Abstract
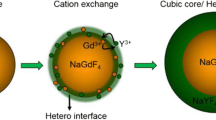
Uniform ytterbium ion and erbium ion codoped gadolinium oxyfluoride (GdOF: Yb3+, Er3+) hollow nanospheres of 100-nm diameter were synthesized via the nanoscale Kirkendall approach, using colloidal nanospheres of ytterbium ion and erbium ion codoped gadolinium hydroxide [Gd(OH)3: Yb3+, Er3+] as sacrificial templates and titanium tetrafluoride as fluorine source under hydrothermal condition. The shell thickness of the as-synthesized GdOF: Yb3+, Er3+ hollow nanospheres can be facilely tuned from 31 to 13 nm by controlling reaction temperature and reaction time. The upconversion emission color could be adjusted from red to yellow to green when the host lattices variedfrom gadolinium (III) oxide to gadolinium oxyfluoride to gadolinium fluoride. Furthermore, the formation mechanism of the hollow GdOF: Yb3+, Er3+ nanospheres was found to depend on the fluorine source.








Similar content being viewed by others
References
J. Hu, T.W. Odom, and C.M. Lieber: Chemistry and physics in one-dimension: Synthesis and properties of nanowires and nanotubes. Acc. Chem. Res. 32, 435 (1999).
J. Goldberger, R. He, S. Lee, Y. Zhang, H. Yan, H. Choi, and P. Yang: Single crystal gallium nitride nanotubes. Nature 422, 599 (2003).
J.T. Zhang, Y. Tang, K. Lee, and M. Ouyang: Nonepitaxial growth of hybrid core-shell nanostructures with large lattice mismatches. Science 327, 1634 (2010).
Y. Yin, R.M. Rioux, C.K. Erdonmez, S. Hughes, G.A. Somorjai, and A.P. Alivisatos: Formation of hollow nanocrystals through the nanoscale Kirkendall effect. Science 304, 711 (2004).
J. Park, K. An, Y. Hwang, J.G. Park, H.J. Noh, J.Y. Kim, J.H. Park, N.M. Hwang, and T. Hyeon: Ultra-large-scale syntheses of monodisperse nanocrystals. Nat. Mater. 3, 891 (2004).
M. Yin, Y. Gu, I.L. Kuskovsky, T. Andelman, Y. Zhu, G.F. Neumark, and S.J. O’Brien: Zinc oxide quantum rods. J. Am. Chem. Soc. 126, 6206 (2004).
F. Shi, M.K. Tse, M.M. Pohl, A. Brückner, M. Zhang, and M. Beller: Tuning catalytic activity between homogeneous and heterogeneous catalysis: Improved activity and selectivity of free nano-Fe2O3 in selective oxidations. Angew. Chem. Int. Ed. 46, 8866 (2007).
Z.X. Li, L.L. Li, H.P. Zhou, Q. Yuan, C. Chen, L.D. Sun, and C.H. Yan: Colour modification action of an upconversion photonic crystal. Chem. Commun. 43, 6616 (2009).
X.M. Lu, H.Y. Tuan, J.Y. Chen, Z.Y. Li, B.A. Korgel, and Y.N. Xia: Mechanistic studies on the galvanic replacement reaction between multiply twinned particles of Ag and HAuCl4 in an organic medium. J. Am. Chem. Soc. 129, 1733 (2007).
B. Liu and H.C. Zeng: Fabrication of ZnO “Dandelions” via a modified Kirkendall process. J. Am. Chem. Soc. 126, 16744 (2004).
C.J. Jia, L.D. Sun, Z.G. Yan, L.P. You, F. Luo, X.D. Han, Y.C. Pang, Z. Zhang, and C.H. Yan: Single-crystalline iron oxide nanotubes. Angew. Chem. Int. Ed. 44, 4328 (2005).
X. Wang, J. Zhuang, Q. Peng, and Y.D. Li: A general strategy for nanocrystal synthesis. Nature 437, 121 (2005).
X.H. Li, D.H. Zhang, and J.S. Chen: Synthesis of amphiphilic superparamagnetic ferrite/block copolymer hollow submicrospheres. J. Am. Chem. Soc. 128, 8382 (2006).
