Abstract
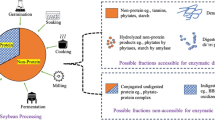
The aim of this study was to optimize the conditions for the extraction of low-abundance proteins (LAPs) and the removal of abundant proteins (APs; β-conglycinin and glycinin) from soybean meal. Single factor and orthogonal experiments were designed to determine the effects of four factors (isopropanol concentration, total extraction time, ultrasonic power, and ultrasonic time) on protein concentration in isopropanol extracts. Proteins in the isopropanol supernatant and the cold acetone precipitate of isopropanol were identified by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF-MS). The results showed that the optimal conditions were 50% isopropanol, ultrasonic pretreatment for 15 min at 350 W, and a total extraction time of 1 h. Under these conditions, the protein concentration in the isopropanol extracts reached 0.8081 g/L. Many LAPs were detected, including β-amylase, soybean agglutinin, soybean trypsin inhibitor, fumarylacetoacetase-like, phospholipase D alpha 1-like, oleosin, and even some unknown soybean proteins. The soybean APs (β-conglycinin and glycinin) were not found. The method may be useful for discovering new soybean proteins and extracting enough LAPs of soybean to allow further studies of their physiological effects on animals without the influence of APs.
摘要
目 的
优化低温脱脂大豆粕中的低丰度蛋白提取和高丰度蛋白去除条件, 为进一步探讨大豆低丰度蛋白对动物的生理功能影响提供试验材料。
创新点
结合提取时间和异丙醇浓度, 将超声波 (超声时间、 功率) 用于辅助异丙醇去除大豆高丰度蛋白和富集低丰度蛋白的研究。
方 法
在单因素试验基础上, 设计了超声时间、 超声功率、 提取时间和异丙醇浓度等四因素三水平的正交试验。 并通过测定提取液中的蛋白浓度和通过聚丙烯酰胺凝胶电泳 (SDS-PAGE) 、 质谱分析 (MS) 等蛋白质组学手段鉴定提取液或丙酮沉淀物中的蛋白质含量, 实现了大豆高丰度蛋白去除和低丰度蛋白提取条件的优化。
结 论
50% 异丙醇, 350 W 超声 15 min, 提取1 h 是大豆低丰度蛋白提取的最佳条件。 提取物含多种低丰度蛋白 (油质蛋白、 大豆未知蛋白、 磷酸酯D和无特征大豆蛋白) , 且无大豆高丰度蛋白 (大豆球蛋白和β-伴大豆球蛋白) 。
Similar content being viewed by others
References
Adams, M.R., Anthony, M.S., Chen, H., et al., 2008. Replacement of dietary soy protein isolate with concentrates of soy 7S or 11S globulin has minimal or no effects on plasma lipoprotein profiles and biomarkers of coronary risk in monkeys. Atherosclerosis, 196(1): 76–80. http://dx.doi.org/10.1016/j.atherosclerosis.2007.01.037
Bajpai, S., Sharma, A., Gupta, M.N., 2005. Removal and recovery of antinutritional factors from soybean flour. Food Chem., 89(4): 497–501. http://dx.doi.org/10.1016/j.foodchem.2004.02.055
Bu, G., Liu, H., Chen, F., et al., 2012. Effects of different factors on the forward extraction of soy protein in reverse micelle systems. Afr. J. Biotechnol., 11(28): 7247–7257.
Cornish, S.M., Wood, C.M., L'Abbe, M.R., et al., 2011. Sexand age-specific immunomodulatory effects of dietary soya protein isolate and isoflavones in rats. Brit. J. Nutr., 106(5): 683–687. http://dx.doi.org/10.1017/S0007114511000766
Deak, N.A., Murphy, P.A., Johnson L.A., et al., 2006. Fractionating soybean storage proteins using Ca2+ and NaHSO3. J. Food Sci., 71(7): C413–C424. http://dx.doi.org/10.1111/j.1750-3841.2006.00132.x
Fasina, Y.O., Classen, H.L., Garlich, J.D., et al., 2006. Response of turkey pouty to soybean lectin levels typically encountered in commercial diets. 2. Effect on intestinal development and lymphoid organs. Poult. Sci., 85: 870–877. http://dx.doi.org/10.1093/ps/85.5.870
Fulton, D.C., Stettler, M., Mettler, T., et al., 2008. β-AMYLASE4, a noncatalytic protein required for starch breakdown, acts upstream of three active β-amylases in Arabidopsis chloroplasts. Plant Cell, 20(4): 1040–1058. http://dx.doi.org/10.1105/tpc.107.056507
Hart, S.D., Bharadwaj, A.S., Brown, P.B., 2010. Soybean lectins and trypsin inhibitors, but not oligosaccharides or the interactions of factors, impact weight gain of rainbow trout (Oncorhynchus mykiss). Aquaculture, 306(1–4): 310–314. http://dx.doi.org/10.1016/j.aquaculture.2010.03.027
Jankowski, J., Juskiewicz, J., Gulewicz, K., et al., 2009. The effect of diets containing soybean meal, soybean protein concentrate, and soybean protein isolate of different oligosaccharide content on growth performance and gut function of young turkeys. Poult. Sci., 88(10): 2132–2140. http://dx.doi.org/10.3382/ps.2009-00066
Karki, B., Lamsal, B.P., Jung, S., et al., 2009a. Enhancing protein and sugar release from defatted soy flakes using ultrasound technology. J. Food Eng., 96(2): 270–278. http://dx.doi.org/10.1016/j.jfoodeng.2009.07.023
Karki, B., Lamsal, B.P., Grewell, D., et al., 2009b. Functional properties of soy protein isolates produced from ultrasonicated defatted soy flakes. J. Am. Oil Chem. Soc., 86(10): 1021–1028. http://dx.doi.org/10.1007/s11746-009-1433-0
Krishnan, B.B., 2004. A simple and rapid method to isolate low molecular weight proteinase inhibitors from soybean. Korean J. Crop Sci., 49: 342–348.
