Abstract
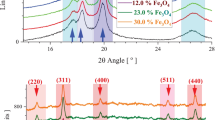
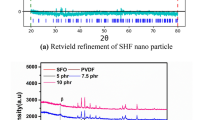
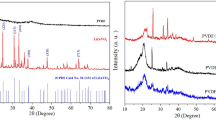
Nanocomposites of polyvinylidene fluoride loaded with various amounts of γ-Fe2O nanoparticles, with an average size ranging between 20 and 40 nm, have been obtained by melt mixing and investigated using various experimental techniques [Superconducting Quantum Interference Device, Mössbauer, and Thermogravimetric Analysis]. Magnetic and Mössbauer measurements confirmed the presence of maghemite and a trace of a paramagnetic iron compound. Magnetic data are consistent with a blocking temperature close to room temperature (RT), showing a decrease in the coercive field as the temperature is increased. A weak exchange bias was noticed in all nanocomposites investigated at all temperatures and tentatively ascribed to surface spin disorder. The temperature dependence of the coercive field obeys the Kneller law. The nanocomposites exhibit superparamagnetic behavior near RT. Most magnetic measurements have been performed below the blocking temperature, revealing thus a complex behavior. The dependence of the mass loss derivative versus temperature, as obtained by thermogravimetric analysis, exhibits a single peak due to the thermal degradation of the polymeric matrix. A weak increase in the thermal stability of the polymeric matrix upon loading with maghemite is reported.







Similar content being viewed by others
References
T. Furukawa: Ferroelectric properties of vinylidene fluoride copolymers. Phase Transitions 18, 143 (1989).
S. Baskaran, N. Ramachandran, X. He, S. Thiruvannamalai, H.J. Lee, H. Heo, Q. Chen, and J.Y. Fu: Giant flexoelectricity in polyvinylidene fluoride films. Phys. Lett. A 375, 2082 (2011).
M. Poulsen, S. Ducharme, M. Poulsen, and S. Ducharme: Why ferroelectric polyvinylidene fluoride is special. IEEE Trans. Dielectr. Electr. Insul. 17, 1028 (2010).
F. He, J. Fan, and L.H. Chan: Preparation and characterization of electrospun poly(vinylidene fluoride)/poly(methyl methacrylate) membrane. High Perform. Polym. 26, 817 (2014).
M. Li, I. Katsouras, C. Piliego, G. Glasser, I. Lieberwirth, P.W.M. Blom, and D.M. de Leeuw: Controlling the microstructure of poly(vinylidene-fluoride) (PVDF) thin films for microelectronics. J. Mater. Chem. C 46, 7695 (2013).
A. Biswas, K. Henkel, D. Schmeißer, and D. Mandal: Comparison of the thermal stability of the α, β and γ phases in poly(vinylidene fluoride) based on in situ thermal Fourier transform infrared spectroscopy. Phase Transitions 90, 1205 (2017).
S. Abdalla, A. Obaid, and F.M. Al-Marzouki: Preparation and characterization of poly(vinylidene fluoride): A high dielectric performance nano-composite for electrical storage. Results Phys. 6, 617 (2016).
V. Sencadas, M.V. Moreira, S. Lanceros-Méndez, A.S. Pouzada, and R. Gregório Filho: Α- to β transformation on PVDF films obtained by uniaxial stretch. Mater. Sci. Forum 514–516, 872 (2006).
N. Cai, J. Zhai, C. Nan, Y. Lin, and Z. Shi: Dielectric, ferroelectric, magnetic, and magnetoelectric properties of multiferroic laminated composites. Phys. Rev. B 68, 224103 (2003).
C. Nan, N. Cai, L. Liu, J. Zhai, Y. Ye, and Y. Lin: Coupled magnetic–electric properties and critical behavior in multiferroic particulate composites. J. Appl. Phys. 94, 5930 (2003).
C.W. Nan, M.I. Bichurin, S. Dong, D. Viehland, and G. Srinivasan: Multiferroic magnetoelectric composites: Historical perspective, status. J. Appl. Physiol. 103, 031101 (2008).
O.M. Lemine, K. Omri, M. Iglesias, V. Velasco, P. Crespo, P. de la Presa, L. El Mir, H. Bouzid, A. Yousif, and A. Al-Hajry: γ-Fe2O3 by sol–gel with large nanoparticles size for magnetic hyperthermia application. J. Alloys Compd. 607, 125 (2014).
D. Cao, H. Li, L. Pan, J. Li, X. Wang, P. Jing, X. Cheng, W. Wang, J. Wang, and Q. Liu: High saturation magnetization of γ-Fe2O3 nano-particles by a facile one-step synthesis approach. Sci. Rep. 6, 1 (2016).
R. Strobel and S.E. Pratsinis: Direct synthesis of maghemite, magnetite and wustite nanoparticles by flame spray pyrolysis. Adv. Powder Technol. 20, 190 (2009).
H. Khurshid, M.H. Phan, P. Mukherjee, and H. Srikanth: Tuning exchange bias in Fe/γ-Fe2O3 core–shell nanoparticles: Impacts of interface and surface spins. Appl. Phys. Lett. 104, 1 (2014).