G.F. Wang, Q. Peng, and Y.D. Li: Upconversion luminescence of monodisperse CaF2:Yb3+/Er3+ nanocrystals. J. Am. Chem. Soc. 131, 14200 (2009).
L.H. Hu, Q. Peng, and Y.D. Li: Selective synthesis of Co3O4 nanocrystal with different shape and crystal plane effect on catalytic property for methane combustion. J. Am. Chem. Soc. 130, 16136 (2008).
K.L. Ai, B.H. Zhang, and L.H. Lu: Europium-based fluorescence nanoparticle sensor for rapid and ultrasensitive detection of an anthrax biomarker. Angew. Chem. Int. Ed. 48, 304 (2009).
H. Zeng and S.H. Sun: Syntheses, properties, and potential applications of multicomponent magnetic nanoparticles. Adv. Funct. Mater. 18, 391 (2008).
C.X. Li, Z.Y. Hou, C.M. Zhang, P.P. Yang, G.G. Li, Z.H. Xu, Y. Fan, and J. Lin: Controlled synthesis of Ln3+ (Ln = Tb, Eu, Dy) and V5+ ion-doped YPO4 nano-/microstructures with tunable luminescent colors. Chem. Mater. 21, 4598 (2009).
G. Jia, H.P. You, M. Yang, L.H. Zhang, and H.J. Zhang: Uniform lanthanide orthoborates LnBO3 (Ln = Gd, Nd, Sm, Eu, Tb, and Dy) microplates: General synthesis and luminescence properties. J. Phys. Chem. C 113, 16638 (2009).
M. Yada, M. Mihara, S. Mouri, M. Kuroki, and T. Kijima: Rare earth (Er, Tm, Yb, Lu) oxide nanotubes templated by dodecylsulfate assemblies. Adv. Mater. 14, 309 (2002).
F. Cao, W.D. Shi, L.J. Zhao, S.Y. Song, J.H. Yang, Y.Q. Lei, and H.J. Zhang: Hydrothermal synthesis and high photocatalytic activity of 3D wurtzite ZnSe hierarchical nanostruetures. J. Phys. Chem. C 112, 17095 (2008).
K. Binnemans: Lanthanide-based luminescent hybrid materials. Chem. Rev. 109, 4283 (2009).
T. Nakashima and N. Kimizuka: Interfacial synthesis of hollow TiO2 microspheres in ionic liquids. J. Am. Chem. Soc. 125, 6386 (2003).
Q. Peng, Y. Dong, and Y.D. Li: ZnSe semiconductor hollow microspheres. Angew. Chem. Int. Ed. 42, 3027 (2003).
H.G. Yang and H.C. Zeng: Creation of intestine-like interior space for metal-oxide nanostructures with a quasi-reverse emulsion. Angew. Chem. Int. Ed. 43, 5206 (2004).
S.W. Kim, M. Kim, W.Y. Lee, and T. Hyeon: Fabrication of hollow palladium spheres and their successful application to the recyclable heterogeneous catalyst for suzuki coupling reactions. J. Am. Chem. Soc. 124, 7642 (2002).
X.M. Sun and Y.D. Li: Ga2O3 and GaN semiconductor hollow spheres. Angew. Chem. Int. Ed. 43, 3827 (2004).
Y. Sun and Y. Xia: Shape-controlled synthesis of gold and silver nanoparticles. Science 298, 2176 (2002).
S. Ikeda, S. Ishino, T. Harada, N. Okamoto, T. Sakata, H. Mori, S. Kuwabata, T. Torimoto, and M. Matsumura: Ligand-free platinum nanoparticles encapsulated in a hollow porous carbon shell as a highly active heterogeneous hydrogenation catalyst. Angew. Chem. Int. Ed. 45, 7063 (2006).
X.L. Xu and S.A. Asher: Synthesis and utilization of monodisperse hollow polymeric particles in photonic crystals. J. Am. Chem. Soc. 126, 7940 (2004).
X.W. Lou, Y. Wong, C. Yuan, J.Y. Lee, and L.A. Archer: Template-free synthesis of SnO2 hollow nanostructures with high lithium storage capacity. Adv. Mater. 18, 2325 (2006).
