Abstract
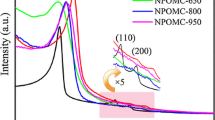
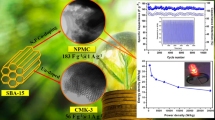
Ordered mesoporous carbons (OMCs) are appealing alternatives to conventional porous activated carbon applied to electronic energy storage and conversion devices. Nitrogen-doped OMC (NOMC) was prepared with a soft-template strategy directly using task-specific ionic liquid with dicyanamide anion as the nitrogen dopant, and utilized as supercapacitors for the first time. Compared with pristine OMC, NOMC showed excellent electrochemical capacitive behavior in 6 M KOH electrolyte. NOMC possessed a high specific capacitance of 427 F/g at a current density of 1 A/g and exhibited a stable cycle life (almost 98% retained at a current density of 5 A/g after 2000 cycles). The outstanding capacitive performance of NOMC was ascribed to the synergetic effects of its bimodal mesoporous structure, large specific surface area (1919 m2/g), and nitrogen doping (3.52 wt%), which help to accelerate the ion diffusion, increase the surface charge storage, and intensify pseudo-capacitive reactions.







Similar content being viewed by others
References
P. Simon and Y. Gogotsi: Materials for electrochemical capacitors. Nat. Mater. 7(11), 845 (2008).
L.L. Zhang and X.S. Zhao: Carbon-based materials as supercapacitor electrodes. Chem. Soc. Rev. 38(9), 2520 (2009).
A. Elmouwahidi, Z. Zapata-Benabithe, F. Carrasco-Marin, and C. Moreno-Castilla: Activated carbons from KOH-activation of argan (Argania spinosa) seed shells as supercapacitor electrodes. Bioresour. Technol. 111, 185 (2012).
D. Zhang, Y. Hao, L. Zheng, Y. Ma, H. Feng, and H. Luo: Nitrogen and sulfur co-doped ordered mesoporous carbon with enhanced electrochemical capacitance performance. J. Mater. Chem. A 1(26), 7584 (2013).
R. Ryoo, S.H. Joo, M. Kruk, and M. Jaroniec: Ordered mesoporous carbons. Adv. Mater. 13(9), 677 (2001).
Y. Zhai, Y. Dou, D. Zhao, P.F. Fulvio, R.T. Mayes, and S. Dai: Carbon materials for chemical capacitive energy storage. Adv. Mater. 23(42), 4828 (2011).
S.R.P. Gnanakan, K. Karthikeyan, S. Amaresh, S.J. Cho, G.J. Park, and Y.S. Lee: New application and electrochemical characterization of a nickel-doped mesoporous carbon for supercapacitors. J. Alloys Compd. 509(41), 9858 (2011).
W. Xiong, M. Liu, L. Gan, Y. Lv, Y. Li, L. Yang, Z. Xu, Z. Hao, H. Liu, and L. Chen: A novel synthesis of mesoporous carbon microspheres for supercapacitor electrodes. J. Power Sources 196(23), 10461 (2011).
J.X. Xu, Y. Zhao, C. Shen, and L.H. Guan: Sulfur- and nitrogen-doped, ferrocene-derived mesoporous carbons with efficient electrochemical reduction of oxygen. ACS Appl. Mater. Interfaces 5(23), 12594 (2013).
H. Guo and Q. Gao: Boron and nitrogen co-doped porous carbon and its enhanced properties as supercapacitor. J. Power Sources 186(2), 551 (2009).
T. Zhou, H. Wang, J. Key, S. Ji, V. Linkov, and R. Wang: Highly dispersed ultrafine Pt nanoparticles on hydrophilic N-doped carbon tubes for improved methanol oxidation. RSC Adv. 3(38), 16949 (2013).
J. Goscianska, A. Olejnik, and R. Pietrzak: Adsorption of L-phenylalanine on ordered mesoporous carbons prepared by hard template method. J. Taiwan Inst. Chem. Eng. 45(2), 347 (2014).
