Abstract
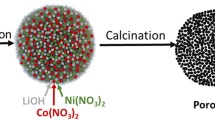
The porous Li1.2Ni0.13Co0.13Mn0.54O2 nanoplate is prepared by colloidal crystal template assembled by the poly (methyl methacrylate) (PMMA) beads. Scanning electron microscopy and transmission electron microscopy results show that the nanoplates of porous solid solution cathodes are composed of nanoparticles with a size range of 30 nm, which interweave together forming an open porous structure. Electrochemical tests show that porous Li1.2Ni0.13Co0.13Mn0.54O2 cathode could deliver higher discharge capacity than that of bulk Li1.2Ni0.13Co0.13Mn0.54O2 cathode at all C-rates. The enhanced structural stability reflected by high ratios of integrated Intensity I(003)/I(104) and lattice parameters c/a, high specific surface area, a fast reaction and ionic diffusion kinetics of the nanoplates are considered attributable to the improved electrochemical properties.







Similar content being viewed by others
REFERENCES
C.S. Johnson, J-S. Kim, C. Lefief, N. Li, J.T. Vaughey, and M.M. Thackeray: The significance of the Li2MnO3 component in ‘composite’ xLi2MnO3·(1-x)LiMn0.5Ni0.5O2 electrodes. Electrochem. Commun. 6, 1085 (2004).
J. Liu, Q.Y. Wang, B.R. Jayan, and A. Manthiram: Carbon-coated high capacity layered Li[Li0.2Mn0.54Ni0.13Co0.13]O2 cathodes. Electrochem. Commun. 12, 750 (2010).
S-H. Kang, P. Kempgens, S. Greenbaum, A.J. Kropf, K. Amine, and M.M. Thackeray: Interpreting the structural and electrochemical complexity of 0.5Li2MnO3·0.5LiMO2 electrodes for lithium batteries (M = Mn0.5−xNi0.5−xCo2x, 0 ≤ x ≤ 0.5). J. Mater. Chem. 17, 2069 (2007).
Z. Lu and J.R. Dahn: Understanding the anomalous capacity of Li/Li [NixLi (1/3− 2x/3) Mn (2/3− x/3)]O2 cells using in situ x-ray diffraction and electrochemical studies. J. Electrochem. Soc. 149, A815 (2002).
J. Liu, B.R. Jayan, and A. Manthiram: Conductive surface modification with aluminum of high capacity layered Li[Li0.2Mn0.54Ni0.13Co0.13]O2 cathodes. J. Phys. Chem. C 114, 9528 (2010).
N. Yabuuchi, K. Yoshii, S.T. Myung, I. Nakai, and S. Komaba: Detailed studies of a high-capacity electrode material for rechargeable batteries, Li2MnO3-LiCo1/3Ni1/3Mn1/3O2. J. Am. Chem. Soc. 133, 4404 (2011).
P.D. Yang, D.Y. Zhao, D.I. Margolese, B.F. Chmelka, and G.D. Stucky: Generalized syntheses of large-pore mesoporous metal oxides with semicrystalline frameworks. Nature 396, 152 (1998).
P.D. Yang, D.Y. Zhao, D.I. Margolese, B.F. Chmelka, and G.D. Stucky: Block copolymer templating syntheses of mesoporous metal oxides with large ordering lengths and semicrystalline framework. Chem. Mater. 11, 2813 (1999).
F.B. Su, J.H. Zeng, P. Bai, L. Lv, P. Guo, H. Sun, H. Li, J. Yu, J. Lee, and X. Zhao: Template synthesis of mesoporous carbon microfibers as a catalyst support for methanol electrooxidation. Ind. Eng. Chem. Res. 46, 9097 (2007).
J.W. Long, M.B. Sassin, A.E. Fischer, and D.R. Rolison: Multifunctional MnO2−carbon nanoarchitectures exhibit battery and capacitor characteristics in alkaline electrolytes. J. Phys. Chem. C 113, 17595 (2009).
