Abstract
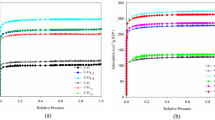
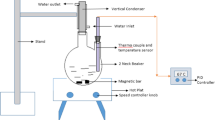
The process of extracting aluminum with sulfuric acid from high-alumina fly ash activated by roasting with Na2CO3 and NaOH as raw materials ware studied. The results revealed that the aluminum in fly ash activated by sodium carbonate reached 93.2% under the optimum conditions (30% H2SO4, 90°C, 120 min, and liquid-to-solid ratio was 5:1). Simultaneously, the leaching rate of aluminum in fly ash activated by sodium hydroxide reached 92%. The study of kinetics suggested that the leaching process with sulfuric acid was controlled through chemical reactions; it conformed to the kinetic equation: 1 − (1 − X) 1/3 = 22.924 exp[− 25174/(RT)]t.
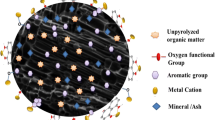
Graphical abstract

Extracting aluminum with sulfuric acid from high-alumina fly ash activated by roasting with Na2CO3 and NaOH as raw materials.



Similar content being viewed by others
Data availability
The authors declare that datasets supporting the conclusions of this article are available within the article.
References
L. Yang, D.L. Li, Z.N. Zhu, M. Xu, X.K. Yan, H.J. Zhang, Effect of the intensification of preconditioning on the separation of unburned carbon from coal fly ash. Fuel 242, 174–183 (2019)
M. Ahmaruzzaman, A review on the utilization of fly ash. Prog. Energy Combustion. Sci. 36, 327–363 (2010)
R.S. Blissett, N.A. Rowson, A review of the multi-component utilisation of coal fly ash. Fuel 97, 1–23 (2012)
Z.Q. Jiang, J. Yang, H.W. Ma, L. Wang, X. Ma, Reaction behaviour of Al2O3 and SiO2 in high alumina coal fly ash during alkali hydrothermal process. Trans. Nonferrous Met. Soc. China. 25, 2065–2072 (2015)
R.C. Bhangare, M. Tiwari, P.Y. Ajmal, S.K. Sahu, G.G. Pandit, Distribution of natural radioactivity in coal and combustion residues of thermal power plants. J. Radioanal. Nucl. Chem. 300, 17–22 (2014)
X.F. Lei, G.X. Qi, Y.I. Sun, H. Xu, Y. Wang, Removal of uranium and gross radioactivity from coal bottom ash by CaCl2 roasting followed by HNO3 leaching. J. Hazard. Mater. 276, 346–352 (2014)
A. González, R. Navia, N. Moreno, Fly ashes from coal and petroleum coke combustion: current and innovative potential applications. Waste Manage. Res. 27, 976–987 (2009)
B.D. Wang, Y.X. Zhou, L. Li, H. Xu, Y.L. Sun, Y. Wang, Novel synthesis of cyano-func-tionalized mesoporous silica nanospheres (MSN) from coal fly ash for removal oftoxic metals from wastewater. J. Hazard. Mater. 345, 76–86 (2018)
J.W. Chen, B.L. Yuan, J.W. Shi, J.C. Yang, M.L. Fu, Reduced graphene oxide and titaniananosheet cowrapped coal fly ash microspheres alternately as a novel photocatalystfor water treatment. Catal. Today 315, 247–254 (2018)
M. Erol, S. Küçükbayrak, A. Ersoy-Meriçboyu, Comparison of the properties of glass, glass-ceramic and ceramic materials produced from coal fly ash. J. Hazard. Mater. 153, 418–425 (2008)
Z.F. Wang, Y.J. Feng, L.N. Zhang, Advances in studies in effects of fine coal ash on agricultural crops. J. Shandong Agric. Univ. (Nat. Sci. Ed.) 34, 152–156 (2003)
A.M. Liu, Z.N. Shi, X.W. Hu, B. Kubikova, M. Boca, B.L. Gao et al., Production of metalsand oxygen from coal fly ash by aluminothothermic and electrochemical reduction process. J. Alloys Compd. 718, 279–287 (2017)
