Abstract
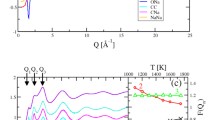
Carbonate glasses can be formed routinely in the system K2CO3–MgCO3. The enthalpy of formation for one such 0.55K2CO3–0.45MgCO3 glass was determined at 298 K to be 115.00 ± 1.21 kJ/mol by drop solution calorimetry in molten sodium molybdate (3Na2O·MoO3) at 975 K. The corresponding heat of formation from oxides at 298 K was −261.12 ± 3.02 kJ/mol. This ternary glass is shown to be slightly metastable with respect to binary crystalline components (K2CO3 and MgCO3) and may be further stabilized by entropy terms arising from cation disorder and carbonate group distortions. This high degree of disorder is confirmed by 13C MAS NMR measurement of the average chemical shift tensor values, which show asymmetry of the carbonate anion to be significantly larger than previously reported values. Molecular dynamics simulations show that the structure of this carbonate glass reflects the strong interaction between the oxygen atoms in distorted carbonate anions and potassium cations.





Similar content being viewed by others
References
C.A. Angell: Formation of glasses from liquids and biopolymers. Science 267, 1924–1935 (1995).
M.C. Wilding, M. Wilson, O.L.G. Alderman, C. Benmore, J.K.R. Weber, J.B. Parise, A. Tamalonis, and L. Skinner: Low-dimensional network formation in molten sodium carbonate. Sci. Rep. 6, 1–7 (2016).
M.C. Wilding, M. Wilson, M.C.C. Ribeiro, C.J. Benmore, J.K.R. Weber, O.L.G. Alderman, A. Tamalonis, and J.B. Parise: The structure of liquid alkali nitrates and nitrites. Phys. Chem. Chem. Phys. 19, 21625–21638 (2017).
M. Wilson, M.C.C. Ribeiro, M.C. Wilding, C. Benmore, J.K.R. Weber, O. Alderman, A. Tamalonis, and J.B. Parise: Structure and liquid fragility in sodium carbonate. J. Phys. Chem. A 122, 1071–1076 (2018).
M.J. Genge, A.P. Jones, and G.D. Price: An infrared and Raman study of carbonate glasses-implications for carbonatite magmas. Geochim. Cosmochim. Acta 59, 927–937 (1995).
S. Sen, D.C. Kaseman, B. Colas, D.E. Jacob, and S.M. Clark: Hydrogen bonding induced distortion of CO3 units and kinetic stabilization of amorphous calcium carbonate: Results from 2D C-13 NMR spectroscopy. Phys. Chem. Chem. Phys. 18, 20330–20337 (2016).
W. Eitel and W. Skaliks: Double carbonates of alkalis and alkaline earths. Z. Anorg. Allg. Chem. 183, 263–286 (1929).
M.J. Genge, A.P. Jones, and G.D. Price: An infrared and Raman study of carbonate glasses-implications for the structure of carbonatite magmas. Geochim. Cosmochim. Acta 59, 927–937 (1995).
M.J. Genge, G.D. Price, and A.P. Jones: Molecular dynamics simulations of CaCO3 melts to mantle pressures and temperatures—Implications for carbonatite magmas. Earth Planet. Sci. Lett. 131, 225–238 (1995).
D.P. Dobson, A.P. Jones, R. Rabe, T. Sekine, K. Kurita, T. Taniguchi, T. Kondo, T. Kato, O. Shimomura, and S. Urakawa: In situ measurement of viscosity and density of carbonate melts at high pressure. Earth Planet. Sci. Lett. 143, 207–215 (1996).
S.E. Ragone, R.K. Datta, D.M. Roy, and O.F. Tuttle: The system potassium carbonate-magnesium carbonate. J. Phys. Chem. 70, 3360–3361 (1966).
R.K. Datta, D.M. Roy, S.P. Faile, and O.F. Tuttle: Glass formation in carbonate systems. J. Am. Ceram. Soc. 47, 153 (1964).
T. Forland and W.A. Weyl: formation of a sulfate glass. J. Am. Ceram. Soc. 33, 186–187 (1950).
D.R. MacFarlane: Attempted glass formation in pure KHSO4. J. Am. Ceram. Soc. 67, C–28 (1984).
L.G. van Uitert and W.H. Grodkiewicz: Nitrate glasses. Mater. Res. Bull. 6, 283–292 (1971).
A.P. Jones, M. Genge, and L. Carmody: Carbonate melts and carbonatites. In Carbon in Earth, R.M. Hazen, A.P. Jones, and J.A. Baross, eds. (The Mineralogical Society of America, Chantilly, Virginia, 2013); pp. 289–322.
S.K. Sharma and B. Simons: Raman study of K2CO3–MgCO3 glasses. In Carnegie Institute of Washington Yearbook, Vol. 79, H.S. Yoder, ed. (The Carnegie Institution of Washington, Washington DC, 1980); pp. 322–326.
