Abstract
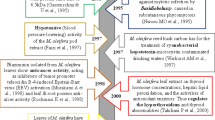
Chlortetracycline (CTC), one kind of common antibiotic for prevention and treatment of various diseases, also exhibits good performance in accelerating the growth of livestock. Macleaya cordata, a traditional Chinese medicine, is usually used as a natural additive in livestock because of its anti-microbial, anti-fungal, anti-inflammatory, and pesticidal activity. In this work, we studied whether M. cordata helps regulate the growth-promoting effect of CTC on broiler chickens. It is demonstrated that M. cordata improves the growth-promoting effect of CTC on growth performance indices of broiler chickens, such as survival rate, daily weight, and feed to weight rate. M. cordata also delays the maximum of CTC residues in plasma. It may depend on the higher values of operational taxonomic unit (OTU) and the indices of α diversity driven by simultaneous use of CTC and M. cordata.
中文概要
目的
通过同时与金霉素饲喂,验证博落回在肉鸡生长促进中的作用。
创新点
通过博落回与金霉素的同时饲喂改进肉鸡生长, 优化肉鸡表观指标的同时,检测体内金霉素残留,提高肉鸡饲养的经济利益。
方法
在肉鸡饲养过程中记录体重和采食量等基础数据,通过换算并比较存活率、日增重及料重比, 进行肉鸡生长表观指标的比较;在给药饲喂过程中以及结束后,采取动物的组织及血液,通过高 效液相色谱法和高效液相色谱-串联质谱法检测 金霉素含量,同时应用16S 测序检测饲喂后十二指肠肠道微生物菌群组成结构。
结论
证实博落回虽然增加了肝脏中金霉素残留量,但可以通过调节十二指肠肠道微生物菌群来提高 金霉素的促生长作用。
Similar content being viewed by others
References
Aarestrup FM, Bager F, Jensen NE, et al., 1998. Resistance to antimicrobial agents used for animal therapy in pathogenic-, zoonotic-and indicator bacteria isolated from different food animals in Denmark: a baseline study for the Danish Integrated Antimicrobial Resistance Monitoring Programme (DANMAP). APMIS, 106(7-12):745–770. https://doi.org/10.1111/j.1699-0463.1998.tb00222.x
Anadón A, Gamboa F, Martínez MA, et al., 2012. Plasma disposition and tissue depletion of chlortetracycline in the food producing animals, chickens for fattening. Food Chem Toxicol, 50(8):2714–2721. https://doi.org/10.1016/j.fct.2012.05.007
Cerf-Bensussan N, Gaboriau-Routhiau V, 2010. The immune system and the gut microbiota: friends or foes? Nat Rev Immunol, 10(10):735–744. https://doi.org/10.1038/nri2850
Chopra I, Roberts M, 2001. Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol Biol Rev, 65(2): 232–260. https://doi.org/10.1128/MMBR.65.2.232-260.2001
de Ruyck H, de Ridder H, van Renterghem R, et al., 1999. Validation of HPLC method of analysis of tetracycline residues in eggs and broiler meat and its application to a feeding trial. Food Addit Contam, 16(2):47–56. https://doi.org/10.1080/026520399284190
Dibner JJ, Richards JD, 2005. Antibiotic growth promoters in agriculture: history and mode of action. Poultry Sci, 84(4):634–643. https://doi.org/10.1093/ps/84.4.634
Gaskins HR, Collier CT, Anderson DB, 2002. Antibiotics as growth promotants: mode of action. Anim Biotechnol, 13(1):29–42. https://doi.org/10.1081/ABIO-120005768
Gerritsen J, Smidt H, Rijkers GT, et al., 2011. Intestinal microbiota in human health and disease: the impact of probiotics. Genes Nutr, 6(3):209–240. https://doi.org/10.1007/s12263-011-0229-7
Gu N, Ge KL, Hao C, et al., 2017. Neuregulin1β effects on brain tissue via ERK5-dependent MAPK pathway in a rat model of cerebral ischemia-reperfusion injury. J Mol Neurosci, 61(4):607–616. https://doi.org/10.1007/s12031-017-0902-4
