Abstract
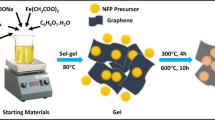
The graphene/mesocarbon microbead (MCMB) composite is assessed as an anode material with a high capacity for lithium-ion batteries. The composite electrode exhibits improved cycling stability and rate capability, delivering a high initial charge/discharge capacity of 421.4 mA·h/g/494.8 mA·h/g as well as an excellent capacity retention over 500 cycles at a current density of 40 mA/g. At a higher current density of 800 mA/g, the electrode still retains 35% of its initial capacity which exceeds the capacity retention of pure graphene or MCMB reference electrodes. Cyclic voltammetry and electrochemical impedance spectroscopy reveal that the composite electrode favors electrochemical kinetics as compared with graphene and MCMB separately. Superior electrochemical properties suggest a strong synergetic effect between highly conductive graphene and MCMB.
目的
电动汽车和大规模储能的发展对锂离子电池的能 量密度提出了更高的要求,但现有商业石墨负极 容量难以满足要求。本文结合石墨烯高电导和高 容量的优点以及中间相碳微球材料循环稳定性 优良的优势,研究和报道一种容量高和循环性能 好的石墨烯/中间相碳微球复合负极材料。
方法:1. 通过选择高电导率石墨烯和中间相碳微球,制 备石墨烯和中间相碳微球复合负极材料。2. 选用 商业聚偏氟乙烯(PVDF)粘结剂,制备复合材 料电极极片,测试和表征电极的形貌、电导以及 半电池的充放电等电化学性能,并优化复合材料 质量比。3. 选择优化的复合负极材料(GMC (8:2)),研究其长循环性能。
结论:中间相碳微球的球形结构能有效防止石墨烯的折 叠团聚,从而发挥石墨烯的高电导性能。因此, 石墨烯/中间相碳微球复合负极材料表现出了 很好的倍率性能和循环性能,且其容量达到了 421 mA∙h/g 以上,高于商业石墨的理论容量,具 有潜在的应用前景。
Similar content being viewed by others
References
Alcántara R, Fernández Madrigal FJ, Lavela P, et al., 2000. Characterisation of mesocarbon microbeads (MCMB) as active electrode material in lithium and sodium cells. Carbon, 38(7):1031–1041. https://doi.org/10.1016/S0008-6223(99)00215-8
Allen MJ, Tung VC, Kaner RB, 2010. Honeycomb carbon: a review of graphene. Chemical Reviews, 110(1):132–145. https://doi.org/10.1021/cr900070d
Birrozzi A, Maroni F, Raccichini R, et al., 2015. Enhanced stability of SnSb/graphene anode through alternative binder and electrolyte additive for lithiumion batteries application. Journal of Power Sources, 294:248–253. https://doi.org/10.1016/j.jpowsour.2015.06.065
Cai MZ, Thorpe D, Adamson DH, et al., 2012. Methods of graphite exfoliation. Journal of Materials Chemistry, 22(48):24992–25002. https://doi.org/10.1039/c2jm34517j
Cai XY, Lai LF, Shen ZX, et al., 2017. Graphene and graphene-based composites as Li-ion battery electrode materials and their application in full cells. Journal of Materials Chemistry A, 5(30):15423–15446. https://doi.org/10.1039/C7TA04354F
Collins J, Gourdin G, Foster M, et al., 2015. Carbon surface functionalities and SEI formation during Li intercalation. Carbon, 92:193–244. https://doi.org/10.1016/j.carbon.2015.04.007
Compton OC, Nguyen ST, 2010. Graphene oxide, highly reduced graphene oxide, and graphene: versatile building blocks for carbon-based materials. Small, 6(6):711–723. https://doi.org/10.1002/smll.200901934
Dahn JR, Zheng T, Liu YH, et al., 1995. Mechanisms for lithium insertion in carbonaceous materials. Science, 270(5236):590–593. https://doi.org/10.1126/science.270.5236.590
Du YH, Tang YF, Huang FQ, et al., 2016. Preparation of three-dimensional free-standing nano-LiFePO4/graphene composite for high performance lithiumion battery. RSC Advances, 6(57):52279–52283. https://doi.org/10.1039/c6ra08937b
Dunn B, Kamath H, Tarascon JM, 2011. Electrical energy storage for the grid: a battery of choices. Science, 334(6058):928–935. https://doi.org/10.1126/science.1212741
Guo P, Song HH, Chen XH, 2009. Electrochemical performance of graphene nanosheets as anode material for lithium-ion batteries. Electrochemistry Communications, 11(6):1320–1324. https://doi.org/10.1016/j.elecom.2009.04.036