Krishnan, H.B., Kim, W.S., Jang, S., et al., 2009a. All three subunits of soybean β-conglycinin are potential food allergens. J. Agric. Food Chem., 57(3): 938–943. http://dx.doi.org/10.1021/jf802451g
Krishnan, H.B., Oehrle, N.W., Natarajan, S.S., 2009b. A rapid and simple procedure for the depletion of abundant storage proteins from legume seeds to advance proteome analysis: a case study using Glycine max. Proteomics, 9(11): 3174–3188. http://dx.doi.org/10.1002/pmic.200800875
Li, H.Z., Pordesimo, L., Weiss, J., 2004. High intensity ultrasound-assisted extraction of oil from soybeans. J. Food Res. Intern., 37(7): 731–738. http://dx.doi.org/10.1016/j.foodres.2004.02.016
Liu, M.M., Zhao, G.Q., Qi, B., et al., 2016. Effects of ultrasonic treatment on removal of abundant proteins and enrichment of low-abundance proteins in defatted soybean meal by isopropanol. Biotechnol. Eq., 30(3): 521–528. http://dx.doi.org/10.1080/13102818.2016.1149518
Natarajan, S.S., Xu, C.P., Bae, H., et al., 2006a. Characterization of storage proteins in wild (Glycine soja) and cultivated (Glycine max) soybean seeds using proteomics analysis. J. Agric. Food Chem., 54(8): 3114–3120. http://dx.doi.org/10.1021/jf052954k
Natarajan, S.S., Xu, C.P., Bae, H., et al., 2006b. Proteomic analysis of allergen and antinutritional proteins in wild and cultivated soybean seeds. J. Plant Biochem. Biotechnol., 15(2): 103–108. http://dx.doi.org/10.1007/BF03321912
Natarajan, S.S., Krishnan, H.B., Sukla, L., 2009. An efficient extraction method to enhance analysis of low abundant proteins from soybean seed. Anal. Biochem., 394(2): 259–268. http://dx.doi.org/10.1016/j.ab.2009.07.048
Rackis, J.J., Wolf, W.J., Baker, E.C., 1986. Protease Inhibitors in Plant Foods: Content and Inactivation. Nutritional and Toxicological Significance of Enzyme Inhibitors in Foods. Plenum Press, New York, p.299–349.
Saki, A.A., Abbasinezhad, M., Ghazi, S., et al., 2012. Intestinal characteristics, alkaline phosphatase and broilers performance in response to extracted and mechanical soybean meal replaced by fish meal. Agric. Sci. Technol., 14(1): 105–114.
Sun, P., Li, D.F., Li, Z.J., 2008. Effects of glycinin on IgEmediated increase of mast cell numbers and histamine release in the small intestine. J. Nutr. Biochem., 19(9): 627–633. http://dx.doi.org/10.1016/j.jnutbio.2007.08.007
Tang, R., Nancollas, G.H., 2002. New mechanism for the dissolution of sparingly soluble minerals. Pure Appl. Chem., 74(10): 1851–1857. http://dx.doi.org/10.1351/pac200274101851
Tibaldi, E., Hakim, Y., Uni, Z., et al., 2006. Effects of the partial substitution of dietary fish meal by differently processed soybean meals on growth performance, nutrient digestibility and activity of intestinal brush border enzymes in the European sea bass (Dicentrarchus labrax). Aquaculture, 261(1): 182–193. http://dx.doi.org/10.1016/j.aquaculture.2006.06.026
Wang, Y., Kong, L.J., Li, C., et al., 2006. Effect of replacing fish meal with soybean meal on growth, feed utilization and carcass composition of cuneate drum (Nibea miichthioides). Aquaculture, 261(4): 1307–1313. http://dx.doi.org/10.1016/j.aquaculture.2006.08.045
Zang, J.J., Li, D.F., Piao, X.S., 2006. Effects of soybean agglutinin on body composition and organ weight in rats. Arch. Anim. Nutr., 60(3): 245–253. http://dx.doi.org/10.1080/17450390600679082
Acknowledgements
We are very grateful to the Testing Center of Yangzhou University for providing the 5800 MALDI-TOF/TOF analyzer and Yu-yang WANG (Testing Center of Yangzhou University) for the guidance in its operation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the China Agriculture Research System (No. CARS-36) and the National Natural Science Foundation of China (No. 31572430)
Rights and permissions
About this article
Cite this article
Liu, Mm., Qi, B., Liu, Zx. et al. Optimization of low-abundance protein extraction and abundant protein removal from defatted soybean meal. J. Zhejiang Univ. Sci. B 18, 878–885 (2017). https://doi.org/10.1631/jzus.B1600293
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600293