Z.Y. Wang, E.R. Kiesel, and A. Stein: Silica-free syntheses of hierarchically ordered macroporous polymer and carbon monoliths with controllable mesoporosity. J. Mater. Chem. 18, 2194 (2008).
Z.Y. Wang, M.A. Fierke, and A. Stein: Porous carbon/Tin (IV) oxide monoliths as anodes for lithium-ion batteries. J. Electrochem. Soc. 155, A658 (2008).
A.H. Lu, W. Schmidt, B. Spliethoff, and F. Schüth: Synthesis of ordered mesoporous carbon with bimodal pore system and high pore volume. Adv. Mater. 15, 1602 (2003).
H. Yan, C.F. Blanford, J.C. Lytle, B. Carter, W.H. Smyrl, and A. Stein: Influence of processing conditions on structures of 3D ordered macroporous metals prepared by colloidal crystal templating. Chem. Mater. 13, 4314 (2001).
C.M. Doherty, R.A. Caruso, B.M. Smarsly, and C.J. Drummond: Colloidal crystal templating to produce hierarchically porous LiFePO4 electrode materials for high power lithium ion batteries. Chem. Mater. 21, 2895 (2009).
C.M. Doherty, R.A. Caruso, and C.J. Drummond: High performance LiFePO4 electrode materials: Influence of colloidal particle morphology and porosity on lithium-ion battery power capability. Energy Environ. Sci. 3, 813 (2010).
G.X. Wang, H. Liu, J. Liu, S. Qiao, G. Lu, P. Munroe, and H. Ahn: Mesoporous LiFePO4/C nanocomposite cathode materials for high power lithium ion batteries with superior performance. Adv. Mater. 22, 4944 (2010).
A. Vu and A. Stein: Multiconstituent synthesis of LiFePO4/C composites with hierarchical porosity as cathode materials for lithium ion batteries. Chem. Mater. 23, 3237 (2011).
S. Yuvaraj, L. Fan-Yuan, C. Tsong-Huei, and Y. Chuin-Tih: Thermal decomposition of metal nitrates in air and hydrogen environments. J. Phys. Chem. B 107, 1044 (2003).
B.T. Holland, C.F. Blanford, T. Do, and A. Stein: Synthesis of highly ordered, three-dimensional, macroporous structures of amorphous or crystalline inorganic oxides, phosphates, and hybrid composites. Chem. Mater. 11, 795 (1999).
T.W. Wang, O. Sel, I. Djerdj, and B. Smarsly: Preparation of a large mesoporous CeO2 with crystalline walls using PMMA colloidal crystal templates. Colloid Polym. Sci. 285, 1 (2006).
W. Liu, G.C. Farrington, F. Chaput, and B. Dunn: Synthesis and electrochemical studies of spinel phase LiMn2O4 cathode materials prepared by the pechini process. J. Electrochem. Soc. 143, 879 (1996).
D. Tonti, M.J. Torralvo, E. Enciso, I. Sobrados, and J. Sanz: Three-dimensionally ordered macroporous lithium manganese oxide for rechargeable lithium batteries. Chem. Mater. 20, 4783 (2008).
H.W. Yan, C.F. Blanford, B.T. Holland, W.H. Smyrl, and A. Stein: General synthesis of periodic macroporous solids by templated salt precipitation and chemical conversion. Chem. Mater. 12, 1134 (2000).
J.M. Zheng, X.B. Wu, and Y. Yang: A comparison of preparation method on the electrochemical performance of cathode material Li[Li0.2Mn0.54Ni0.13Co0.13]O2 for lithium ion battery. Electrochim. Acta 56, 3071 (2011).
Z.H. Lu, L.Y. Beaulieu, R.A. Donaberger, C.L. Thomas, and J.R. Dahn: Synthesis, structure, and electrochemical behavior of Li[NixLi1/3-2x/3Mn2/3-x/3]O2. J. Electrochem. Soc. 149, A778 (2002).
