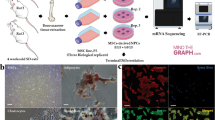
Abstract
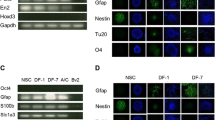
Bone mesenchymal stem cells (BMSCs) differentiated into neurons have been widely proposed for use in cell therapy of many neurological disorders. It is therefore important to understand the molecular mechanisms underlying this differentiation. We screened differentially expressed genes between immature neural tissues and untreated BMSCs to identify the genes responsible for neuronal differentiation from BMSCs. GSE68243 gene microarray data of rat BMSCs and GSE18860 gene microarray data of rat neurons were received from the Gene Expression Omnibus database. Transcriptome Analysis Console software showed that 1248 genes were up-regulated and 1273 were down-regulated in neurons compared with BMSCs. Gene Ontology functional enrichment, protein-protein interaction networks, functional modules, and hub genes were analyzed using DAVID, STRING 10, BiNGO tool, and Network Analyzer software, revealing that nine hub genes, Nrcam, Sema3a, Mapk8, Dlg4, Slit1, Creb1, Ntrk2, Cntn2, and Pax6, may play a pivotal role in neuronal differentiation from BMSCs. Seven genes, Dcx, Nrcam, Sema3a, Cntn2, Slit1, Ephb1, and Pax6, were shown to be hub nodes within the neuronal development network, while six genes, Fgf2, Tgfβ1, Vegfa, Serpine1, Il6, and Stat1, appeared to play an important role in suppressing neuronal differentiation. However, additional studies are required to confirm these results.
摘要
目的
通过筛选差异基因,获得控制骨髓间充质干细胞 向神经细胞分化及神经发育的中心基因,为治疗 神经系统疾病提供参考。
方法
从基因表达综合数据库(Gene Expression Omnibus database)中获得芯片数据,利用生物信息学软件 筛选差异基因,并对差异基因进行GO 功能富集、 蛋白互作网络分析和中心基因分析。
结论
通过分析,初步推测Nrcam、Sema3a、Mapk8、 Dlg4、Slit1、Creb1、Ntrk2、Cntn2 和Pax6 等中 心基因在调控骨髓间充质干细胞向神经细胞的 分化中发挥重要作用;Dcx、Nrcam、Sema3a、 Cntn2、Slit1、Ephb1 和Pax6 等中心基因在神经 发育过程中发挥作用;Fgf2、Tgfβ1、Vegfa、 Serpine1、Il6 和Stat1 等中心基因在抑制神经分化 过程中发挥作用。
Similar content being viewed by others
References
Ahn, J., Lee, J., Kim, S., 2015. Interferon-gamma inhibits the neuronal differentiation of neural progenitor cells by inhibiting the expression of Neurogenin2 via the JAK/ STAT1 pathway. Biochem. Biophys. Res. Commun., 466(1):52–59. http://dx.doi.org/10.1016/j.bbrc.2015.08.104
Assenov, Y., Ramirez, F., Schelhorn, S.E., et al., 2008. Computing topological parameters of biological networks. Bioinformatics, 24(2):282–284. http://dx.doi.org/10.1093/bioinformatics/btm554
Bae, K.S., Park, J.B., Kim, H.S., et al., 2011. Neuron-like differentiation of bone marrow-derived mesenchymal stem cells. Yonsei Med. J., 52(3):401–412. http://dx.doi.org/10.3349/ymj.2011.52.3.401
Balasubramaniam, B., Carter, D.A., Mayer, E.J., et al., 2009. Microglia derived IL-6 suppresses neurosphere generation from adult human retinal cell suspensions. Exp. Eye Res., 89(5):757–766. http://dx.doi.org/10.1016/j.exer.2009.06.019
Belvindrah, R., Nissant, A., Lledo, P.M., 2011. Abnormal neuronal migration changes the fate of developing neurons in the postnatal olfactory bulb. J. Neurosci., 31(20): 7551–7562. http://dx.doi.org/10.1523/JNEUROSCI.6716-10.2011
Bertani, N., Malatesta, P., Volpi, G., et al., 2005. Neurogenic potential of human mesenchymal stem cells revisited: analysis by immunostaining, time-lapse video and microarray. J. Cell Sci., 118(17):3925–3936. http://dx.doi.org/10.1242/jcs.02511
Brown, J.P., Couillard-Despres, S., Cooper-Kuhn, C.M., et al., 2003. Transient expression of doublecortin during adult neurogenesis. J. Comp. Neurol., 467(1):1–10. http://dx.doi.org/10.1002/cne.10874
Çapkin, M., Cakmak, S., Kurt, F.O., et al., 2012. Random/ aligned electrospun PCL/PCL-collagen nanofibrous membranes: comparison of neural differentiation of rat AdMSCs and BMSCs. Biomed. Mater., 7(4):045013. http://dx.doi.org/10.1088/1748-6041/7/4/045013