J.P. Li, J.H. Gan, X.W. Li, Leaching of aluminum and iron from boiler slag generated from a typical Chinese Steel Plant. J. Hazard. Mater. 166, 1096–1101 (2009)
Z.T. Yao, M.S. Xia, P.K. Sarker et al., A Review of the alumina recovery from coal fly ash, with a focus in china. Fuel 120, 74–85 (2014)
P.P. Hua, X.J. Hou, J.B. Zhang, S.P. Li, H. Wu, J.D. Anne et al., Distribution and occurrenceof lithium in high-alumina-coal fly ash. Int. J. Coal Geol. 189, 27–34 (2018)
A. Shemi, R.N. Mpana, S. Ndlovu, L.D. van Dyk, V. Sibanda, L. Seepe, Alternative techniques for extracting alumina from coal fly ash. Miner. Eng. 34, 30–37 (2012)
D. Bojinova, R. Teodosieva, Leaching of valuable elements from thermal power plant bottom ash using a thermo-hydrometallurgical process. Waste Manage. Res. 34, 511–517 (2016)
H.Q. Li, J.B. Hui, C.Y. Wang, W.J. Bao, Z.H. Sun, Etraction of alumina from coal fly ash by mixed-alkaline hydrothermal method. Hydrometallurgy 147, 183–187 (2014)
G.H. Bai, Y.H. Qiao, B. Chen, S.L. Chen, Thermal decomposition of coal fly ash by concentrated sulfuric acid and alumina extraction process based on it. Fuel Process. Technol. 92, 1213–1219 (2011)
R.C. Wang, Y.C. Zhai, X.W. Wu, Z.Q. Ning, P.H. Ma, Extraction of alumina from fly ash by ammonium hydrogen sulfate roasting technology. Trans. Nonferrous Met. Soc. China 24, 1596–1603 (2014)
Q.C. Yang, S.H. Ma, S.L. Zheng, R. Zheng, Recovery of alumina from circulating fluidized bed combustion Al-rich fly ash using mild hydrochemical process. Trans. Nonferrous Met. Soc. China 24, 1187–1195 (2014)
G.H. Bai, W. Teng, X.G. Wang, J.G. Qin, P. Xu, P.C. Li, Alkali desilicated coal fly ash as substitute of bauxite in lime-soda sintering process for aluminum production. T Trans. Nonferrous Met. Soc. China. 20, 169–175 (2010)
J.D. Wang, Y.C. Zhai, X.Y. Shen, Extracting Al2O3 from desiliconized fly ash with alkali lime sintering process. Light. Met. 6, 14–16 (2009)
Acknowledgments
The present study was financially supported by the National Natural Science Foundation of China (No. U1710257), Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (No. 2019L0656), Doctoral Research Foundation of Taiyuan University of Science and Technology, China (No. 20142001), Open Foundation Program of Key Laboratory for Ecological Metallurgy of Multimetallic Mineral, Ministry of Education, China (No. 2020003), Supported by Fundamental Research Program of Shanxi Province, China (No. 202103021224281), and graduate education innovation project of Taiyuan University of Science and Technology (SY2022012).
Funding
The National Natural Science Foundation of China (No. U1710257) and Fundamental Research Program of Shanxi Province, China (No. 202103021224281).
Author information
Authors and Affiliations
Contributions
Conceptualization: ACZ and TAZ. Methodology: ACZ and CJL. Software: CJL and XY. Validation and formal analysis: XRY. Writing of the original draft preparation: CJL. Writing, reviewing, and editing of the manuscript: CJL and ZZ. Visualization: CJL. Supervision: YMQ. Project administration: ACZ. Funding acquisition: ACZ. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, CJ., Zhao, AC., Ye, X. et al. Kinetics of aluminum extraction from roasting activated fly ash by sulfuric acid leaching. MRS Communications 14, 48–55 (2024). https://doi.org/10.1557/s43579-023-00494-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43579-023-00494-4