Abstract
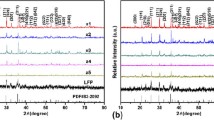
Two novel series of cathode materials LiFe1−xMxPO4/C (x ≈ 0.0040; M = Mn, Fe, Co, Ni, Cu, and Zn) composites based on metal phthalocyanines (MPc) and metal tetrasulfophthalocyanines (MPcTs) to modify lithium iron phosphate (LiFePO4) for lithium-ion batteries (LIBs) are in situ prepared by solvothermal and calcination techniques. Structures and morphologies of all the composites are characterized by normal methods. To evaluate the electrochemical performance of the composites, the charge/discharge capabilities, rate performance, cycling stabilities, cyclic voltammetry profiles, and electrochemical impedance spectroscopy plots of the LIBs using them as cathode materials are measured carefully. The results indicate that most of the composites deliver highly improved initial discharge capacity and show remarkable reversibility and cycling stabilities. Especially, composites using MPcTs as additives are more efficient for the improvement of specific capacity, rate capability, reversibility, and cycling stability.









Similar content being viewed by others
References
A. Stein: Energy storage: Batteries take charge. Nat. Nanotechnol. 6, 262 (2011).
L.X. Yuan, Z.H. Wang, W.X. Zhang, X.L. Hu, J.T. Chen, Y.H. Huang, and J.B. Goodenough: Development and challenges of LiFePO4 cathode material for lithium-ion batteries. Energy Environ. Sci. 4, 269 (2011).
A.K. Padhi, K.S. Nanjundaswamy, and C. Masquelier: Effect of structure on the Fe3+/Fe2+ redox couple in iron phosphates. J. Electrochem. Soc. 144, 1609 (1997).
J. Wang and X. Sun: Understanding and recent development of carbon coating on LiFePO4 cathode materials for lithium-ion batteries. Energy Environ. Sci. 5, 5163 (2012).
H.C. Shin, K.Y. Chung, W.S. Min, D.J. Byun, H. Jang, and B.W. Cho: Asymmetry between charge and discharge during high rate cycling in LiFePO4–in situ x-ray diffraction study. Electrochem. Commun. 10, 536 (2008).
G. Wang, B. Wang, X. Wang, J. Park, S. Dou, H. Ahn, and K. Kim: Sn/graphene nanocomposite with 3D architecture for enhanced reversible lithium storage in lithium ion batteries. J. Mater. Chem. 19, 8378 (2009).
D. Baster, K. Zheng, W. Zając, K. Świerczek, and J. Molenda: Toward elucidation of delithiation mechanism of zinc-substituted LiFePO4. Electrochim. Acta 92, 79 (2013).
H. Zhang, D. Liu, X. Qian, C. Zhao, and Y. Xu: A novel nano structured LiFePO4/C composite as cathode for Li-ion batteries. J. Power Sources 249, 431 (2014).
W.K. Kim, W.H. Ryu, D.W. Han, S. Lim, J.Y. Eom, and H.S. Kwon: Fabrication of graphene embedded LiFePO4 using a catalyst assisted self assembly method as a cathode material for high power lithium-ion batteries. ACS Appl. Mater. Interfaces 6, 4731 (2014).
N. Zhou, E. Uchaker, H.Y. Wang, M. Zhang, S.Q. Liu, Y.N. Liu, X. Wu, G. Cao, and H. Li: Additive-free solvothermal synthesis of hierarchical flower-like LiFePO4/C mesocrystal and its electrochemical performance. RSC Adv. 3, 19366 (2013).
M.Y. Cho, H. Kim, H. Kim, Y.S. Lim, K.B. Kim, J.W. Lee, K. Kang, and K.C. Roh: Size-selective synthesis of mesoporous LiFePO4/C microspheres based on nucleation and growth rate control of primary particles. J. Mater. Chem. A 2, 5922 (2014).
G. Zeng, R. Caputo, D. Carriazo, L. Luo, and M. Niederberger: Tailoring two polymorphs of LiFePO4 by efficient microwave-assisted synthesis: A combined experimental and theoretical study. Chem. Mater. 25, 3399 (2013).
G. Qin, S. Xue, Q. Ma, and C. Wang: The morphology controlled synthesis of 3D networking LiFePO4 with multiwalled-carbon nanotubes for Li-ion batteries. CrystEngComm 16, 260 (2014).
S. Yang, M. Hu, L. Xi, R. Ma, Y. Dong, and C.Y. Chung: Solvothermal synthesis of monodisperse LiFePO4 micro hollow spheres as high performance cathode material for lithium ion batteries. ACS Appl. Mater. Interfaces 5, 8961–8967 (2013).
A. Vu and A. Stein: Multiconstituent synthesis of LiFePO4/C composites with hierarchical porosity as cathode materials for lithium ion batteries. Chem. Mater. 23, 3237 (2011).
H. Ni, J. Liu, and L.Z. Fan: Carbon-coated LiFePO4–porous carbon composites as cathode materials for lithium ion batteries. Nanoscale 5, 2164 (2013).
D. Bhuvaneswari and N. Kalaiselvi: In situ carbon coated LiFePO4/C microrods with improved lithium intercalation behavior. Phys. Chem. Chem. Phys. 16, 1469 (2014).
