Abstract
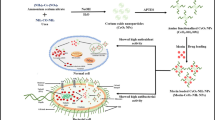
A crucial balance between oxidative eustress and distress is important for maintaining redox homeostasis in the cellular milieu. Therefore, sustaining the intracellular redox buffer condition with exogenous agents could be a therapeutic strategy against diseases caused by redox imbalance. Here, we synthesized chitosan-functionalized Mn3O4 nanoparticles (Ch-Mn3O4 NPs) and tested their redox buffering capability in in-vitro and in-cellulo. Chitosan is easily absorbed by the intestine and can be used as a target-specific delivery agent to the intestine, while Mn3O4 NPs have redox modulatory properties. Therefore, combination of chitosan and Mn3O4 NPs provide the opportunity for targeted redox buffering. Targeted delivery of a drug and remediation of the corresponding indication qualify the drug to be a theranostic agent. Our spectroscopic studies suggest ROS generation as well as antioxidant ability of Ch-Mn3O4 NPs. In-cellulo studies using A549 cell lines confirmed the efficacy of the nanohybrid in redox homeostasis. The outcomes suggest that Ch-Mn3O4 nanohybrid has the potential to function as a target-specific redox buffering agent in both in-vitro and in-cellulo systems.
Graphic abstract





Similar content being viewed by others
References
A.J. Kowaltowski, A.E. Vercesi, Free Radical Biol. Med. 26, 463 (1999)
C. Li, R.M. Jackson, Am. J. Physiol. Cell Physiol. 282, C227 (2002)
K. Brieger, S. Schiavone, F. Miller, K.-H. Krause, Swiss Med Weekly 142, w13659 (2012)
B. D’Autréaux, M.B. Toledano, Nat. Rev. Mol. Cell Biol. 8, 813 (2007)
D.P. Jones, Antioxid. Redox Signal. 8, 1865 (2006)
H. Sies, D.P. Jones, Nat. Rev. Mol. Cell Biol. 21, 363 (2020)
G.G. Martinovich, I.V. Martinovich, S.N. Cherenkevich, H. Sauer, Cell Biochem. Biophys. 58, 75 (2010)
S. Yan, D. Zhang, N. Gu, J. Zheng, A. Ding, Z. Wang et al., J. Nanosci. Nanotechnol. 5, 1185 (2005)
D. Bagchi, V.S. Rathnam, P. Lemmens, I. Banerjee, S.K. Pal, ACS Omega 3, 10877 (2018)
N. Polley, S. Saha, A. Adhikari, S. Banerjee, S. Darbar, S. Das et al., Nanomedicine 10, 2349 (2015)
A. O’Shea, A. Parakh, R.M. Lahoud, S. Hedgire, M.G. Harisinghani, MRS Adv. 5, 2157 (2020)
A. Adhikari, P. Biswas, S. Mondal, M. Das, S. Darbar, A.M. Hameed et al., ChemMedChem 15, 420 (2020)
A. Adhikari, M. Das, S. Mondal, S. Darbar, A.K. Das, S.S. Bhattacharya et al., Biomater. Sci. 7, 4491 (2019)
W. Wang, X. Ji, A. Kapur, H. Mattoussi, MRS Adv. 1, 3741 (2016)
C.R. Reczek, N.S. Chandel, Cancer Cell 33, 949 (2018)
D.K. Singh, A.R. Ray, J. Macromol. Sci. C 40, 69 (2000)
X. Qu, A. Wirsen, A.-C. Albertsson, Polymer 41, 4589 (2000)
J.-Y. Je, S.-K. Kim, in Advances in Food and Nutrition Research, vol. 65, ed. by S.-K. Kim (Academic Press, New York, 2012), p. 321
C. Muanprasat, P. Wongkrasant, S. Satitsri, A. Moonwiriyakit, P. Pongkorpsakol, T. Mattaveewong et al., Biochem. Pharmacol. 96, 225 (2015)
B. Huang, D. Xiao, B. Tan, H. Xiao, J. Wang, J. Yin et al., J. Agric. Food Chem. 64, 245 (2016)
M. Thanou, J.C. Verhoef, H.E. Junginger, Adv. Drug Deliv. Rev. 50, S91 (2001)
H. Hyun, S. Hashimoto-Hill, M. Kim, M.D. Tsifansky, C.H. Kim, Y. Yeo, ACS Biomater. Sci. Eng. 3, 1853 (2017)
S.-J. Yang, F.-H. Lin, H.-M. Tsai, C.-F. Lin, H.-C. Chin, J.-M. Wong et al., Biomaterials 32, 2174 (2011)
C.F. Babbs, Free Radic. Biol. Med. 13(2), 169–181 (1992)
A. Zinger, S. Soriano, G. Baudo, E. De Rosa, F. Taraballi, S. Villapol, Adv. Funct. Mater. 2100722 (2021)
C. Wang, S. Ravi, U.S. Garapati, M. Das, M. Howell, J. Mallela et al., J. Mater. Chem. B 1, 4396 (2013)
S. Lei, K. Tang, Z. Fang, H. Zheng, Cryst. Growth Des. 6, 1757 (2006)
R. Nandi, S. Mishra, T.K. Maji, K. Manna, P. Kar, S. Banerjee et al., J. Mater. Chem. B 5, 3927 (2017)
C.P. LeBel, H. Ischiropoulos, S.C. Bondy, Chem. Res. Toxicol. 5, 227 (1992)
R. Cathcart, E. Schwiers, B.N. Ames, Anal. Biochem. 134, 111 (1983)
S. Mondal, A. Adhikari, M. Das, S. Darbar, A. Alharbi, S.A. Ahmed et al., RSC Adv. 9, 30216 (2019)
A. Giri, N. Goswami, M. Pal, M.T.Z. Myint, S. Al-Harthi, A. Singha et al., J. Mater. Chem. C 1, 1885 (2013)
C. Song, H. Yu, M. Zhang, Y. Yang, G. Zhang, Int. J. Biol. Macromol. 60, 347 (2013)
A. Datta, D. Mandal, S.K. Pal, K. Bhattacharyya, J. Phys. Chem. B 101, 10221 (1997)
S. Sardar, S. Chaudhuri, P. Kar, S. Sarkar, P. Lemmens, S.K. Pal, Phys. Chem. Chem. Phys. 17, 166 (2015)
A. Adhikari, N. Polley, S. Darbar, D. Bagchi, S.K. Pal, Future Sci. OA, 2, FSO146 (2016)
A. Giri, N. Goswami, C. Sasmal, N. Polley, D. Majumdar, S. Sarkar et al., RSC Adv. 4, 5075 (2014)
A. Khorobrykh, V. Terentyev, S. Zharmukhamedov, V. Klimov, Philos. Trans. R. Soc. B 363, 1245 (2008)
Acknowledgments
MD thanks University Grants Commission (UGC), Govt. of India for Junior Research Fellowship. SKP thanks the Indian National Academy of Engineering (INAE) for the Abdul Kalam Technology Innovation National Fellowship, INAE/121/AKF. The authors thank the DBT (WB)-BOOST scheme for the financial grant, 339/WBBDC/1P-2/2013. The authors would like to thank Dr. Uttam Pal for helping in graphical abstract preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mondal, S., Adhikari, A., Ghosh, R. et al. Synthesis and spectroscopic characterization of a target-specific nanohybrid for redox buffering in cellular milieu. MRS Advances 6, 427–433 (2021). https://doi.org/10.1557/s43580-021-00087-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43580-021-00087-0