Hu J, Li H, Huang XJ, 2007. Electrochemical behavior and microstructure variation of hard carbon nano-spherules as anode material for Li-ion batteries. Solid State Ionics, 178(3–4):265–271. https://doi.org/10.1016/j.ssi.2006.12.014
Huang X, Zeng ZY, Fan ZX, et al., 2012. Graphene-based electrodes. Advanced Materials, 24(45):5979–6004. https://doi.org/10.1002/adma.201201587
Imtiaz M, Zhu CL, Li Y, et al., 2017. Functionalized bioinspired porous carbon with graphene sheets as anode materials for lithium-ion batteries. Journal of Alloys and Compounds, 724:296–305. https://doi.org/10.1016/j.jallcom.2017.07.005
Jaguemont J, Boulon L, Dubé Y, 2016. A comprehensive review of lithium-ion batteries used in hybrid and electric vehicles at cold temperatures. Applied Energy, 164:99–114. https://doi.org/10.1016/j.apenergy.2015.11.034
Li N, Chen ZP, Ren WC, et al., 2012. Flexible graphene-based lithiumion batteries with ultrafast charge and discharge rates. Proceedings of the National Academy of Sciences of the United States of America, 109(43):17360–17365. https://doi.org/10.1073/pnas.1210072109
Li Z, Li GY, Ouyang J, et al., 2019. Defective lithium storage boosts high rate and long-life span of carbon fibers. ChemistrySelect, 4(19):5768–5775. https://doi.org/10.1002/slct.201901140
Linden D, Reddy TB, 2002. Handbook of Batteries, 3rd Edition. McGraw-Hill, New York, USA.
Liu HL, Tang YF, Zhao W, et al., 2018. Facile synthesis of nitrogen and halogen dual-doped porous graphene as an advanced performance anode for lithium-ion batteries. Advanced Materials Interfaces, 5(5):1701261. https://doi.org/10.1002/admi.201701261
Mochida I, Korai Y, Ku CH, et al., 2000. Chemistry of synthesis, structure, preparation and application of aromaticderived mesophase pitch. Carbon, 38(2):305–328. https://doi.org/10.1016/s0008-6223(99)00176-1
Novoselov KS, Geim AK, Morozov SV, et al., 2004. Electric field effect in atomically thin carbon films. Science, 306(5696):666–669. https://doi.org/10.1126/science.1102896
Ogumi Z, Inaba M, 1998. Electrochemical lithium intercalation within carbonaceous materials: intercalation processes, surface film formation, and lithium diffusion. Bulletin of the Chemical Society of Japan, 71(3):521–534. https://doi.org/10.1246/bcsj.71.521
Peled E, Eshkenazi V, Rosenberg Y, 1998. Study of lithium insertion in hard carbon made from cotton wool. Journal of Power Sources, 76(2):153–158. https://doi.org/10.1016/S0378-7753(98)00148-7
Qie L, Chen WM, Wang ZH, et al., 2012. Nitrogen-doped porous carbon nanofiber webs as anodes for lithium ion batteries with a superhigh capacity and rate capability. Advanced Materials, 24(15):2047–2050. https://doi.org/10.1002/adma.201104634
Wang BW, Li Z, Zhang JJ, et al., 2019. N-doped 3D interconnected carbon bubbles as anode materials for lithiumion and sodium-ion storage with excellent performance. Journal of Nanoscience and Nanotechnology, 19(11): 7301-7307. https://doi.org/10.1166/jnn.2019.16655
Wang GX, Shen XP, Yao J, et al., 2009. Graphene nanosheets for enhanced lithium storage in lithium ion batteries. Carbon, 47(8):2049–2053. https://doi.org/10.1016/j.carbon.2009.03.053
Wang H, Abe T, Maruyama S, et al., 2005. Graphitized carbon nanobeads with an onion texture as a lithium-ion battery negative electrode for high-rate use. Advanced Materials, 17(23):2857–2860. https://doi.org/10.1002/adma.200500320
Wang JG, Jin DD, Zhou R, et al., 2016. Highly flexible graphene/Mn3O4 nanocomposite membrane as advanced anodes for Li-ion batteries. ACS Nano, 10(6):6227–6234. https://doi.org/10.1021/acsnano.6b02319
Wang W, Guo SR, Ozkan M, et al., 2013. Chrysanthemum like carbon nanofiber foam architectures for supercapacitors. Journal of Materials Research, 28(7):912–917. https://doi.org/10.1557/jmr.2012.412
