Abstract
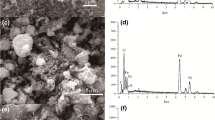
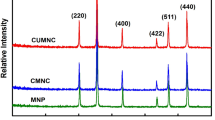
A new Cs+ adsorbent, Fe3O4-chitosan particles combined with ammonium molybdophosphate (Fe3O4–CTS–AMP composite) were synthesized using a facile method through coordination between CTS and AMP. The surface morphology and chemical composition characterization indicated that Fe3O4–CTS particles and AMP particles were bonded tightly and the composite behaved superparamagnetically. Cs+ adsorption properties of Fe3O4–CTS–AMP composites were studied. The adsorption data fitted well with pseudo-second-order kinetic model and Langmuir–Freundlich adsorption isotherms model. The adsorption equilibrium can be reached fast at 5 min. The maximum adsorption capacity was calculated as 177.8 mg g−1. The composite also showed high selectivity for Cs+ in coexistence with high concentration of competing cations. The adsorption mechanism of Cs+ for the composite may mainly be the exchange of Cs+ with NH4+. Therefore, the newly developed adsorbent is a kind of potential efficient composite for Cs+ removal from wastewater.
Graphic abstract










Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Code availability
No software application or custom code is involved in the article.
References
J. Wang, S. Zhuang, Removal of cesium ions from aqueous solutions using various separation technologies. Rev. Environ. Sci. Bio/Technol. 18(2), 231–269 (2019)
Y. Dai et al., Adsorption of cesium using supermolecular impregnated XAD-7 composite: isotherms, kinetics and thermodynamics. J. Radioanal. Nucl. Chem. 321(2), 473–480 (2019)
J. Wang, S. Zhuang, Cesium separation from radioactive waste by extraction and adsorption based on crown ethers and calixarenes. Nucl. Eng. Technol. 52(2), 328–336 (2019)
J. Kiener et al., Activated carbon/transition metal (Ni, In, Cu) hexacyanoferrate nanocomposites for cesium adsorption. Materials 12(8), 1253 (2019)
X. Liu et al., Adsorption removal of cesium from drinking waters: a mini review on use of biosorbents and other adsorbents. Biores. Technol. 160, 142–149 (2014)
M.R. Awual, T. Yaita, T. Taguchi et al., Selective cesium removal from radioactive liquid waste by crown ether immobilized new class conjugate adsorbent. J. Hazard. Mater. 278, 227–235 (2014)
H. Dan et al., Fabrication of AMP/SBA-15 with various morphologies for cesium removal from aqueous solution. J. Sol-Gel. Sci. Technol. 91, 165–177 (2019)
M. Feng, L. Wang, Y. Zhao, C. Liu, Z. Chen, L. Yan et al., Synthesis and characterization of a new activated carbon supported ammonium molybdophosphate composite and its cesium-selective adsorption properties. Radiochim. Acta 98(1), 39–44 (2010)
R. Chakravarty et al., Ammonium molybdophosphate impregnated alumina microspheres as a new generation sorbent for chromatographic 137Cs/137mBa generator. J. Chromatogr. A 1220, 82–91 (2012)
H. Buchwald, W.P. Thistlethwaite, Some cation exchange properties of ammonium 12-molybdophosphate. J. Inorg. Nucl. Chem. 5(4), 341–343 (1958)
J. Krtil, Exchange properties of ammonium salts of 12-heteropolyacids—IV: Cs exchange on ammonium phosphotungstate and phosphomolybdate. J. Inorg. Nucl. Chem. 24(9), 1139–1144 (1962)
T.A. Todd et al., Cesium sorption from concentrated acidic tank wastes using ammonium molybdophosphate-polyacrylonitrile composite sorbents. J. Radioanal. Nucl. Chem. 254(1), 47–52 (2002)
D. Ding et al., Selective removal of cesium by ammonium molybdophosphate—polyacrylonitrile bead and membrane. J. Hazard. Mater. 324, 753–761 (2017)
D. Hao et al., An efficient composite ion exchanger of silica matrix impregnated with ammonium molydbophosphate for cesium uptake from aqueous solution. Chem. Eng. J. 286, 25–35 (2016)
C. Sun et al., Facile preparation of ammonium molybdophosphate/Al-MCM-41 composite material from natural clay and its use in cesium ion adsorption. Eur. J. Inorg. Chem. 2015(12), 2125–2131 (2015)
J. Satyanarayana, G.S. Murthy, P. Sasidhar, Adsorption studies of cesium on a new inorganic exchanger ammonium molybdophosphate—alumina (AMP-Al2O3). J. Radioanal. Nucl. Chem. 242(1), 11–16 (1999)
X. Ye et al., Rubidium and cesium ion adsorption by an ammonium molybdophosphate–calcium alginate composite adsorbent. Colloids Surf. A 342(1–3), 76–83 (2009)
W. Ling, M. Wang, C. Xiong et al., Synthesis, surface modification, and applications of magnetic iron oxide nanoparticles. J. Mater. Res. 34(11), 1–17 (2019)
M. Yamada, A. Fujisawa, K. Morishige et al., Preparation of DNA-immobilized magnetic particles and their utilization as an accumulative material of metal ions. J. Mater. Res. 31(3), 360–369 (2016)
N. Hamed, J. Elham, S. Marziyeh, Biocompatibility and anticancer activity of L-phenyl alanine-coated iron oxide magnetic nanoparticles as potential chrysin delivery system. J. Mater. Res. 33(11), 1602–1611 (2018)
Z. Hui, J. Zhang, R. Sun et al., Preparation of magnetic and photocatalytic cenosphere deposited with Fe3O4/SiO2/Eu-doped TiO2 core/shell nanoparticles. J. Mater. Res. 30(23), 3700–3709 (2015)
