms and atherosclerosis in angiotensin II-infused, apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol, 23(9):1621–1626. https://doi.org/10.1161/01.ATV.0000085631.76095.64
Schilling ED, Strong FM, 1954. Isolation, structure and synthesis of a Lathyrus factor from L. odoratus. J Am Chem Soc, 76:2848. https://doi.org/10.1111/j.1753-4887.1976.tb05779.x
Suehiro C, Suzuki J, Hamaguchi M, et al., 2019. Deletion of interleukin-18 attenuates abdominal aortic aneurysm formation. Atherosclerosis, 289:14–20. https://doi.org/10.1016/j.atherosclerosis.2019.08.003
Takayanagi T, Crawford KJ, Kobayashi T, et al., 2014. Caveolin 1 is critical for abdominal aortic aneurysm formation induced by angiotensin II and inhibition of lysyl oxidase. Clin Sci (Lond), 126(11):785–800. https://doi.org/10.1042/CS20130660
Tieu BC, Lee C, Sun H, et al., 2009. An adventitial IL-6/MCP1 amplification loop accelerates macrophage-mediated vascular inflammation leading to aortic dissection in mice. J Clin Invest, 119(12):3637–3651. https://doi.org/10.1172/JCI38308
Tomida S, Aizawa K, Nishida N, et al., 2019. Indomethacin reduces rates of aortic dissection and rupture of the abdominal aorta by inhibiting monocyte/macrophage accumulation in a murine model. Sci Rep, 9:10751. https://doi.org/10.1038/s41598-019-46673-z
van Laer L, Dietz H, Loeys B, 2014. Loeys-dietz syndrome. In: Halper J (Ed.), Progress in Heritable Soft Connective Tissue Diseases. Springer, Dordrecht, p.95–105. https://doi.org/10.1007/978-94-007-7893-1_7
Waibel PE, Lovelady HG, Liener IE, 1964. Influence of beta-aminopropionitrile on dissecting aneurysm and on plasma amino acids in the Turkey. Metabolism, 13(5):473–479. https://doi.org/10.1016/0026-0495(64)90121-0
Wang SS, Liu YT, Zhao GZ, et al., 2018. Postnatal deficiency of ADAMTS1 ameliorates thoracic aortic aneurysm and dissection in mice. Exp Physiol, 103(12):1717–1731. https://doi.org/10.1113/EP087018
Wang Y, Zhao ZM, Zhang GX, et al., 2016. Dynamic autophagic activity affected the development of thoracic aortic dissection by regulating functional properties of smooth muscle cells. Biochem Biophys Res Commun, 479(2):358–364. https://doi.org/10.1016/j.bbrc.2016.09.080
Wawzonek S, Ponseti IV, Shepard RS, et al., 1955. Epiphyseal plate lesions, degenerative arthritis, and dissecting aneurysm of the aorta produced by aminonitriles. Science, 121(3133):63–65. https://doi.org/10.1126/science.121.3133.63
Xu K, Xu C, Zhang YZZ, et al., 2018. Identification of type IV collagen exposure as a molecular imaging target for early detection of thoracic aortic dissection. Theranostics, 8(2): 437–449. https://doi.org/10.7150/thno.22467
Yanagisawa H, Wagenseil J, 2020. Elastic fibers and biomechanics of the aorta: insights from mouse studies. Matrix Biol, 85–86:160–172. https://doi.org/10.1016/j.matbio.2019.03.001
Zhang L, Pei YF, Wang L, et al., 2012. Dramatic decrease of aortic longitudinal elastic strength in a rat model of aortic dissection. Ann Vasc Surg, 26(7):996–1001. https://doi.org/10.1016/j.avsg.2012.02.004
Zhao ZM, Wang Y, Li SH, et al., 2019. HSP90 inhibitor 17-DMAG effectively alleviated the progress of thoracic aortic dissection by suppressing smooth muscle cell phenotypic switch. Am J Transl Res, 11(1):509–518.
Zhou B, Li W, Zhao GZ, et al., 2019. Rapamycin prevents thoracic aortic aneurysm and dissection in mice. J Vasc Surg, 69(3):921–932.e3. https://doi.org/10.1016/j.jvs.2018.05.246
Acknowledgments
The authors thank all Jian-an WANG laboratory members for critical comments on the manuscript.
Author information
Authors and Affiliations
Contributions
Jian-an WANG and Hong S. LU provided the theme and design, and edited the manuscript. Hai-qiong ZHENG participated in searching and summarizing the relevant literature as well as designing and writing the manuscript. Jia-bing RONG, Fei-ming YE, and Yin-chuan XU provided comments and edited the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Hai-qiong ZHENG, Jia-bing RONG, Fei-ming YE, Yin-chuan XU, Hong S. LU, and Jian-an WANG declare that they have no conflicts of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Project supported by the National Natural Science Foundation of China (Nos. 81870292 and 81971860) and the National Key Research and Development Program of China (No. 2016YFC1301204)
Rights and permissions
About this article
Cite this article
Zheng, Hq., Rong, Jb., Ye, Fm. et al. Induction of thoracic aortic dissection: a mini-review of β-aminopropionitrile-related mouse models. J. Zhejiang Univ. Sci. B 21, 603–610 (2020). https://doi.org/10.1631/jzus.B2000022
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000022
Key words
- Thoracic aortic dissection (TAD)
- β-Aminopropionitrile (BAPN)
- Angiotensin II (AngII)
- Mouse model
- Hypertension