Ceber, M., Mihmanli, A., Kilic, U., et al., 2015. Changes in expression of Slit1 and its receptor Robo2 in trigeminal ganglion and inferior alveolar nerve following inferior alveolar nerve axotomy in adult rats: a pilot study. Int. J. Oral. Maxillofac. Surg., 44(4):518–527. http://dx.doi.org/10.1016/j.ijom.2014.09.022
Demyanenko, G.P., Mohan, V., Zhang, X., et al., 2014. Neural cell adhesion molecule NrCAM regulates Semaphorin 3F-induced dendritic spine remodeling. J. Neurosci., 34(34):11274–11287. http://dx.doi.org/10.1523/JNEUROSCI.1774-14.2014
Edamura, K., Nakano, R., Fujimoto, K., et al., 2014. Effects of cryopreservation on the cell viability, proliferative capacity and neuronal differentiation potential of canine bone marrow stromal cells. J. Vet. Med. Sci., 76(4): 573–577. http://dx.doi.org/10.1292/jvms.13-0296
Francis, F., Koulakoff, A., Boucher, D., et al., 1999. Doublecortin is a developmentally regulated, microtubuleassociated protein expressed in migrating and differentiating neurons. Neuron, 23(2):247–256. http://dx.doi.org/10.1016/S0896-6273(00)80777-1
Hatori, M., Shimozawa, N., Yasmin, L., et al., 2014. Role of retinoic acid and fibroblast growth factor 2 in neural differentiation from cynomolgus monkey (Macaca fascicularis) embryonic stem cells. Comp. Med., 64(2):140–147.
Irizarry, R.A., Hobbs, B., Collin, F., et al., 2003. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics, 4(2):249–264. http://dx.doi.org/10.1093/biostatistics/4.2.249
Jiang, X., Xu, R., Guo, Z., et al., 2005. Neurobiochemistry and neuroelectrophysiology of neuron-like cells differentiated from neural BMSCs-D-NSCs. 2005 IEEE Engineering in Medicine and Biology 27th Annual Conference, Shanghai, p.5215–5218. http://dx.doi.org/10.1109/IEMBS.2005.1615654
Liu, W.T., Han, Y., Li, H.C., et al., 2009. An in vivo mouse model of long-term potentiation at synapses between primary afferent C-fibers and spinal dorsal horn neurons: essential role of EphB1 receptor. Mol. Pain, 5:29. http://dx.doi.org/10.1186/1744-8069-5-29
Lustig, M., Sakurai, T., Grumet, M., 1999. Nr-CAM promotes neurite outgrowth from peripheral ganglia by a mechanism involving axonin-1 as a neuronal receptor. Dev. Biol., 209(2):340–351. http://dx.doi.org/10.1006/dbio.1999.9250
McGowan, L.D., Alaama, R.A., Striedter, G.F., 2013. FGF2 delays tectal neurogenesis, increases tectal cell numbers, and alters tectal lamination in embryonic chicks. PLoS ONE, 8(11):e79949. http://dx.doi.org/10.1371/journal.pone.0079949
Mishra, S., Maurya, S.K., Srivastava, K., et al., 2015. Pax6 influences expression patterns of genes involved in neurodegeneration. Ann. Neurosci., 22(4):226–231. http://dx.doi.org/10.5214/ans.0972.7531.220407
Mlechkovich, G., Peng, S.S., Shacham, V., et al., 2014. Distinct cytoplasmic domains in Plexin-A4 mediate diverse responses to semaphorin 3A in developing mammalian neurons. Sci. Signal., 7(316):ra24. http://dx.doi.org/10.1126/scisignal.2004734
Nair, J., Ghatge, M., Kakkar, V.V., et al., 2014. Network analysis of inflammatory genes and their transcriptional regulators in coronary artery disease. PLoS ONE, 9(4): e94328. http://dx.doi.org/10.1371/journal.pone.0094328
Nandy, S.B., Mohanty, S., Singh, M., et al., 2014. Fibroblast Growth Factor-2 alone as an efficient inducer for differentiation of human bone marrow mesenchymal stem cells into dopaminergic neurons. J. Biomed. Sci., 21(1):83. http://dx.doi.org/10.1186/s12929-014-0083-1
Nelson, A.D., Svendsen, C.N., 2006. Low concentrations of extracellular FGF-2 are sufficient but not essential for neurogenesis from human neural progenitor cells. Mol. Cell. Neurosci., 33(1):29–35. http://dx.doi.org/10.1016/j.mcn.2006.06.003
Parivar, K., Baharara, J., Sheikholeslami, A., 2015. Neural differentiation of mouse bone marrow-derived mesenchymal stem cells treated with sex steroid hormones and basic fibroblast growth factor. Cell J., 17(1):27–36.