Y.D. Xia and R. Mokaya: Synthesis of ordered mesoporous carbon and nitrogen-doped carbon materials with graphitic pore walls via a simple chemical vapor deposition method. Adv. Mater. 16(17), 1553 (2004).
J. Liu, T.Y. Yang, D.W. Wang, G.Q.M. Lu, D.Y. Zhao, and S.Z. Qiao: A facile soft-template synthesis of mesoporous polymeric and carbonaceous nanospheres. Nat. Commun. 4(1), 94 (2013).
Y.P. Zhai, Y.Q. Dou, X.X. Liu, S.S. Park, C.S. Ha, and D.Y. Zhao: Soft-template synthesis of ordered mesoporous carbon/nanoparticle nickel composites with a high surface area. Carbon 49(2), 545 (2011).
A. Vinu: Two-dimensional hexagonally-ordered mesoporous carbon nitrides with tunable pore diameter, surface area and nitrogen content. Adv. Funct. Mater. 18(5), 816 (2008).
H. Chen, M. Zhou, Z. Wang, S.Y. Zhao, and S.Y. Guan: Rich nitrogen-doped ordered mesoporous phenolic resin-based carbon for supercapacitors. Electrochim. Acta 148, 187 (2014).
J. Yu, M. Guo, F. Muhammad, A. Wang, F. Zhang, Q. Li, and G. Zhu: One-pot synthesis of highly ordered nitrogen-containing mesoporous carbon with resorcinol-urea-formaldehyde resin for CO2 capture. Carbon 69, 502 (2014).
A. Lu, A. Kiefer, W. Schmidt, and F. Schüth: Synthesis of polyacrylonitrile-based ordered mesoporous carbon with tunable pore structures. Chem. Mater. 16(1), 100 (2004).
J. Wei, D. Zhou, Z. Sun, Y. Deng, Y. Xia, and D. Zhao: A controllable synthesis of rich nitrogen doped ordered mesoporous carbon for CO2 capture and supercapacitors. Adv. Funct. Mater. 23(18), 2322 (2013).
Á. Sánchez-Sánchez, F. Suárez-García, A. Martínez-Alonso, and J. Tascón: Synthesis, characterization and dye removal capacities of N-doped mesoporous carbons. J. Colloid Interface Sci. 450, 91 (2015).
T.Q. Lin, I.W. Chen, F.X. Liu, C.Y. Yang, H. Bi, F.F. Xu, and F.Q. Huang: Nitrogen-doped mesoporous carbon of extraordinary capacitance for electrochemical energy storage. Science. 350(6267), 1508 (2015).
T. Welton: Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev. 99(8), 2071 (1999).
Z. Ma, J. Yu, and S. Dai: Preparation of inorganic materials using ionic liquids. Adv. Mater. 22(2), 261 (2010).
C. Liao, R. Liu, X.S. Hou, X.G. Sun, and S. Dai: Easy synthesis of poly(ionic liquid) for use as a porous carbon precursor. Carbon 29(1), 78 (2014).
X. Wang and S. Dai: Ionic liquids as versatile precursors for functionalized porous carbon and carbon–oxide composite materials by confined carbonization. Angew. Chem., Int. Ed. 49(37), 6664 (2010).
J.S. Lee, X. Wang, H. Luo, G.A. Baker, and S. Dai: Facile ionothermal synthesis of microporous and mesoporous carbons from task specific ionic liquids. J. Am. Chem. Soc. 131(13), 4596 (2009).
J.P. Paraknowitsch, J. Zhang, D.S. Su, A. Thomas, and M. Antonietti: Ionic liquids as precursors for nitrogen-doped graphitic carbon. Adv. Mater. 22(1), 87 (2010).
