Abstract
Objective
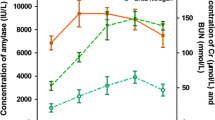
This study demonstrated that dexamethasone (DEX) protects the endothelial glycocalyx from damage induced by the inflammatory stimulus tumor necrosis factor-α (TNF-α) during severe acute pancreatitis (SAP), and improves the renal microcirculation.
Methods
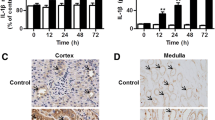
Ninety mice were evenly divided into 3 groups (Sham, SAP, and SAP+DEX). The SAP mice model was established by ligature of pancreatic duct and intraperitoneal injection of cerulein. Renal perfusion and function, and morphological changes of the glycocalyx were evaluated by laser Doppler velocimetry, electron microscopy, and histopathology (hematoxylin and eosin (H&E) staining), respectively. Serum levels of syndecan-1 and TNF-α were assessed by enzyme-linked immunosorbent assay (ELISA). The protective effects of dexamethasone on the glycocalyx and renal microcirculation were evaluated.
Results
Significantly high levels of serum TNF-α were detected 3 h after the onset of SAP. These levels might induce degradation of the glycocalyx and kidney hypoperfusion, resulting in kidney microcirculation dysfunction. The application of dexamethasone reduced the degradation of the glycocalyx and improved perfusion of kidney.
Conclusions
Dexamethasone protects the endothelial glycocalyx from inflammatory degradation possibly initiated by TNF-α during SAP. This is might be a significant discovery that helps to prevent tissue edema and hypoperfusion in the future.
概 要
目 的
明确地塞米松可以减少重症急性胰腺炎 (SAP) 引起的肿瘤坏死因子 (TNF-α) 的释放, 减轻 TNF-α 导致的肾脏血管内皮糖萼的降解, 从而改善肾脏微循环和缓解肾损伤。
创新点
本研究通过小鼠活体研究的方法, 建立小鼠重症急性胰腺炎模型, 并用地塞米松进行干预对照, 采用透射电镜、激光多谱勒和酶联免疫的方法, 检测了各组小鼠肾脏血管内皮糖萼的完整性、肾血流灌注和 TNF-α 表达情况, 阐明了地塞米松对内皮糖萼的保护作用。
方 法
通过 “胰管结扎+腹腔内雨蛙素注射” 的方法建立SAP 模型, 分别留取各组小鼠的血液和组织标本, 采用透射电镜观察内皮糖萼的损伤情况, 用酶联免疫检测血清 TNF-α 和糖萼成份多配体聚糖的浓度, 并用激光多谱勒检测活体小鼠肾脏的灌注, 分析地塞米松对内皮糖萼的保护和改善肾脏灌注的作用。
结 论
SAP 可以引起 TNF-α 的大量释放, 并导致内皮糖萼的降解和肾脏灌注下降, 而地塞米松可以减少 TNF-α 的释放, 减轻糖萼的降解, 改善肾脏血流灌注。
Similar content being viewed by others
References
Barabutis N, Khangoora V, Marik PE, et al., 2017. Hydrocortisone and ascorbic acid synergistically prevent and repair lipopolysaccharide-induced pulmonary endothelial barrier dysfunction. Chest, 152(5):954–962. https://doi.org/10.1016/jxhest.2017.07.014
Chappell D, Dörfler N, Jacob M, et al., 2010. Glycocalyx protection reduces leukocyte adhesion after ischemia/reperfusion. Shock, 34(2):133–139. https://doi.org/10.1097/SHK.0b013e3181cdc363
Curry FRE, Adamson RH, 2010. Vascular permeability modulation at the cell, microvessel, or whole organ level: towards closing gaps in our knowledge. Cardiovasc Res, 87(2):218–229. https://doi.org/10.1093/cvr/cvq115
de Leeuw K, Niemeijer AS, Eshuis J, et al., 2016. Effect and mechanism of hydrocortisone on organ function in patients with severe burns. J Crit Care, 36:200–206. https://doi.org/10.1016/j.jcrc.2016.06.007
Ergin B, Kapucu A, Demirci-Tansel C, et al., 2015. The renal microcirculation in sepsis. Nephrol Dial Transplant, 30(2):169–177. https://doi.org/10.1093/ndt/gfu105
Gao SL, Zhang Y, Zhang SY, et al., 2015. The hydrocortisone protection of glycocalyx on the intestinal capillary endothelium during severe acute pancreatitis. Shock, 43(5): 512–517. https://doi.org/10.1097/SHK.0000000000000326
Glasner DR, Ratnasiri K, Puerta-Guardo H, et al., 2017. Dengue virus NS1 cytokine-independent vascular leak is dependent on endothelial glycocalyx components. PLoS Pathog, 13(11):e1006673. https://doi.org/10.1371/journal.ppat.1006673
Henry CB, Duling BR, 2000. TNF-α increases entry of macromolecules into luminal endothelial cell glycocalyx. Am J Physiol Heart Circ Physiol, 279(6):H2815–H2823. https://doi.org/10.1152/ajpheart.2000.279.6.H2815
Ince C, 2014. The central role of renal microcirculatory dysfunction in the pathogenesis of acute kidney injury. Nephron Clin Pract, 127(1–4):124–128. https://doi.org/10.1159/000363203