Pereira, L., Medina, R., Baena, M., et al., 2015. IFN gamma regulates proliferation and neuronal differentiation by STAT1 in adult SVZ niche. Front. Cell. Neurosci., 9:270. http://dx.doi.org/10.3389/fncel.2015.00270
Ratie, L., Ware, M., Jagline, H., et al., 2014. Dynamic expression of Notch-dependent neurogenic markers in the chick embryonic nervous system. Front. Neuroanat., 8:158. http://dx.doi.org/10.3389/fnana.2014.00158
Richards, A.B., Scheel, T.A., Wang, K., et al., 2007. EphB1 null mice exhibit neuronal loss in substantia nigra pars reticulata and spontaneous locomotor hyperactivity. Eur. J. Neurosci., 25(9):2619–2628. http://dx.doi.org/10.1111/j.1460-9568.2007.05523.x
Round, J.E., Sun, H., 2011. The adaptor protein Nck2 mediates Slit1-induced changes in cortical neuron morphology. Mol. Cell. Neurosci., 47(4):265–273. http://dx.doi.org/10.1016/j.mcn.2011.04.009
Sakurai, T., 2012. The role of NrCAM in neural development and disorders—beyond a simple glue in the brain. Mol. Cell. Neurosci., 49(3):351–363. http://dx.doi.org/10.1016/j.mcn.2011.12.002
Shannon, P., Markiel, A., Ozier, O., et al., 2003. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res., 13(11): 2498–2504. http://dx.doi.org/10.1101/gr.1239303
Sherman, B.T., Huangda, W., Tan, Q., et al., 2007. DAVID Knowledgebase: a gene-centered database integrating heterogeneous gene annotation resources to facilitate high-throughput gene functional analysis. BMC Bioinformatics, 8(1):8426. http://dx.doi.org/10.1186/1471-2105-8-426
Snel, B., Lehmann, G., Bork, P., et al., 2000. STRING: a web-server to retrieve and display the repeatedly occurring neighbourhood of a gene. Nucleic Acids Res., 28(18): 3442–3444. http://dx.doi.org/10.1093/nar/28.18.3442
Sun, T., Li, W., Ling, S., 2016. miR-30c and semaphorin 3A determine adult neurogenesis by regulating proliferation and differentiation of stem cells in the subventricular zones of mouse. Cell Prolif., 49(3):270–280. http://dx.doi.org/10.1111/cpr.12261
Torres, L., Juarez, U., Garcia, L., et al., 2015. External ear microRNA expression profiles during mouse development. Int. J. Dev. Biol., 59(10-12):497–503. http://dx.doi.org/10.1387/ijdb.150124sf
Wu, C.Y., Persaud, S.D., Wei, L.N., 2016. Retinoic acid induces ubiquitination-resistant RIP140/LSD1 complex to fine-tune Pax6 gene in neuronal differentiation. Stem Cells, 34(1):114–123. http://dx.doi.org/10.1002/stem.2190
Wu, K.Y., He, M., Hou, Q.Q., et al., 2014. Semaphorin 3A activates the guanosine triphosphatase Rab5 to promote growth cone collapse and organize callosal axon projections. Sci. Signal., 7(340):ra81. http://dx.doi.org/10.1126/scisignal.2005334
Yang, J., Yang, C., Liu, C., et al., 2016. Paradoxical effects of VEGF on synaptic activity partially involved in notch1 signaling in the mouse hippocampus. Hippocampus, 26(5):589–600. http://dx.doi.org/10.1002/hipo.22544
Zhang, X., Zheng, H., Zhu, H.Y., et al., 2016. Acute effects of transforming growth factor-ß1 on neuronal excitability and involvement in the pain of rats with chronic pancreatitis. J. Neurogastroenterol. Motil., 22(2):333–343. http://dx.doi.org/10.5056/jnm15127
Zhou, X., Zoller, T., Krieglstein, K., et al., 2015. TGFß1 inhibits IFN?-mediated microglia activation and protects mDA neurons from IFNγ-driven neurotoxicity. J. Neurochem., 134(1):125–134. http://dx.doi.org/10.1111/jnc.13111
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Key Project of Hebei North University (No. 120177) and the Science and Technology Research Project of Hebei Province Department Institutions of Higher Learning (No. Z2015047), China
Electronic supplementary materials: The online version of this article (http://dx.doi.org/10.1631/jzus.B1600109) contains supplementary materials, which are available to authorized users
ORCID: Li-ning SU, http://orcid.org/0000-0001-8118-7601
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Su, Ln., Song, Xq., Wei, Hp. et al. Identification of neuron-related genes for cell therapy of neurological disorders by network analysis. J. Zhejiang Univ. Sci. B 18, 172–182 (2017). https://doi.org/10.1631/jzus.B1600109
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600109
Key words
- Neuronal differentiation
- Bone mesenchymal stem cells (BMSCs)
- Protein-protein interaction network
- Differentially expressed genes