Abstract
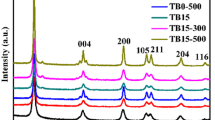
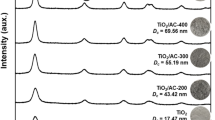
A novel metallo-organic chemical vapor deposition (MOCVD) technique has been applied to the preparation of the photocatalytic titanium dioxide supported on activated carbon. The effects of various condition parameters such as carrier gas flow rate, source temperature and deposition temperature on the deposition rate were investigated. The maximum deposition rate of 8.2 mg/(g·h) was obtained under conditions of carrier gas flow rate of 400 ml/min, source temperature of 423 K and deposition temperature of 913 K. The deposition rate followed Arrhenius behavior at temperature of 753 K to 913 K, corresponding to activation energy E a of 51.09 kJ/mol. TiO2 existed only in anatase phase when the deposition temperature was 773 K to 973 K. With increase of deposition temperature from 1073 K to 1273 K, the rutile content sharply increased from 7% to 70%. It was found that a deposition temperature of 773 K and a higher source temperature of 448 K resulted in finely dispersed TiO2 particles, which were mainly in the range of 10–20 nm.
Similar content being viewed by others
References
Choy, K.L., 2003. Chemical vapor deposition of coating. Prog. Mater. Sci., 48:57–170.
Fujishima, A., Honda, K., 1972. Electrochemical photolysis of water at a semiconductor electrode. Nature, 37(1):238–245.
Hierso, J.C., Feurer, R., Kalck, P., 2000. Platinum andpalladium films obtained by low-temperature MOCVD for the formation of small particles on divided supports as catalytic materials. Chem. Mater., 12:390–399.
Lei, L., Chu, H., Hu, X.J., Yue, P.L., 1999. Preparation of heterogeneous photocatalyst (TiO2/Alumina) by MOCVD. Ind. Eng. Chem. Res., 38:3381–3385.
Moene, R., Makkee, M., Moulijn, J.A., 1993. Chemical vapor deposition as a novel technique for catalyst preparation: modeling of active phase profiles. Chem. Eng. J., 53:13–24.
Serp, P., Feurer, R., Morancho, R., Kalck, P., 1995. A versatile one-step method for the preparation of highly dispersed metal supported catalysts. J. Mol. Catal. A: Chem., 101:107–110.
Snell, F.R.D., Ettre, L.S., 1974. Encyclopedia of Industrial Chemical Analysis. Interscience, New York, U.S.A, p. 107–109.
Sopyan, I., Watanabe, M., Murasawa, S., Hashimoto, K., Fujishima, A., 1996. An efficient TiO2 thin-film photocatalyst: photocatalytic properties in gas-phase acetaldehyde degradation. J. Photochem. Photobiol. A., 98: 79–86.
Spur, R.A., Myers, H., 1957. Quantitative analysis of anantase-rutile mixtures with an X-ray diffractometer. Anal. Chem., 29:760–762.
Tsukasa, t., Shigeyoshi, I., Susumu, K., Yoneyama, H., 1996. Effects of adsorbents used as supports for titanium dioxide loading on photocatalytic degradation of propyzamide. Environ. Sci. Technol., 30:1275–1281.
Zhang, Q., Griffin, G.L., 1995. Gas-phase kinetics for TiO2 CVD: hot-wall reactor results. Thin Solid Films, 263:65–71.
Zhang, L.Y., Sun, Y.J., Yao, X., 1998. Preparation of nano TiO2 powder by MOCVD Process TiO2. Chinese J. Silicon, 17(5):22–32.
Author information
Authors and Affiliations
Additional information
Project (No. 90206007) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Zhang, Xw., Zhou, Mh., Lei, Lc. et al. Synthesis of TiO2 supported on activated carbon by MOCVD: operation parameters study. J. Zheijang Univ.-Sci. 5, 1548–1553 (2004). https://doi.org/10.1631/jzus.2004.1548
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.2004.1548