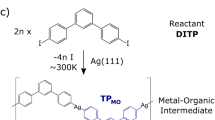
Abstract
The initial steps of the thermal chemistry of Cu(I)-2-(tert-butylimino)-5,5-dimethyl-pyrrolidinate on metal surfaces were characterized using temperature-programmed desorption experiments and density functional theory (DFT). The relative stability of the initial dimer relative to its dissociation on metal surfaces was evaluated. Several molecular desorption temperatures were identified on Ni(110), but all correspond to dimers, either containing the initial Cu ions or after their removal; no monomer was ever detected. DFT calculations also indicated preferential bonding on Cu(110) as a dimer, albeit with a distorted configuration, via the Cu atoms and in registry with the lattice of the substrate. A potential dissociation pathway of the adsorbed dimer was identified involving the partial detachment of the ligands via the scission of one Cu–N bond at the time and migration to adjacent surface sites. This process is accompanied by the reduction of the Cu centers of the metal–organic complex, indicating that it may be the rate-limiting reaction that leads to further fragmentation of the ligands.











Similar content being viewed by others
References
B.C. Gates, M. Flytzani-Stephanopoulos, D.A. Dixon, and A. Katz: Atomically dispersed supported metal catalysts: Perspectives and suggestions for future research. Catal. Sci. Technol. 7, 4259 (2017).
M.K. Samantaray, E. Pump, A. Bendjeriou-Sedjerari, V. D’Elia, J.D.A. Pelletier, M. Guidotti, R. Psaro, and J-M. Basset: Surface organometallic chemistry in heterogeneous catalysis. Chem. Soc. Rev. 47, 8403 (2018).
J. Hämäläinen, M. Ritala, and M. Leskelä: Atomic layer deposition of noble metals and their oxides. Chem. Mater. 26, 786 (2014).
R.G. Gordon: ALD precursors and reaction mechanisms, in atomic layer deposition for semiconductors. In Atomic Layer Deposition for Semiconductors, S.C. Hwang, ed. (Springer, Boston, Massachusetts, 2014); p. 15.
S.E. Koponen, P.G. Gordon, and S.T. Barry: Principles of precursor design for vapour deposition methods. Polyhedron 108 (Suppl. C), 59 (2016).
S.T. Barry, A.V. Teplyakov, and F. Zaera: The chemistry of inorganic precursors during the chemical deposition of films on solid surfaces. Acc. Chem. Res. 51, 800 (2018).
R.G. Gordon, U. Riaz, and D.M. Hoffman: Chemical vapor deposition of aluminum nitride thin films. J. Mater. Res. 7, 1679 (1992).
A.W. Laubengayer and W.F. Gilliam: The alkyls of the third group elements. I. Vapor phase studies of the alkyls of aluminum, gallium and indium1. J. Am. Chem. Soc. 63, 477 (1941).
M.A. Malik, P. O’Brien, M. Motevalli, A.C. Jones, and T. Leedham: X-ray crystal structures of bis-2,2,6,6-tetramethylheptane-3,5-dionatolead(II) and bis-2,2-dimethyl-6,6,7,7,8,8,8-heptafluorooctane-3,5-dionatolead(II): Compounds important in the metalorganic chemical vapour deposition (MOCVD) of lead-containing films. Polyhedron 18, 1641 (1999).
B.S. Lim, A. Rahtu, and R.G. Gordon: Atomic layer deposition of transition metals. Nat. Mater. 2, 749 (2003).
A. Devi: ‘Old Chemistries’ for new applications: Perspectives for development of precursors for MOCVD and ALD applications. Coord. Chem. Rev. 257, 3332 (2013).
M. Xu, H. Tiznado, B-C. Kang, M. Bouman, I. Lee, and F. Zaera: Mechanistic details of atomic layer deposition (ALD) processes. J. Korean Phys. Soc. 51, 1063 (2007).
F. Zaera: The surface chemistry of thin film atomic layer deposition (ALD) processes for electronic device manufacturing. J. Mater. Chem. 18, 3521 (2008).
Q. Ma, H. Guo, R.G. Gordon, and F. Zaera: Uptake of copper acetamidinate ALD precursors on nickel surfaces. Chem. Mater. 22, 352 (2010).
Q. Ma, H. Guo, R.G. Gordon, and F. Zaera: Surface chemistry of copper(I) acetamidinates in connection with atomic layer deposition (ALD) processes. Chem. Mater. 23, 3325 (2011).
T. Kim, Y. Yao, J.P. Coyle, S.T. Barry, and F. Zaera: Thermal chemistry of Cu(I)-iminopyrrolidinate and Cu(I)-guanidinate atomic layer deposition (ALD) precursors on Ni(110) single-crystal surfaces. Chem. Mater. 25, 3630 (2013).
Y. Yao, J.P. Coyle, S.T. Barry, and F. Zaera: Thermal decomposition of copper iminopyrrolidinate atomic layer deposition (ALD) precursors on silicon oxide surfaces. J. Phys. Chem. C 120, 14149 (2016).
Y. Yao and F. Zaera: Thermal chemistry of copper acetamidinate atomic layer deposition precursors on silicon oxide surfaces studied by XPS. J. Vac. Sci. Technol., A 34, 01A101 (2016).
B. Chen, Y. Duan, Y. Yao, Q. Ma, J.P. Coyle, S.T. Barry, A.V. Teplyakov, and F. Zaera: Activation of the dimers and tetramers of metal amidinate atomic layer deposition precursors upon adsorption on silicon oxide surfaces. J. Vac. Sci. Technol., A 35, 01B124 (2017).
