Abstract
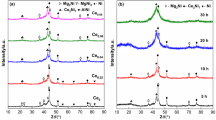
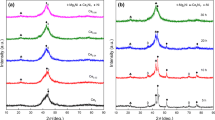
Mg2Ni0.8Cr0.2−x wt.% CoO/Al2O3 (x=0.5, 1, 2 and 3) composites were prepared by mechanically milling sintered Mg2Ni0.8Cr0.2 alloy and CoO/Al2O3 compound for 45 h. The addition of CoO/Al2O3 compound resulted in the good kinetics properties of hydriding/dehydriding reaction of the composites. The composite with 1.0 wt.% CoO/Al2O3 catalyst could reach the maximum hydrogen absorption capacity (2.9 wt.%) within 5 min at 393 K under H2 pressure of 4 MPa, and can desorb rapidly at 493 K. The decomposition and synthesis of hydrogen molecule on Mg2Ni0.8Cr0.2 alloy surface was promoted by addition of CoO/Al2O3 catalyst. In addition, the formation of metallic Ni particles, strain and defects during the ball milling process also resulted in the improved hydrogenation performance of Mg2Ni-based alloys.
Similar content being viewed by others
References
Chen, X.G., Shen, W., Xu, H.L., Xiang, Y.F., 2000. Preparation of γ-Butyrolactone from 1,4-Butanediol dehydrogenation over Cu/ZnO/Al2O3 catalyst.Chinese Journal of Catalysis,21(3):259–263 (in Chinese)
Cong, Y., Tin, K.C., Huang, N.B., Xu, C.H., Zhang, T., 2000. Preparation of ultrafine Cu-ZnO-ZrO2 catalysts and CO2 hydrogenation performance.Chinese Journal of Catalysis,21(3):247–250 (in Chinese).
Lee, D.S., Kwon, I.K., Bobet, J.L., Song, M.Y., 2004. Effects on the H2-sorption properties of Mg of Co (with various sizes) and CoO addition by reactive grinding.J. Alloys and Comps.,366:279–288.
Liu, B.J., Xiong, G.X., Pan, X.L., Sheng, S.S., Yang, W.S., 2002. Effect of noble metal on selective hydrogenation of cinnamaldehyde over cobalt-based catalyst.Chinese Journal of Catalysis,23(5):481–484 (in Chinese).
Oelerich, W., Klassen, T., Bormann, R., 2001. Metal oxides as catalysts for improved hydrogen sorption in nanocrystal-line Mg-based materials.J. Alloys Comp.,315:237–242.
Orimo, S., Fujii, H., Ikeda, K., 1997. Notable hydriding properties of a nanostructured composite material of the Mg2Ni−H system synthesized by reactive mechanical grinding.Acta Mater.,45(1):331–341.
Pedersen, A.S., Larsen, B., 1993. The storage of industrially pure hydrogen in magnesium.Int. J. Hydrogen Energy,18:297–300.
Reilly, J.J., Wiswall, R.H., 1968. The reaction of hydrogen with alloys of magnesium and nickel and the formation of Mg2NiH4.Inorg. Chem. 7:2254–2256.
Song, M.Y., Bobet, J.L., Darriet, B., 2002. Improvement in hydrogen sorption properties of Mg by reactive mechanical grinding with Cr2O3, Al2O3 and CeO2.J. Alloys Comp.,340:256–262.
Spassov, T., Köster, U., 1998. Thermal stability and hydriding properties of nanocrystalline melt-spun Mg63Ni30Y7 alloy.J. Alloys Comp.,279:279–286.
Tsushio, Y., Enoki, H., Akiba, E., 1998. Hydrogenation properties of MgNi0.86M10.03 (M1=Cr, Fe, Co, Mn) alloy.J. Alloys Comp.,281:301–305.
Wang, P., Wang, A.M., Zhang, H.F., Ding, B.Z., Hu, Z.Q., 2000. Hydrogenation characteristics of Mg−TiO2 (rutile) composite.J. Alloys Comp.,313:218–223.
Wang, X.L., Tu, J.P., Zhang, X.B., Gao, R.G., Chen, C.P., 2002. Hydrogen storage properties of nanocrystalline Mg2Ni alloys with Cr additions.The Chinese Journal of Nonferrous Metals,12(5):907–911 (in Chinese).
Zhu, H.J., Li, H.B., Tong, J.H., Sheng, S.S., Yang, W.S., Lin, L.W., 2002. Oxidative dehydrogenation of propane over vanadia catalysts supported on TiO2−ZrO2 composite oxide.Chinese Journal of Catalysis,23(5):391–392 (in Chinese).
Author information
Authors and Affiliations
Corresponding author
Additional information
Project (No. TG20000264-06) supported by the Special Funds for Major States Basic Research Project of MOST, China
Rights and permissions
About this article
Cite this article
Wang, Xl., Tu, Jp., Chen, Cp. et al. Hydrogenation properties of mechanically milled Mg2Ni0.8Cr0.2-CoO/Al2O3 composites. J Zheijang Univ Sci B 6, 208–212 (2005). https://doi.org/10.1631/jzus.2005.B0208
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.2005.B0208