W.F. Dong, A. Kishimura, Y. Anraku, and K. Kataoka: Monodispersed polymeric nanocapsules: Spontaneous evolution and morphology transition from reducible hetero-PEG PICmicelles by controlled degradation. J. Am. Chem. Soc. 131, 3804 (2009).
D.H. Son, S. Hughes, Y.D. Yin, and A.P. Alivisatos: Cation exchange reactions in ionic nanocrystals. Science 306, 1009 (2004).
C.C. Huang, T.Y. Liu, C.H. Su, Y.W. Lo, J.R. Chen, and C.S. Yeh: Superparamagnetic hollow and paramagnetic porous Gd2O3 particles. Chem. Mater. 20, 3840 (2008).
C.M. Zhang, C.X. Li, C. Peng, R.T. Chai, S.S. Huang, D.M. Yang, Z.Y. Cheng, and J. Lin: Facile and controllable synthesis of monodisperse CaF2 and CaF2:Ce3+/Tb3+ hollow spheres as efficient luminescent materials and smart drug carriers. Chem. Eur. J. 16, 5672 (2010).
K.A. Abel, J.C. Boyer, and F.C.J.M. Van Veggel: Hard proof of the NaYF4/NaGdF4 nanocrystal core/shell structure. J. Am. Chem. Soc. 131, 14644 (2009).
H. Schafer, P. Ptacek, H. Eickmeier, and M. Haase: Synthesis of hexagonal Yb3+, Er3+-doped NaYF4 nanocrystals at low temperature. Adv. Funct. Mater. 19, 3091 (2009).
Y.P. Li, J.H. Zhang, X. Zhang, Y.S. Luo, X.G. Ren, H.F. Zhao, X.J. Wang, L.D. Sun, and C.H. Yan: Near-infrared to visible upconversion in Er3+ and Yb3+ codoped Lu2O3 nanocrystals: Enhanced red color upconversion and three-photon process in green color upconversion. J. Phys. Chem. C 113, 4413 (2009).
F. Zhang and D.Y. Zhao: Synthesis of uniform rare earth fluoride (NaMF4) nanotubes by in situ ion exchange from their hydroxide [M(OH)3] parents. ACS Nano 3, 159 (2009).
Y.P. Li, J.H. Zhang, X. Zhang, Y.S. Luo, S.Z. Lu, Z.D. Hao, and X.J. Wang: Spectral probing of surface luminescence of cubic Lu2O3:Eu3+ nanocrystals synthesized by hydrothermal approach. J. Phys. Chem. C 113, 17705 (2009).
Y.P. Du, Y.W. Zhang, Z.G. Yan, L.D. Sun, and C.H. Yan: Highly luminescent self-organized sub-2-nm EuOF nanowires. J. Am. Chem. Soc. 131, 16364 (2009).
S. Fujihara, S. Koji, and T. Kimura: Structure and optical properties of (Gd, Eu)F3-nanocrystallized sol–gel silica films. J. Mater. Chem. 14, 1331 (2004).
S. Fujihara and K. Tokumo: Chemical processing for inorganic fluoride and oxyfluoride materials having optical functions. J. Fluorine Chem. 130, 1106 (2009).
E. Antic-Fidancev, J. Holsa, J.C. Krupa, and M. Lastusarri: Crystal fields in ROF: Tb3+ (R = La, Gd). J. Alloys Compd. 380, 303 (2004).
F. Zhang, Y.F. Shi, X.H. Sun, D.Y. Zhao, and G.D. Stucky: Formation of hollow upconversion rare-earth fluoride nanospheres: Nanoscale Kirkendall effect during ion exchange. Chem. Mater. 21, 5237 (2009).
Q. Wu, Y. Chen, P. Xiao, F. Zhang, X.Z. Wang, and Z. Hu: Hydrothermal synthesis of cerium fluoride hollow nanostructures in a controlled growth microenvironment. J. Phys. Chem. C 112, 9604 (2008).
Z.J. Yang, D.Q. Han, D.L. Ma, H. Liang, L. Liu, and Y.Z. Yang: Fabrication of monodisperse CeO2 hollow spheres assembled by nano-octahedra. Cryst. Growth Des. 10, 291 (2010).
G. Jia, H.P. You, K. Liu, Y.H. Zheng, N. Guo, and H.J. Zhang: Highly uniform Gd2O3 hollow microspheres: Template-directed synthesis and luminescence properties. Langmuir 26, 5122 (2010).