Y. Yao, J.P. Coyle, S.T. Barry, and F. Zaera: Effect of the nature of the substrate on the surface chemistry of atomic layer deposition precursors. J. Chem. Phys. 146, 052806 (2017).
B. Chen, J.P. Coyle, S.T. Barry, and F. Zaera: Rational design of metalorganic complexes for the deposition of solid films: Growth of metallic copper with amidinate precursors. Chem. Mater. 31, 1681 (2019).
J. Guerrero-Sánchez, N. Takeuchi, and F. Zaera: Density functional theory study of the surface adsorption and dissociation of copper(I) acetamidinates on Cu(110) surfaces. J. Phys. Chem. C 123, 4341 (2019).
H. Tiznado, M. Bouman, B.C. Kang, I. Lee, and F. Zaera: Mechanistic details of atomic layer deposition (ALD) processes for metal nitride film growth. J. Mol. Catal. A: Chem. 281, 35 (2008).
F. Zaera: The surface chemistry of atomic layer depositions of solid thin films. J. Phys. Chem. Lett. 3, 1301 (2012).
F. Zaera: Mechanisms of surface reactions in thin solid film chemical deposition processes. Coord. Chem. Rev. 257, 3177 (2013).
K. Christmann, O. Schober, G. Ertl, and M. Neumann: Adsorption of hydrogen on nickel single crystal surfaces. J. Chem. Phys. 60, 4528 (1974).
M. Bouman and F. Zaera: The surface chemistry of atomic layer deposition (ALD) processes for metal nitride and metal oxide film growth. ECS Trans. 33, 291 (2010).
M. Bouman and F. Zaera: Reductive eliminations from amido metal complexes: Implications for metal film deposition. J. Electrochem. Soc. 158, D524 (2011).
Z. Li, S.T. Barry, and R.G. Gordon: Synthesis and characterization of copper(I) amidinates as precursors for atomic layer deposition (ALD) of copper metal. Inorg. Chem. 44, 1728 (2005).
T.J.J. Whitehorne, J.P. Coyle, A. Mahmood, W.H. Monillas, G.P.A. Yap, and S.T. Barry: Group 11 amidinates and guanidinates: Potential precursors for vapour deposition. Eur. J. Inorg. Chem. 2011, 3240 (2011).
F.T. Edelmann: Chapter two—Recent progress in the chemistry of metal amidinates and guanidinates: Syntheses, catalysis and materials, in advances in organometallic chemistry. In Advances in Organometallic Chemistry, Vol. 61, F.H. Anthony and J.F. Mark, eds. (Academic Press, Amsterdam, 2013); p. 55.
J.P. Coyle, A. Kurek, P.J. Pallister, E.R. Sirianni, G.P.A. Yap, and S.T. Barry: Preventing thermolysis: Precursor design for volatile copper compounds. Chem. Commun. 48, 10440 (2012).
Q. Ma, F. Zaera, and R.G. Gordon: Thermal chemistry of copper(I)-N,N’-di-sec-butylacetamidinate on Cu(110) single-crystal surfaces. J. Vac. Sci. Technol. A 30, 01A114 (2012).
F. Zaera: A thermal desorption and X-ray photoelectron spectroscopy study of the surface chemistry of iron pentacarbonyl. J. Vac. Sci. Technol. A 7, 640 (1989).
Y. Yao, J. Guerrero-Sánchez, N. Takeuchi, and F. Zaera: Coadsorption of formic acid and hydrazine on Cu(110) single-crystal surfaces. J. Phys. Chem. C 123, 7584 (2019).
H.E. Farnsworth, R.E. Schlier, T.H. George, and R.M. Burger: Application of the ion bombardment cleaning method to titanium, germanium, silicon, and nickel as determined by low-energy electron diffraction. J. Appl. Phys. 29, 1150 (1958).
K. Klier, A.C. Zettlemoyer, and H. Leidheiser, Jr.: Chemisorption of carbon monoxide on Ni(110) and (100) nickel crystal faces. J. Chem. Phys. 52, 589 (1970).
P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, S. Cavazzoni, D. Ceresoli, G. Chiarotti, L., M. Cococcioni, I. Dabo, A.D. Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A.P. Seitsonen, A. Smogunov, P. Umari and R.M. Wentzcovitch: Quantum espresso: A modular and open-source software project for quantum simulations of materials. J. Phys.: Condens. Matter. 21, 395502 (2009).
I.A. Pašti, A. Jovanović, A.S. Dobrota, S.V. Mentus, B. Johansson, and N.V. Skorodumova: Atomic adsorption on pristine graphene along the Periodic Table of Elements—From PBE to non-local functionals. Appl. Surf. Sci. 436, 433 (2018).
Acknowledgments
Financial support for this project was provided by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, Materials Sciences and Engineering (MSE) Division, under Award No. DE-SC0001839. N.T. and J.G.S. thank DGAPA-UNAM Project IN100516, and Conacyt Grant A1-S-9070 of the Call of Proposals for Basic Scientific Research 2017–2018 for partial financial support. N.T. thanks DGAPA-UNAM for a scholarship at the University of California, Riverside. Calculations were performed in the DGCTIC-UNAM Supercomputing Center, project LANCAD-UNAM-DGTIC-051.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guerrero-Sánchez, J., Chen, B., Takeuchi, N. et al. Role of oligomer structures in the surface chemistry of amidinate metal complexes used for atomic layer deposition of thin films. Journal of Materials Research 35, 720–731 (2020). https://doi.org/10.1557/jmr.2019.293
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2019.293