W. Xu, J. Yu, Application of Fe3O4 /graphene oxide composite for the separation of Cs(I) and Sr(II) from aqueous solution. J. Radioanal. Nucl. Chem. 303(1), 807–813 (2015)
W. Mu et al., Efficient removal of Cs+ and Sr2+ from aqueous solution using hierarchically structured hexagonal tungsten trioxide coated Fe3O4. Chem. Eng. J. 319, 170–178 (2017)
T. Xia et al., Efficiently remove of Cs(I) by metals hexacyanoferrate modified magnetic Fe3O4-chitosan nanoparticles. Chem. Phys. Lett. 746, 137293 (2020)
H. Yang, H. Yu, J. Sun et al., Facile synthesis of mesoporous magnetic AMP polyhedric composites for rapid and highly efficient separation of Cs+ from water. Chem. Eng. J. 317, 533–543 (2017)
H. Yang et al., A simple synthesis of magnetic ammonium 12-molybdophosphate/graphene oxide nanocomposites for rapid separation of Cs+ from water. J. Radioanal. Nucl. Chem. 318, 955–966 (2018)
A. Aslan, S. Elanthikkal, A. Bozkurt, Chitosan/hollow silica sphere nanocomposites for wound healing application. J. Mater. Res. 34(2), 231–239 (2019)
A. Zhu, X. Luo, S. Dai, Chitosan-poly(acrylic acid) complex modified paramagnetic Fe3O4 nanoparticles for camptothecin loading and release. J. Mater. Res. 24(7), 2307–2315 (2009)
R. Schmuhl, H. Krieg, K. Keizer, Adsorption of Cu(II) and Cr(VI) ions by chitosan: kinetics and equilibrium studies. Water S A 27(1), 1–8 (2001)
R. Brion-Roby, J. Gagnon, S. Nosrati et al., Adsorption and desorption of molybdenum(VI) in contaminated water using a chitosan sorbent. J. Water Process Eng. 23, 13–19 (2018)
L. Dermeche et al., Preparation and characterization of mixed ammonium salts of Keggin phosphomolybdate. Inorg. Chim. Acta 362(11), 3896–3900 (2009)
B. Zhang, R. Hu, D. Sun, T. Wu, Y. Li, Fabrication of magnetite-graphene oxide/MgAl-layered double hydroxide composites for efficient removal of emulsified oils from various oil-in-water emulsions. J. Chem. Eng. Data 63(12), 4689–702 (2018)
D. Hao et al., An efficient composite ion exchanger of silica matrix impregnated with ammonium molydbophosphate for cesium uptake from aqueous solution. Chem. Eng. J. 286, 25–35 (2015)
H. Wei, Q. Xi, X. Chen et al., Molybdenum carbide nanoparticles coated into the graphene wrapping N-doped porous carbon microspheres for highly efficient electrocatalytic hydrogen evolution both in acidic and alkaline media. Adv. Sci. 5(3), 1700733 (2018)
H. Zhang et al., Removal of cesium from low-level radioactive wastewaters using magnetic potassium titanium hexacyanoferrate. Chem. Eng. J. 275, 262–270 (2015)
S. Periyasamy, V. Gopalakannan, N. Viswanathan, Enhanced chromium sorption and quick separation of magnetic hydrotalcite anchored biopolymeric composites using the hydrothermal method. J. Chem. Eng. Data 63(5), 1268–1299 (2018)
D. Ding, Y. Zhao, S. Yang et al., Adsorption of cesium from aqueous solution using agricultural residue-Walnut shell: equilibrium, kinetic and thermodynamic modeling studies. Water Res. 47(7), 2563–2571 (2013)
E.A. Deliyanni, N.A. Travlou, G.Z. Kyzas et al., Graphite oxide/chitosan composite for reactive dye removal. Chem. Eng. J. 217(1), 256–265 (2013)
H. Deng, Y. Li, Y. Huang, et al., An efficient composite ion exchanger of silica matrix impregnated with ammonium molydbophosphate for cesium uptake from aqueous solution. Chem. Eng. J. (2015). S1385894715014515
G.S. Murthy et al., Adsorption of cesium on a composite inorganic exchanger zirconium phosphate—ammonium molybdophosphate. J. Radioanal. Nucl. Chem. 260(1), 109–114 (2004)
Ada K, Ergene A, et al. (2009) Adsorption of Remazol Brilliant Blue R using ZnO fine powder: equilibrium, kinetic and thermodynamic modeling studies. J. Hazard. Mater. 165(1–3):637-44
Y. Park et al., Removal of cobalt, strontium and cesium from radioactive laundry wastewater by ammonium molybdophosphate–polyacrylonitrile (AMP–PAN). Chem. Eng. J. 162(2), 685–695 (2010)
T. Xia et al., Synthesis of polyethylenimine modified Fe3O4 nanoparticles with immobilized Cu2+ for highly efficient proteins adsorption. Colloids Surf., A 443, 552–559 (2014)
Acknowledgments
The authors acknowledge financial supported by Chinese Ministry of Science and Technology (No. 2014FY211000).
Funding
The authors received support from Chinese Ministry of Science and Technology (No. 2014FY211000) for the submitted work.
Author information
Authors and Affiliations
Contributions
Conceptualization: YJ, TX; methodology: TX, LY; formal analysis and investigation: TX, HW; writing original draft preparation: TX; writing review and editing: YJ; resources: YJ; supervision: YJ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing financial interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xia, T., Wu, H., Yin, L. et al. Selective removal of cesium by ammonium molybdophosphate-magnetic Fe3O4-chitosan composites. Journal of Materials Research 36, 2926–2935 (2021). https://doi.org/10.1557/s43578-021-00279-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43578-021-00279-2