A. Navrotsky: Progress and new directions in calorimetry: A 2014 perspective. J. Am. Ceram. Soc. 97, 3349–3359 (2014).
S.K. Sahu, L.A. Boatner, and A. Navrotsky: Formation and dehydration enthalpy of potassium hexaniobate. J. Am. Ceram. Soc. 100, 304–311 (2017).
R. Shivaramaiah and A. Navrotsky: Energetics of order-disorder in layered magnesium aluminum double hydroxides with inter layer carbonate. Inorg. Chem. 54, 3253–3259 (2015).
L.A. Chai and A. Navrotsky: Thermochemistry of carbonate-pyroxene equilibria. Contrib. Mineral. Petrol. 114, 139–147 (1993).
I. Kiseleva, A. Navrotsky, I.A. Belitsky, and B.A. Fursenko: Thermochemistry of natural potassium sodium calcium leonhardite and its cation-exchanged forms. Am. Mineral. 81, 668–675 (1996).
A. Navrotsky, R.L. Putnam, C. Winbo, and E. Rosen: Thermochemistry of double carbonates in the K2CO3–CaCO3 system. Am. Mineral. 82, 546–548 (1997).
I. Tarina, A. Navrotsky, and H. Gan: Direct calorimetric measurment of enthalpics in diopside-anorthite-wollastonaite melts at 1773 K. Geochim. Cosmochim. Acta 58, 3665–3673 (1994).
A. Navrotsky, P. Maniar, and R. Oestrike: Energetics of glasses in the system diopside-anorthite-forsterite. Contrib. Mineral. Petrol. 105, 81–86 (1990).
R. Hon, D.F. Weill, R.B. Kasper, and A. Navrotsky: Enthalpies of mixing of glasses in the system albite-anorthite-diopside. Trans., Am. Geophys. Union 58, 1243 (1977).
A. Golubkova, M. Merlini, and M.W. Schmidt: Crystal structure, high-pressure, and high-temperature behavior of carbonates in the K2Mg(CO3)2–Na2Mg(CO3)2 join. Am. Mineral. 100, 2458–2467 (2015).
A. Shatskiy, K.D. Litasov, Y.N. Palyanov, and E. Ohtani: Phase relations on the K2CO3–CaCO3–MgCO3 join at 6 GPa and 900–1400 °C: Implications for incipient melting in carbonated mantle domains. Am. Mineral. 101, 437–447 (2016).
A. Shatskiy, Y.M. Borzdov, K.D. Litasov, I.S. Sharygin, Y.N. Palyanov, and E. Ohtani: Phase relationships in the system K2CO3–CaCO3 at 6 GPa and 900–1450 °C. Am. Mineral. 100, 223–232 (2015).
A. Shatskiy, I.S. Sharygin, P.N. Gavryushkin, K.D. Litasov, Y.M. Borzdov, A.V. Shcherbakova, Y. Higo, K-i. Funakoshi, Y.N. Palyanov, and E. Ohtani: The system K2CO3–MgCO3 at 6 GPa and 900–1450 °C. Am. Mineral. 98, 1593–1603 (2013).
A.I. Alekseev, L.D. Barinova, N.P. Rogacheva, and O.V. Kulinich: Thermodynamic values of binary carbonate salts K2CO3·MgCO3·nH2O. J. Appl. Chem. USSR 57, 1168–1172 (1984).
H.W. Papenguth, R.J. Kirkpatrick, B. Montez, and P.A. Sandberg: C-13 MAS NMR-spectroscopy of inorganic and biogenic carbonates. Am. Mineral. 74, 1152–1158 (1989).
F. Marc Michel, J. MacDonald, J. Feng, B.L. Phillips, L. Ehm, C. Tarabrella, J.B. Parise, R.J. Reeder: Structural characteristics of synthetic amorphous calcium carbonate. Chem. Mater. 20, 4720–4728 (2008).
F.M. Michel, J. McDonald, J. Feng, B.L. Phillips, L. Ehm, C. Tarabrella, J.B. Parise, and R.J. Reeder: Structural characteristics of synthetic amorphous calcium carbonate. Geochim. Cosmochim. Acta 72, A626 (2008).
T.F. Sevelsted, D. Herfort, and J. Skibsted: C-13 chemical shift anisotropies for carbonate ions in cement minerals and the use of C-13, Al-27 and Si-29 MAS NMR in studies of Portland cement including limestone additions. Cem. Concr. Res. 52, 100–111 (2013).
J.K. Moore, J.A. Surface, A. Brenner, L.S. Wang, P. Skemer, M.S. Conradi, and S.E. Hayes: Quantitative identification of metastable magnesium carbonate minerals by solid-state C-13 NMR spectroscopy (vol 49, pg 657, 2015). Environ. Sci. Technol. 49, 1986 (2015).