Wang ZX, Huang XJ, Xue RJ, et al., 1999. A new possible mechanism of lithium insertion and extraction in lowtemperature pyrolytic carbon electrode. Carbon, 37(4): 685-692. https://doi.org/10.1016/S0008-6223(98)00245-0
Welna DT, Qu LT, Taylor BE, et al., 2011. Vertically aligned carbon nanotube electrodes for lithium-ion batteries. Journal of Power Sources, 196(3):1455–1460. https://doi.org/10.1016/j.jpowsour.2010.08.003
Wu YP, Zhang TF, Zhang F, et al., 2012. In situ synthesis of graphene/single-walled carbon nanotube hybrid material by arc-discharge and its application in supercapacitors. Nano Energy, 1(6):820–827. https://doi.org/10.1016/j.nanoen.2012.07.001
Wu ZS, Ren WC, Xu L, et al., 2011. Doped graphene sheets as anode materials with superhigh rate and large capacity for lithiumion batteries. ACS Nano, 5(7):5463–5471. https://doi.org/10.1021/nn2006249
Yao J, Wang GX, Ahn JH, et al., 2003. Electrochemical studies of graphitized mesocarbon microbeads as an anode in lithium-ion cells. Journal of Power Sources, 114(2):292–297. https://doi.org/10.1016/S0378-7753(02)00585-2
Yoo E, Kim J, Hosono E, et al., 2008. Large reversible Li storage of graphene nanosheet families for use in rechargeable lithiumion batteries. Nano Letters, 8(8):2277–2282. https://doi.org/10.1021/nl800957b
Yun YS, Le VD, Kim H, et al., 2014. Effects of sulfur doping on graphene-based nanosheets for use as anode materials in lithium-ion batteries. Journal of Power Sources, 262: 79-85. https://doi.org/10.1016/j.jpowsour.2014.03.084
Zhao JF, Wen XM, Xu HS, et al., 2019. Salting-out and salting-in of protein: a novel approach toward fabrication of hierarchical porous carbon for energy storage application. Journal of Alloys and Compounds, 788:397–406. https://doi.org/10.1016/j.jallcom.2019.02.252
Zhu S, Li JJ, Ma LY, et al., 2016. Three-dimensional network of N-doped carbon ultrathin nanosheets with closely packed mesopores: controllable synthesis and application in electrochemical energy storage. ACS Applied Materials & Interfaces, 8(18):11720–11728. https://doi.org/10.1021/acsami.6b02386
Zhu YW, Murali S, Cai WW, et al., 2010. Graphene and graphene oxide: synthesis, properties, and applications. Advanced Materials, 22(35):3906–3924. https://doi.org/10.1002/adma.201001068
Author information
Authors and Affiliations
Contributions
Inna SMOLIANOVA conducted research and prepared the paper; Jin-long HU and Xin-yue ZHAO helped for guidance of partial electrochemical tests; Viacheslav DEMENTIEV donated the graphene sample and initialized the collaboration of this research; Ling-zhi ZHANG is responsible for all the research and the publication of the paper including revision of manuscript and all communication during the whole process.
Corresponding author
Additional information
Conflict of interest
Inna SMOLIANOVA, Jin-long HU, Xin-yue ZHAO, Viacheslav DEMENTIEV, and Ling-zhi ZHANG declare that they have no conflict of interest.
Project supported by the National Natural Science Foundation of China (No. 21573239), the Guangdong Provincial Project for Science and Technology (Nos. 2014TX01N14, 2015B010135008, and 2016B010114003), the Guangzhou Municipal Project for Science and Technology (No. 201509010018), and the K. C. WONG Education Foundation, China
Rights and permissions
About this article
Cite this article
Smolianova, I., Hu, Jl., Zhao, Xy. et al. A high-capacity graphene/mesocarbon microbead composite anode for lithium-ion batteries. J. Zhejiang Univ. Sci. A 21, 392–400 (2020). https://doi.org/10.1631/jzus.A1900600
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.A1900600