Abstract
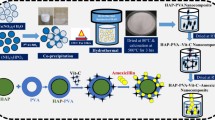
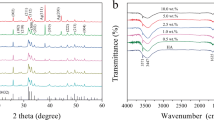
In this work, a multifunctional system was developed in which antibacterial and luminescent properties were inserted into the matrix of hydroxyapatite (HAp) and its efficiency as a support for an antineoplastic drug was evaluated, aiming its application in the treatment of osseous diseases. The precipitation method was used for the synthesis of HAp, EuCl3 was used for the incorporation of Eu3+ as imaging agents, silver nanoparticles (AgNPs) with antimicrobial function were used, and a model of drug, 5-fluorouracil (5FU) was used. The developed material is characterized by several techniques, where crystalline peaks attributed to HAp were identified in the X-ray diffractogram, whereas the luminescence spectrum of the material presented emissions attributed to the Eu3+ ion. The identification and the uniform distribution of AgNPs, 5FU, and Eu3+ were confirmed by mapping the sample using energy-dispersive spectroscopy. The measurements indicated that 82% (±2.8) of 5FU was incorporated into the HAp matrix, and a gradual and increasing release of it as a function of time was observed. Assays carried out for different bacteria confirmed the antimicrobial action of the samples and the efficiency of the drug inserted into the matrix. An in vitro assay showed the bioactivity of the material and its potential to bind to living osseous tissue.









Similar content being viewed by others
References
L. Ouyang, D. He, J. Zhang, G. He, B. Jiang, Q. Wang, Z. Chen, J. Pan, Y. Li, and L. Guo: Selective bone targeting 5-fluorouracil prodrugs: Synthesis and preliminary biological evaluation. Bioorg. Med. Chem. 19, 3750–3756 (2011).
J.T. Buijs and G.V. Pluijm: Osteotropic cancers: From primary tumor to bone. Cancer Lett. 273, 177–193 (2009).
H. Hirabayashi and J. Fujisaki: Bone-specific drug delivery systems approaches via chemical modification of bone-seeking agents. Clin. Pharmacokinet. 42, 1319–1330 (2003).
J. Cawthray, E. Wasan, and K. Wasan: Bone-seeking agents for the treatment of bone disorders. Drug Delivery Transl. Res. 7, 466–481 (2017).
E. Andronescu, M. Ficai, G. Voicu, D. Ficai, M. Maganu, and A. Ficai: Synthesis and characterization of collagen/hydroxyapatite: Magnetite composite material for bone cancer treatment. J. Mater. Sci.: Mater. Med. 21, 2237–2242 (2010).
C. Marques, J.M.F. Ferreira, E. Adronescu, D. Ficai, M. Sonmez, and A. Ficai: Multifunctional materials for bone cancer treatment. Int. J. Nanomed. 9, 2713–2725 (2014).
R. Kuehl, P.S. Brunetto, A.K. Woischnig, M. Varisco, Z. Rajacic, Vosbeck, L. Terracciano, K.M. Fromm, and N. Khanna: Preventing implant-associated infections by silver coating. Antimicrob. Agents Chemother. 60, 2467–2475 (2016).
A.M. Costa and V.V. Silva: Nanotechnology strategies for diagnosis and treatment of cancer. R. Saúde Meio Amb. 5, 1–13 (2017).
Y. Liu, H. Miyoshi, and M. Nakamura: Nanomedicine for drug delivery and imaging: A promising avenue for cancer therapy and diagnosis using targeted functional nanoparticles. Int. J. Cancer 120, 2527–2537 (2007).
J.R. Lopes, J.A. Oliveira, and A.A. Esteves: Synthesis and characterization of hydroxyapatite powders [Ca10(PO4)6(OH)2] obtained from the sol–gel process. Foco 6, 55–72 (2015).
K. Lin and J. Chang: Structure and properties of hydroxyapatite for biomedical applications. Hydroxyapatite Biomed. Appl., 3–19 (2015). doi: https://doi.org/10.1016/B978-1-78242-033-0.00001-8.
A. Ashokan, D. Menon, S. Nair, and M. Koyakutty: A molecular receptor targeted, hydroxyapatite nanocrystal based multi-modal contrast agent. Biomaterials 31, 2606–2616 (2010).