Hildebrand F, Nguyen TLA, Brinkman B, et al., 2013. Inflammation-associated enterotypes, host genotype, cage and inter-individual effects drive gut microbiota variation in common laboratory mice. Genome Biol, 14(1):R4. https://doi.org/10.1186/gb-2013-14-1-r4
Hooper LV, Midtvedt T, Gordon JI, 2002. How host-microbial interactions shape the nutrient environment of the mammalian intestine. Annu Rev Nutr, 22:283–307. https://doi.org/10.1146/annurev.nutr.22.011602.092259
Hooper LV, Stappenbeck TS, Hong CV, et al., 2003. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nat Immunol, 4(3):269–273. https://doi.org/10.1038/ni888
Kantas D, Papatsiros VG, Tassis PD, et al., 2015. The effect of a natural feed additive (Macleaya cordata), containing sanguinarine, on the performance and health status of weaning pigs. Anim Sci J, 86(1):92–98. https://doi.org/10.1111/asj.12240
Khadem A, Soler L, Everaert N, et al., 2014. Growth promotion in broilers by both oxytetracycline and Macleaya cordata extract is based on their anti-inflammatory properties. Brit J Nutr, 112(7):1110–1118. https://doi.org/10.1017/S0007114514001871
Kilroy CR, Hall WF, Bane DP, et al., 1990. Chlortetracycline in swine-bioavailability and pharmacokinetics in fasted and fed pigs. J Vet Pharmacol Ther, 13(1):49–58. https://doi.org/10.1111/j.1365-2885.1990.tb00747.x
Kim HB, Borewicz K, White BA, et al., 2012. Microbial shifts in the swine distal gut in response to the treatment with antimicrobial growth promoter, tylosin. Proc Natl Acad Sci USA, 109(38):15485–15490. https://doi.org/10.1073/pnas.1205147109
Kosina P, Gregorova J, Gruz J, et al., 2010. Phytochemical and antimicrobial characterization of Macleaya cordata herb. Fitoterapia, 81(8):1006–1012. https://doi.org/10.1016/j.fitote.2010.06.020
Looft T, Allen HK, Casey TA, et al., 2014. Carbadox has both temporary and lasting effects on the swine gut microbiota. Front Microbiol, 5:276. https://doi.org/10.3389/fmicb.2014.00276
Mazmanian SK, Kasper DL, 2006. The love-hate relationship between bacterial polysaccharides and the host immune system. Nat Rev Immunol, 6(11):849–858. https://doi.org/10.1038/nri1956
Mestecky J, McGhee JR, 1987. Immunoglobulin a (IgA): molecular and cellular interactions involved in IgA biosynthesis and immune response. Adv Immunol, 40:153–245. https://doi.org/10.1016/S0065-2776(08)60240-0
Milbradt EL, Okamoto AS, Rodrigues JCZ, et al., 2014. Use of organic acids and competitive exclusion product as an alternative to antibiotic as a growth promoter in the raising of commercial turkeys. Poultry Sci, 93(7):1855–1861. https://doi.org/10.3382/ps.2013-03593
Motamedi H, Darabpour E, Gholipour M, et al., 2010. In vitro assay for the anti-brucella activity of medicinal plants against tetracycline-resistant Brucella melitensis. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 11(7):506–511. https://doi.org/10.1631/jzus.B0900365
Nelson KL, Brözel VS, Gibson SA, et al., 2011. Influence of manure from pigs fed chlortetracycline as growth promotant on soil microbial community structure. World J Microb Biot, 27(3):659–668. https://doi.org/10.1007/s11274-010-0504-6
Newman SE, Roll MJ, Harkrader RJ, 1999. A naturally occurring compound for controlling powdery mildew of greenhouse roses. Hortscience, 34(4):686–689.