S. Shekhar, E.P. Sajitha, V. Prasad, and S.V. Subramanyam: High coercivity below percolation threshold in polymer nanocomposite. J. Appl. Phys. 104, 083910 (2008).
M. Chipara, T. George, Y. Xu, R. Skomski, L. Yue, N. Ali, and D.J. Sellmyer: Magnetism of FePt nanoclusters in polyimide. J. Nanomater. 2015, 587847 (2015).
Y. Jin, S. Valloppilly, D.M. Chipara, R. Skomski, M. Chipara, W. Zhang, and D.J. Sellmyer: On polystyrene–block polyisoprene–block polystyrene filled with carbon-coated Ni nanoparticles. J. Mater. Sci. 52, 2452 (2017).
A. Lu, E.L. Salabas, and F. Schüth: Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. 46, 1222 (2007).
S. Radhakrishnan, C. Saujanya, P. Sonar, I. Gopalkrishnan, and J. Yakhmi: Polymer-mediated synthesis of γ-Fe2O3 nano-particles. Polyhedron 20, 1489 (2001).
S. Babay, T. Mhiri, and M. Toumi: Synthesis, structural and spectroscopic characterizations of maghemite γ-Fe2O3 prepared by one-step coprecipitation route. J. Mol. Struct. 1085, 286–293 (2015).
G. Ennas, A. Musinu, G. Piccaluga, D. Zedda, D. Gatteschi, C. Sangregorio, J.L. Stanger, G. Concas, and G. Spano: Characterization of iron oxide nanoparticles in an Fe2O3–SiO2 composite prepared by a sol–gel method. Chem. Mater. 10, 495 (1998).
W. Xiao, Z. Wang, H. Guo, X. Li, J. Wang, S. Huang, and L. Gan: Fe2O3 particles enwrapped by graphene with excellent cyclability and rate capability as anode materials for lithium ion batteries. Appl. Surf. Sci. 266, 148 (2013).
C.T. Fleaca, I. Morjan, R. Alexandrescu, F. Dumitrache, I. Soare, and L. Gavrila-florescu: Magnetic properties of core–shell catalyst nanoparticles for carbon nanotube growth. Appl. Surf. Sci. 255, 5386 (2009).
I.V. Ovsienko, L.Y. Matzuy, N.I. Zakharenko, N.G. Babich, T.A. Len, Y.I. Prylutsky, D. Hui, Y.M. Strzhemechny, and P.C. Eklund: Magnetometric studies of catalyst refuses in nanocarbon materials. Nanoscale Res. Lett. 3, 60 (2008).
M.S. Islam, M. Abdulla-Al-Mamun, J. Kurawaki, Y. Kusumoto, and M.Z. Bin Mukhlish: Hydrothermal novel synthesis of neck-structured hyperthermia-suitable magnetic (Fe3O4, γ-Fe2O3, and α-Fe2O3) nanoparticles. J. Sci. Res. 4, 99 (2012).
H. Wu, G. Wu, and L. Wang: Peculiar porous α-Fe2O3, γ-Fe2O3, and Fe3O4 nanospheres: Facile synthesis and electromagnetic properties. Powder Technol. 269, 443 (2015).
A. Tomescu, R. Alexandrescu, I. Morjan, F. Dumitrache, L. Gavrila-Florescu, R. Birjega, I. Soare, G. Prodan, Z. Bastl, A. Galikova, and J. Pola: Structural and sensing properties of a novel Fe/Fe2O3/polyoxocarbosilane core shell nanocomposite powder prepared by laser pyrolysis. J. Mater. Sci. 42, 1838 (2007).
C.W. Jung and P. Jacobs: Physical and chemical properties of superparamagnetic iron oxide MR contrast agents. Ferumoxides, ferumoxtran, ferumoxsil. Magn. Reson. Imaging 13, 661 (1995).
L. Li, W. Jiang, K. Luo, H. Song, F. Lan, Y. Wu, and Z. Gu: Superparamagnetic iron oxide nanoparticles as MRI contrast agents for non-invasive stem cell labeling and tracking. Theranostics 3, 595 (2013).
M.D. Carvalho, F. Henriques, L.P. Ferreira, M. Godinho, and M.M. Cruz: Iron oxide nanoparticles: The influence of synthesis method and size on composition and magnetic properties. J. Solid State Chem. 201, 144 (2013).
K. Rumpf, P. Granitzer, P.M. Morales, P. Poelt, and M. Reissner: Variable blocking temperature of a porous silicon/Fe3O4 composite due to different interactions of the magnetic nanoparticles. Nanoscale Res. Lett. 7, 445 (2012).
Acknowledgments
The authors acknowledge the Department of Defense Grant “Raman Spectrometer for the Characterization of Advanced Materials and Nanomaterials,” W911NF-15-1-0063, the NSF DMR-1523577: UTRGV-UMN Partnership for Fostering Innovation by Bridging Excellence in Research and Student Success.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuncser, V., Chipara, D., Martirosyan, K.S. et al. Magnetic properties and thermal stability of polyvinylidene fluoride—Fe2O3 nanocomposites. Journal of Materials Research 35, 132–140 (2020). https://doi.org/10.1557/jmr.2019.375
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2019.375