J. Mun, H.W. Ha, and W. Choi: LiFePO4 nano in reduced graphene oxide framework for efficient high-rate lithium storage. J. Power Sources 251, 386 (2014).
Z.X. Chi, W. Zhang, F.Q. Cheng, J.T. Chen, A.M. Cao, and L.J. Wan: Optimizing the carbon coating on LiFePO4 for improved battery performance. RSC Adv. 4, 7795 (2014).
D. Arumugam, G.P. Kalaignan, and P. Manisankar: Synthesis and electrochemical characterizations of nano-crystalline LiFePO4 and Mg-doped LiFePO4 cathode materials for rechargeable lithium-ion batteries. J. Solid State Electrochem. 13, 301 (2009).
B. Wang, B. Xu, T. Liu, P. Liu, C. Guo, S. Wang, Q. Wang, Z. Xiong, D. Wang, and X.S. Zhao: Mesoporous carbon-coated LiFePO4 nanocrystals co-modified with graphene and Mg2+ doping as superior cathode materials for lithium ion batteries. Nanoscale 6, 986 (2014).
G. Meligrana, F. Di Lupo, S. Ferrari, M. Destro, S. Bodoardo, N. Garino, and C. Gerbaldi: Surfactant-assisted mild hydrothermal synthesis to nanostructured mixed orthophosphates LiMnyFe1−yPO4/C lithium insertion cathode materials. Electrochim. Acta 105, 99 (2013).
H. Shu, X. Wang, Q. Wu, B. Hu, X. Yang, Q. Wei, Q. Liang, Y. Bai, M. Zhou, C. Wu, M. Chen, A. Wang, and L. Jiang: Improved electrochemical performance of LiFePO4/C cathode via Ni and Mn co-doping for lithium-ion batteries. J. Power Sources 237, 149 (2013).
G. Ramos-Sanchez, A. Callejas-Tovar, L.G. Scanlon, and P.B. Balbuena: DFT analysis of Li intercalation mechanisms in the Fe-phthalocyanine cathode of Li-ion batteries. Phys. Chem. Chem. Phys. 16, 743 (2014).
J. Yamaki and A. Yamaji: Phthalocyanine cathode materials for secondary lithium cells. J. Electrochem. Soc. 129, 5 (1982).
H. Kantekin, G. Dilber, and A. Nas: Microwave-assisted synthesis and characterization of a new soluble metal-free and metallophthalocyanines peripherally fused to four 18-membered tetrathiadiaza macrocycles. J. Organomet. Chem. 695, 1210 (2010).
A. Shaabani, N. Safari, and A. Bazgir: Synthesis of the tetrasulfo- and tetranitrophthalocyanine complexes under solvent-free and reflux conditions using microwave irradiation. Synth. Commun. 33, 1717 (2003).
S.Y. Chung, J.T. Bloking, and Y.M. Chiang: Electronically conductive phospho-olivines as lithium storage electrodes. Nat. Mater. 1, 123 (2002).
G.X. Wang, L. Yang, S.L. Bewlay, Y. Chen, H.K. Liu, and J.H. Ahn: Electrochemical properties of carbon coated LiFePO4 cathode materials. J. Power Sources 146, 521 (2005).
G. Qin, Q. Wu, J. Zhao, Q. Ma, and C. Wang: C/LiFePO4/multi-walled carbon nanotube cathode material with enhanced electrochemical performance for lithium-ion batteries. J. Power Sources 248, 588 (2014).
C. Miao, P. Bai, Q. Jiang, S. Sun, and X. Wang: A novel synthesis and characterization of LiFePO4 and LiFePO4/C as a cathode material for lithium-ion battery. J. Power Sources 246, 232 (2014).
D. Andre, M. Meiler, K. Steiner, Ch. Wimmer, T. Soczka-Guth, and D.U. Sauer: Characterization of high-power lithium-ion batteries by electrochemical impedance spectroscopy. I. Experimental investigation. J. Power Sources 196, 5334 (2011).
R. Chen, Y. Wu, and X.Y. Kong: Monodisperse porous LiFePO4/C microspheres derived by microwave-assisted hydrothermal process combined with carbothermal reduction for high power lithium-ion batteries. J. Power Sources 258, 246 (2014).
Y. Lin, H. Pan, M. Gao, and Y. Liu: Effects of reductive conditions on the microstructure and electrochemical properties of sol-gel derived LiFePO4/C. J. Electrochem. Soc. 154, A1124 (2007).
ACKNOWLEDGMENTS
The authors thank the National Natural Science Foundation of China (Nos. 21401149 and 21371143), the National Basic Research Program (973 Program 2013CB934001), “Science and Technology plan project of Xi’an City [No. CXY1438(6)], Key Laboratory Research and Establish Program of Shaanxi Education Section (No. 11JS112), and Special Foundation of Shaanxi Education Section (No. 12JK0603) for the financial support of this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, R., Zhang, R., Xu, B. et al. Highly improving the electrochemical performance of LiFePO4 modified by metal phthalocyanines as cathode materials. Journal of Materials Research 30, 645–653 (2015). https://doi.org/10.1557/jmr.2014.396
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2014.396