Kes P, Vučičević Ž, Ratković-Gusić I, et al., 1996. Acute renal failure complicating severe acute pancreatitis. Ren Fail, 18(4):621–628. https://doi.org/10.3109/08860229609047686
Lankisch PG, Apte M, Banks PA, 2015. Acute pancreatitis. Lancet, 386(9988):85–96. https://doi.org/10.1016/S0140-6736(14)60649-8
Levick JR, Michel CC, 2010. Microvascular fluid exchange and the revised starling principle. Cardiovasc Res, 87(2): 198–210. https://doi.org/10.1093/cvr/cvq062
Nieuwdorp M, Meuwese MC, Mooij HL, et al., 2009. Tumor necrosis factor-α inhibition protects against endotoxin-induced endothelial glycocalyx perturbation. Atherosclerosis, 202(1):296–303. https://doi.org/10.1016/j.atherosclerosis.2008.03.024
Pavlidis P, Crichton S, Lemmich Smith J, et al., 2013. Improved outcome of severe acute pancreatitis in the intensive care unit. Crit Care Res Pract, 2013:897107. https://doi.org/10.1155/2013/897107
Rhodes A, Evans LE, Alhazzani W, et al., 2017. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med, 45(3):486–552. https://doi.org/10.1097/CCM.0000000000002255
Rosenberg RD, Shworak NW, Liu J, et al., 1997. Heparan sulfate proteoglycans of the cardiovascular system. Specific structures emerge but how is synthesis regulated? J Clin Invest, 100(S11):S67–S75.
Schmidt J, Rattner DW, Lewandrowski K, et al., 1992. A better model of acute pancreatitis for evaluating therapy. Ann Surg, 215(1):44–56. https://doi.org/10.1097/00000658-199201000-00007
Singh DB, Stansby G, Bain I, et al., 2009. Intraoperative measurement of colonic oxygenation during bowel resection. In: Liss P, Hansell P, Bruley DF, et al. (Eds.), Oxygen Transport to Tissue XXX. Springer, Boston, MA, p.261–266. https://doi.org/10.1007/978-0-387-85998-9_39
van Golen RF, van Gulik TM, Heger M, 2012. Mechanistic overview of reactive species-induced degradation of the endothelial glycocalyx during hepatic ischemia/reperfusion injury. Free Radic Biol Med, 52(8):1382–1402. https://doi.org/10.1016/j.freeradbiomed.2012.01.013
van Golen RF, Reiniers MJ, Vrisekoop N, et al., 2014. The mechanisms and physiological relevance of glycocalyx degradation in hepatic ischemia/reperfusion injury. Antioxid Redox Signal, 21(7):1098–1118. https://doi.org/10.1089/ars.2013.5751
Verma SK, Molitoris BA, 2015. Renal endothelial injury and microvascular dysfunction in acute kidney injury. Semin Nephrol, 35(1):96–107. https://doi.org/10.1016/j.semnephrol.2015.01.010
Wan L, Bagshaw SM, Langenberg C, et al., 2008. Pathophysiology of septic acute kidney injury: what do we really know? Crit Care Med, 36(S4):S198–S203. https://doi.org/10.1097/CCM.0b013e318168ccd5
Zhang J, Yao Y, Xiao F, et al., 2013. Administration of dexamethasone protects mice against ischemia/reperfusion induced renal injury by suppressing PI3K/AKT signaling. Int J Clin Exp Pathol, 6(11):2366–2375.
Zheng X, Feng B, Chen G, et al., 2006. Preventing renal ischemia-reperfusion injury using small interfering RNA by targeting complement 3 genes. Am J Transplant, 6(9): 2099–2108. https://doi.org/10.1111/j.1600-6143.2006.01427.x
Zhou JJ, Li Y, Tang Y, et al., 2015. Effect of acute kidney injury on mortality and hospital stay in patient with severe acute pancreatitis. Nephrology (Carlton), 20(7):485–491. https://doi.org/10.1111/nep.12439
Author information
Authors and Affiliations
Corresponding authors
Additional information
Project supported by the National Natural Science Foundation of China (No. 81501644)
Rights and permissions
About this article
Cite this article
Yu, Wq., Zhang, Sy., Fu, Sq. et al. Dexamethasone protects the glycocalyx on the kidney microvascular endothelium during severe acute pancreatitis. J. Zhejiang Univ. Sci. B 20, 355–362 (2019). https://doi.org/10.1631/jzus.B1900006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900006
Key words
- Severe acute pancreatitis (SAP)
- Acute kidney injury (AKI)
- Glycocalyx
- Dexamethasone
- Tumor necrosis factor-α (TNF-α)