J. Zhou, B. Yang, Z.J. Li, L.C. Lei, and X.W. Zhang: Selective adsorption of naphthalene in aqueous solution on mesoporous carbon functionalized by task-specific ionic liquid. Ind. Eng. Chem. Res. 54(8), 2329 (2015).
P. Scovazzo, D. Havard, M. Mcshea, S. Mixon, and D. Morgan: Long-term, continuous mixed-gas dry fed CO2/CH4 and CO2/N2 separation performance and selectivities for room temperature ionic liquid membranes. J. Membr. Sci. 327(s 1–2), 41 (2009).
Y. Meng, D. Gu, F.Q. Zhang, Y.F. Shi, H.F. Yang, Z. Li, C.Z. Yu, B. Tu, and D.Y. Zhao: Ordered mesoporous polymers and homologous carbon frameworks: Amphiphilic surfactant templating and direct transformation. Angew. Chem., Int. Ed. 44(43), 7053 (2005).
M. Xie, H. Dong, D. Zhang, X. Guo, and W. Ding: Simple synthesis of highly ordered mesoporous carbon by self-assembly of phenol–formaldehyde and block copolymers under designed aqueous basic/acidic conditions. Carbon 49(7), 2459 (2011).
H. Chen, M. Zhou, Z. Wang, S. Zhao, and S. Guan: Rich nitrogen-doped ordered mesoporous phenolic resin-based carbon for supercapacitors. Electrochim. Acta 148, 187 (2014).
N. Soin, S.S. Roy, S. Sharma, T. Thundat, and J.A. Mclaughlin: Electrochemical and oxygen reduction properties of pristine and nitrogen-doped few layered graphene nanoflakes (FLGs). J. Solid State Electrochem. 257(3), 300 (2014).
T. Panja, D. Bhattacharjya, and J.S. Yu: Nitrogen and phosphorus co-doped cubic ordered mesoporous carbon as a supercapacitor electrode material with extraordinary cyclic stability. Proc. SPIE 3570(35), 176 (2015).
Q. Shi, R. Zhang, Y. Lv, Y. Deng, A.A. Elzatahrya, D. Zhao, Q. Shi, R. Zhang, Y. Lv, and A.A. Elzatahrya: Nitrogen-doped ordered mesoporous carbons based on cyanamide as the dopant for supercapacitor. Carbon 84(1), 335 (2015).
B. Xu, S. Hou, G. Cao, F. Wu, and Y. Yang: Sustainable nitrogen-doped porous carbon with high surface areas prepared from gelatin for supercapacitors. J. Mater. Chem. 22(36), 19088 (2012).
S. Yang, X. Wu, C. Chen, H. Dong, W. Hu, and X. Wang: Spherical α-Ni(OH)2 nanoarchitecture grown on graphene as advanced electrochemical pseudocapacitor materials. Chem. Commun. 48(22), 2773 (2012).
S.K. Meher and G.R. Rao: Enhanced activity of microwave synthesized hierarchical MnO2 for high performance supercapacitor applications. J. Power Sources 215, 317 (2012).
C. Kim and K.S. Yang: Electrochemical properties of carbon nanofiber web as an electrode for supercapacitor prepared by electrospinning. Appl. Phys. Lett. 83(6), 1216 (2003).
ACKNOWLEDGMENT
The authors are grateful for financial support from the National Natural Science Foundation of China (Project No. 21406044).
Author information
Authors and Affiliations
Corresponding author
Supplementary Material
43578_2017_32020404_MOESM1_ESM.docx
Supplementary Information: Nitrogen-doped Ordered Mesoporous Carbon Using Task-specific Ionic Liquid as a Dopant for High-performance Supercapacitors (approximately 40.0 KB)

Rights and permissions
About this article
Cite this article
Zhou, J., Bao, L., Wu, S. et al. Nitrogen-doped ordered mesoporous carbon using task-specific ionic liquid as a dopant for high-performance supercapacitors. Journal of Materials Research 32, 404–413 (2017). https://doi.org/10.1557/jmr.2016.473
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2016.473