F. Chen, P. Huang, Y.J. Zhu, J. Wu, C.L. Zhang, and D.X. Cui: The photoluminescence, drug delivery and imaging properties of multifunctional Eu3+/Gd3+ dual-doped hydroxyapatite nanorods. Biomaterials 32, 9031–9039 (2011).
S.V. de Oliveira, S.N. Cavalcanti, G.P. Rabelo, E.M. Araújo, and M.V.L. Fook: Hydroxyapatite infrared analysis. In VI Congr. Nac. Eng. Mec. (Conem), Campina Grande, PB, 2010.
C.L. Tseng, J.C. Chen, Y.C. Fang, F.H. Lin, and T.P. Tang: Development of lattice-inserted 5-fluorouracil-hydroxyapatite nanoparticles as a chemotherapeutic delivery system. J. Biomater. Appl. 30, 388–397 (2015).
P. Singh, G. Tyagi, R. Mehrotra, and A. Bakhshi: Thermal stability studies of 5-fluorouracil using diffuse reflectance infrared spectroscopy. Drug Test. Anal. 1, 240–244 (2009).
K.S.W. Sing, D.H. Everett, R.A.W. Haul, L. Moscou, R.A. Pirotti, and J. Rouquerol: Reporting physisorption data for gas/solid systems in. Pure Appl. Chem. 57, 603–619 (1985).
Y. Jin, A. Wang, G. Wu, H. Yin, S. Liu, B. Chen, F. Liu, and X. Li: Synthesis of different sized and porous hydroxyapatite nanorods without organic modifiers and their 5-fluorouracil release performance. Mater. Sci. Eng., C 57, 14–23 (2015).
P. Martin, G. Carlot, A. Chevarier, C. Den-Auwer, and G. Panczer: Mechanisms involved in thermal diffusion of rare earth elements in apatite. J. Nucl. Mater. 275, 268–276 (1999).
L.A. Zavala-Sanchez, G.A. Hirata, E. Novitskaya, K. Karandikar, M. Herrera, and O.A. Graeve: Distribution of Eu2+ and Eu3+ ions in hydroxyapatite: A cathodoluminescence and Raman study. ACS Biomater. Sci. Eng. 1, 1306–1313 (2015).
O.A. Graeve, R. Kanakala, A. Madadi, B.C. Williams, and K.C. Glass: Luminescence variations in hydroxyapatites doped with Eu2+ and Eu3+ ions. Biomaterials 31, 4259–4267 (2010).
ANVISA: Standardization of Antimicrobial Susceptibility Tests by Disk-Diffusion: Standard Approved, Octave ed. (2003); p. 23, ISBN 1-56238-485-6.
K.L. Nair, S. Jagadeeshan, S.A. Nair, and G.S. Kumar: Biological evaluation of 5-fluorouracil nanoparticles for cancer chemotherapy and its dependence on the carrier. PLGA. Int. J. Nanomedic. 6, 1685–1697 (2011).
H. Luo, D. Ji, C. Li, Y. Zhu, G. Xiong, and Y. Wan: Layered nanohydroxyapatite as a novel nanocarrier for controlled delivery of 5-fluorouracil. Int. J. Pharm. 513, 17–25 (2016).
J.G. Hardman, L.E. Limbird, and A.G. Gilman: Goodman & Gilman’s the Pharmacological Basis of Therapeutics, 10th ed. (McGraw-Hill, New York, 2001). Available at: https://pt.wikipedia.org/wiki/Especial:Fontes_de_livros/0071354697. ISBN 0-07-135469-7.
S.D. Solomon, M. Bahadory, A.V. Jeyarajasigam, S.A. Rutkowsky, and C. Boritz: Synthesis and study of silver nanoparticles. J. Chem. Educ. 84, 322–325 (2007).
T. Kokubo and H. Takadama: How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 27, 2907–2915 (2006).
Acknowledgment
This research was supported by Foundation for protection of research in the state of Bahia—FAPESB.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barbosa, A.A., Júnior, S.A. & de Vasconcelos Ferraz, A. Study of a luminescent and antibacterial biomaterial based on hydroxyapatite as support for an antineoplastic drug. Journal of Materials Research 34, 1922–1930 (2019). https://doi.org/10.1557/jmr.2019.139
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2019.139