Nie YF, Hu J, Yan XH, 2015. Cross-talk between bile acids and intestinal microbiota in host metabolism and health. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 16(6):436–446. https://doi.org/10.1631/jzus.B1400327
Ouwehand AC, Salminen S, Isolauri E, 2002. Probiotics: an overview of beneficial effects. Antonie van Leeuwenhoek, 82(1-4):279–289. https://doi.org/10.1023/A:1020620607611
Pang JX, Ma RQ, Liu LM, et al., 2005. Total alkaloid of Macleaya cordata: in vitro cytotoxic effect on Hep3B cells and in vivo antitumor effect in mice. J First Mil Med Univ, 25(3):325–328 (in Chinese). https://doi.org/10.3321/j.issn:1673-4254.2005.03.018
Pryde SE, Duncan SH, Hold GL, et al., 2002. The microbiology of butyrate formation in the human colon. FEMS Microbiol Lett, 217(2):133–139. https://doi.org/10.1111/j.1574-6968.2002.tb11467.x
Psotova J, Vecera R, Zdarilova A, et al., 2006. Safety assessment of sanguiritrin, alkaloid fraction of Macleaya cordata, in rats. Vet Med, 51(4):145–155. https://doi.org/10.17221/5534-VETMED
Rhee KJ, Sethupathi P, Driks A, et al., 2004. Role of commensal bacteria in development of gut-associated lymphoid tissues and preimmune antibody repertoire. J Immunol, 172(2):1118–1124. https://doi.org/10.4049/jimmunol.172.2.1118
Russell DA, Ross RP, Fitzgerald GF, et al., 2011. Metabolic activities and probiotic potential of bifidobacteria. Int J Food Microbol, 149(1):88–105. https://doi.org/10.1016/j.ijfoodmicro.2011.06.003
Salama NA, Abou-Raya SH, Shalaby AR, et al., 2011. Incidence of tetracycline residues in chicken meat and liver retailed to consumers. Food Addit Contam B, 4(2):88–93. https://doi.org/10.1080/19393210.2011.585245
Shen J, Zhang BR, Wei GF, et al., 2006. Molecular profiling of the Clostridium leptum subgroup in human fecal microflora by PCR-denaturing gradient gel electrophoresis and clone library analysis. Appl Environ Microbol, 72(8): 5232–5238. https://doi.org/10.1128/AEM.00151-06
Shi YB, Liu L, Shao W, et al., 2015. Microcalorimetry studies of the antimicrobial actions of Aconitum alkaloids. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 16(8):690–695. https://doi.org/10.1631/jzus.B1500121
Sokol H, Pigneur B, Watterlot L, et al., 2008. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci USA, 105(43): 16731–16736. https://doi.org/10.1073/pnas.0804812105
Unno T, Kim J, Guevarra RB, et al., 2015. Effects of antibiotic growth promoter and characterization of ecological succession in swine gut microbiota. J Microbol Biotechnol, 25(4):431–438. https://doi.org/10.4014/jmb.1408.08063
Ventura M, Canchaya C, Tauch A, et al., 2007. Genomics of Actinobacteria: tracing the evolutionary history of an ancient phylura. Microbol Mol Biol Rev, 71(3):495–548. https://doi.org/10.1128/MMBR.00005-07
Walterová D, Ulrichová J, Válka I, et al., 1995. Benzo[c] phenanthridine alkaloids sanguinarine and chelerythrine: biological activities and dental care applications. Acta Univ Palacki Olomuc Fac Med, 139:7–16.
Xing HC, Li LJ, Xu KJ, et al., 2005. Intestinal microflora in rats with ischemia/reperfusion liver injury. J Zhejiang Univ SCI, 6B(1):14–21. https://doi.org/10.1631/jzus.2005.B0014
Zackular JP, Baxter NT, Iverson KD, et al., 2013. The gut microbiome modulates colon tumorigenesis. mBio, 4(6): e00692–13. https://doi.org/10.1128/mBio.00692-13
Zdarilova A, Vrublova E, Vostalova J, et al., 2008. Natural feed additive of Macleaya cordata: safety assessment in rats a 90-day feeding experiment. Food Chem Toxicol, 46(12):3721–3726. https://doi.org/10.1016/j.fct.2008.09.054
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Natural Science Foundation of Jiangsu Province for Excellent Young Scholars (No. BK20170087) and the National Natural Science Foundation of China (Nos. 31502033, 31472164, and 31672515)
Electronic supplementary materials: The online version of this article (https://doi.org/10.1631/jzus.B1700435) contains supplementary materials, which are available to authorized users
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Li, B., Zhang, Jq., Han, Xg. et al. Macleaya cordata helps improve the growth-promoting effect of chlortetracycline on broiler chickens. J. Zhejiang Univ. Sci. B 19, 776–784 (2018). https://doi.org/10.1631/jzus.B1700435
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1700435