H. Nebel, M. Neumann, C. Mayer, and M. Epple: On the structure of amorphous calcium carbonate—A detailed study by solid-state NMR spectroscopy. Inorg. Chem. 47, 7874–7879 (2008).
S.C. Kohn, R.A. Brooker, and R. Dupree: C-13 MAS NMR—A method for studying CO2 speciation in glasses. Geochim. Cosmochim. Acta 55, 3879–3884 (1991).
R.A. Brooker, S.C. Kohn, J.R. Holloway, P.F. McMillan, and M.R. Carroll: Solubility, speciation and dissolution mechanisms for CO2 in melts on the NaAlO2–SiO2 join. Geochim. Cosmochim. Acta 63, 3549–3565 (1999).
Z.W. Su and P. Coppens: Relativistic X-ray elastic scattering factors for neutral atoms Z = 1–54 from multiconfiguration Dirac–Fock wavefunctions in the 0–12 Å−1 sin θ/λ range, and six-Gaussian analytical expressions in the 0–6 Å−1 range (vol A53, pg 749, 1997). Acta Crystallogr., Sect. A: Found. Crystallogr. 54, 357 (1998).
K.F. Hesse and B. Simons: Crystal structure of synthetic K2Mg(CO3)2. Z. Kristallogr. 161, 289–292 (1982).
P.D. Ihinger: An experimental study of the interaction of water with granitic melt. Ph.D. thesis, California Institute of Technology, Pasedena, California, 1991.
A. Navrotsky: High temperature reaction calorimetry applied to metastable and nanophase materials. J. Therm. Anal. Calorim. 57, 653–658 (1999).
A. Navrotsky: High-temperature oxide melt calorimetry of oxides and nitrides. J. Chem. Thermodyn. 33, 859–871 (2001).
J. Herzfeld and A.E. Berger: Sideband intensities in NMR-spectra of samples spinning at the magic angle. J. Chem. Phys. 73, 6021–6030 (1980).
K. Eichele: HBA. Ph.D. thesis, Universitaet Tuebingen, Tuebingen, Germany, 2015.
J. Tissen, G.J.M. Janssen, and J.P. Vandereerden: Molecular dynamics simulation fo binary mixtures of molten alkali carboantes. Mol. Phys. 82, 101–111 (1994).
M.F. Costa and M.C.C. Ribeiro: Molecular dynamics of molten Li2CO3–K2CO3 (vol 138, pg 61, 2008). J. Mol. Liq. 142, 161 (2008).
M.C.C. Ribeiro: First sharp diffraction peak in the fragile liquid Ca0.4K0.6(NO3)1.4. Phys. Rev. B 61, 3297–3302 (2000).
M.C.C. Ribeiro: Ionic dynamics in the glass-forming liquid Ca0.4K0.6(NO3)1.4: A molecular dynamics study with a polarizable model. Phys. Rev. B 63, 0942051–09420510 (2001).
M.C.C. Ribeiro: Molecular dynamics study on the glass transition in Ca0.4K0.6(NO3)1.4. J. Phys. Chem. B 107, 9520–9527 (2003).
D.T. Cromer and J.B. Mann: X-ray scattering functions compuited from numerical Hartree–Fock functions. Acta Crystallogr. A 24, 321–324 (1968).
G. Shen, V.B. Prakapenka, M.L. Rivers, and S.R. Sutton: Structural investigation of amorphous materials at high pressures using the diamond anvil cell. Rev. Sci. Instrum. 74, 3021–3026 (2003).
Y. Kono, C. Kenney-Benson, D. Hummer, H. Ohfuji, C. Park, G. Shen, Y. Wang, A. Kavner, and C.E. Manning: Ultralow viscosity of carbonate melts at high pressures. Nat. Commun. 5, 5:5091–5 (2014).
Acknowledgments
M.C.W. and P.A.B. would like to acknowledge the funding support from the EPSRC under grant EP/R036225/1. M.W. is grateful for the support from the EPSRC Centre for Doctoral Training, Theory and Modeling in Chemical Sciences, under grant EP/L015722/1. R.A.B. was funded by the NERC Thematic Grant consortium NE/M000419/1. The diffraction study was performed at HPCAT (Sector 16) of the Advanced Photon Source (APS). The Advanced Photon Source is a US DOE Office of Science User facility, operated for the DOE Office of Science by Argonne National Laboratory under contract DE-AC02-06CH11357. Calorimetry at UC Davis was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division under Award DE-FG02ER1474.
Author information
Authors and Affiliations
Corresponding author
Additional information
This author was an editor of this journal during the review and decision stage. For the JMR policy on review and publication of manuscripts authored by editors, please refer to http://www.mrs.org/editor-manuscripts/.
Rights and permissions
About this article
Cite this article
Wilding, M.C., Phillips, B.L., Wilson, M. et al. The structure and thermochemistry of K2CO3–MgCO3 glass. Journal of Materials Research 34, 3377–3388 (2019). https://doi.org/10.1557/jmr.2019.250
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2019.250