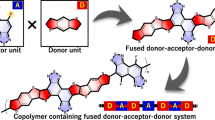
Abstract
The charge transport properties critically depend on the degree of ordering of the chains in the solid state as well as on the density of chemical or structural defects. In general, goodelectronic performance requires strong electronic coupling between adjace nt molecules in the solid-state that yield strong intermolecular π-overlap. Herein, we newly designed and synthesized organic semiconducting materials having both aryl (Ar) and perfluoroaryl (FAr) as substituents for organic electronics along with molecular packing control. Regarding this molecular design, we hypothesized and expected that the Ar and FAr substituents would induce well-defined π-π stacking structure of charge transport units for high performance organic electronics devices.
Similar content being viewed by others
References
(a) Anthony, J. E.; Brooks, J. S.; Eaton, D. L.; Parkin, S. R. J. Am. Chem. Soc. 2001, 123, 9482–9483. (b) Payne, M. M.; Parkin, S. R.; Anthony, J. E.; Kuo, C.-C.; Jackson, T. N. J. Am. Chem. Soc. 2005, 127, 4986–4987.
(a) Patrick, C. R.; Prosser, G. S. Nature 1960, 187, 1021. (b) Williams, J. H. Acc. Chem. Res. 1993, 26, 593–598.
Thalladi, V. R.; Weiss, H.-C.; Bläser, D.; Boese, R.; Nangia, A.; Desiraju, G. R. J. Am. Chem . Soc. 1998, 120, 8702–8710.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okamoto, T., Nakahara, K., Saeki, A. et al. Creation of Face-to-face π-π Stacking of Fused Acene Backbones by Aryl-perfluoroaryl Interactions and Induction of Charge Transport Properties. MRS Online Proceedings Library 1360, 171001 (2011). https://doi.org/10.1557/opl.2012.518
Published:
DOI: https://doi.org/10.1557/opl.2012.518