C.S. Johnson, N. Li, J.T. Vaughey, S.A. Hackney, and M.M. Thackeray: Lithium-manganese oxide electrodes with layered-spinel composite structures xLi2MnO3·(1-x)Li1+yMn2-yO4 (0 < x <1, 0 ≤ y ≤ 0.33) for lithium batteries. Electrochem. Commun. 7, 528 (2005).
D. Pasero, V. McLaren, S.D. Souza, and A.R. West: Oxygen nonstoichiometry in Li2MnO3: An alternative explanation for its anomalous electrochemical activity. Chem. Mater. 17, 345 (2005).
Z.H. Chen and J.R. Dahn: Effect of ZrO2 coating on the structure and electrochemistry of LixCoO2 when cycled to 4.5V. Electrochem. Solid-State Lett. 5, A213 (2002).
X.M. Liu, W. Gao, and B. Ji: Synthesis of LiNi1/3Co1/3Mn1/3O2 nanoparticles by modified Pechini method and their enhanced rate capability. J Sol-Gel Sci. Technol. 61, 56 (2012).
J.H. Kim, C.W. Park, and Y.K. Sun: Synthesis and electrochemical behavior of Li[Li0.1Ni0.35-x/2CoxMn0.55-x/2]O2 cathode material. Solid State Ionics 164, 43 (2003).
V. Subramanian, K. Karki, and B. Rambabu: Synthesis and electrochemical properties of submicron LiNi0.5Co0.5O2. Solid State Ionics 175, 315 (2004).
C.H. Zhao, W.P. Kang, Q.B. Xue, and Q. Shen: Polymerization-pyrolysis-assisted nanofabrication of solid solution Li1.2Ni0.13Co0.13Mn0.54O2 for lithium-ion battery cathodes. J. Nanopart. Res. 14, 1240 (2012).
C. Yu, G.S. Li, X.F. Guan, J. Zheng, L.P. Li, and T.W. Chen: Composites Li2MnO3·LiMn1/3Ni1/3Co1/3O2: Optimized synthesis and applications as advanced high-voltage cathode for batteries working at elevated temperatures. Electrochim. Acta 81, 283 (2012).
Y.J. Hong, S.H. Choi, C.M. Sim, J.K. Lee, and Y.C. Kang: Effect of boric acid on the properties of Li2MnO3·LiNi0.5Mn0.5O2 composite cathode powders prepared by large-scale spray pyrolysis with droplet classifier. MRS Bull. 47, 4359 (2012).
Y.K. Sun, M.J. Lee, C.S. Yoon, J. Hassoun, K. Amine, and B. Scrosati: The role of AlF3 coatings in improving electrochemical cycling of Li-enriched nickel-manganese oxide electrodes for Li-ion batteries. Adv. Mater. 24, 1192 (2012).
D-K. Lee, S-H. Park, K. Amine, H.J. Bang, J. Parakash, and Y-K. Sun: High capacity Li[Li0.2Ni0.2Mn0.6]O2 cathode materials via a carbonate co-precipitation method. J. Power Sources 162, 1346 (2006).
R. Santhanam and B. Rambabu: High rate cycling performance of Li1.05Ni1/3Co1/3Mn1/3O2 materials prepared by sol-gel and co-precipitation methods for lithium-ion batteries. J. Power Sources 196, 4313 (2010).
ACKNOWLEDGMENTS
This work is supported by the Natural Science Foundation of China (Grant Nos. 11275121 and 21241002), Science and Technology Committee of Shanghai (Grant Nos. 11DZ110020 and 10ZR1411300), and Shanghai Leading Academic Disciplines Project (Grant No. S30109).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, Y., Zhuang, H., Ma, Q. et al. Synthesis of porous Li2MnO3-LiNi1/3Co1/3Mn1/3O2 nanoplates via colloidal crystal template. Journal of Materials Research 28, 1505–1511 (2013). https://doi.org/10.1557/jmr.2013.136
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2013.136