L.H. Zhang, G. Jia, H.P. You, K. Liu, M. Yang, Y.H. Song, Y.H. Zheng, Y.H. Huang, N. Guo, and H.J. Zhang: Sacrificial template method for fabrication of submicrometer-sized YPO4:Eu3+ hierarchical hollow spheres. Inorg. Chem. 49, 3305 (2010).
F. Wang and X.G. Liu: Upconversion multicolor fine-tuning: Visible to near-infrared emission from lanthanide-doped NaYF4 nanoparticles. J. Am. Chem. Soc. 130, 5642 (2008).
F. Caruso, R.A. Caruso, and H. Mohwald: Nanoengineering of inorganic and hybrid hollow spheres by colloidal templating. Science 282, 1111 (1998).
W.F. Dong, J.K. Ferri, T. Adalseinsson, M. Schonhoff, G.B. Sukhorukov, and H. Mohwald: Influence of shell structure on stability, integrity, and mesh size of polyelectrolyte capsules: Mechanism and strategy for improved preparation. Chem. Mater. 17, 2603 (2005).
Y.S. Li, J.L. Shi, Z.L. Hua, H.R. Chen, M.L. Ruan, and D.S. Yan: Hollow spheres of mesoporous alumisilicates with a three dimensional pore network and extraordinary hydrothermal stability. Nano Lett. 3, 609 (2003).
T. He, D.R. Chen, X.L. Jiao, Y.Y. Xu, and Y.X. Gu: Surfactant-assisted solvothermal synthesis of Co3O4 hollow spheres with oriented-aggregation nanostructures and tunable particle size. Langmuir 20, 8404 (2004).
C.Z. Wu, Y. Xie, L.Y. Lei, S.Q. Hu, and C.Z. OuYang: Synthesis of new-phased VOOH hollow “dandelions” and their application in lithium-ion batteries. Adv. Mater.. 18, 1727 (2006).
R.L. Penn and J.F. Banfield: Oriented attachment and growth, twinning, polytypism, and formation of metastable phases: Insights from nanocrystalline TiO2. Am. Mineral. 83, 1077 (1998).
R.L. Penn and J.F. Banfield: Imperfect oriented attachment: Dislocation generation in defect-free nanocrystals. Science 281, 969 (1998).
R.L. Penn: Kinetics of oriented aggregation. J. Phys. Chem. B 108, 12707 (2004).
B. Liu and H.C. Zeng: Mesoscale organization of CuO nanoribbons: Formation of “dandelions”. J. Am. Chem. Soc. 126, 8124 (2004).
W. Ostwald: On the assumed isomerism of red and yellow mercury oxide and the surface tension of solid bodies. Z. Phys. Chem. Stoechiom. Verwandtschafts. 34, 495 (1900).
Y. Chang, J.J. Teo, and H.C. Zeng: Formation of colloidal CuO nanocrystallites and their spherical aggregation and reductive transformation to hollow Cu2O nanospheres. Langmuir 21, 1074 (2005).
H.Y. Yu, J.G. Yu, S.W. Liu, and S. Mann: Template-free hydrothermal synthesis of CuO/Cu2O composite hollow microspheres. Chem. Mater. 19, 4327 (2007).
E. Kirkendall, L. Thomassen, and C. Upthegrove: Rates of diffusion of copper and zinc in alpha brass. Trans. AIME 133, 186 (1939).
E.O. Kirkendall: Diffusion of zinc in alpha brass. Trans. AIME 147, 104 (1942).
A.D. Smigelskas and E.O. Kirkendall: Zinc diffusion in alpha brass. Trans. AIME 171, 130 (1947).
R.K. Chiang and R.T. Chiang: Formation of hollow Ni2P nanoparticles based on the nanoscale Kirkendall effect. Inorg. Chem. 46, 369 (2007).
Y. Wang, X. Bai, T. Liu, B. Dong, L. Xu, Q. Liu, and H.W. Song: Solvothermal synthesis and luminescence properties of monodisperse Gd2O3:Eu3+ and Gd2O3:Eu3+@SiO2 nanospheres. J. Solid State Chem. 183, 2779 (2010).
M. Pang, Q. Wang, and H.C. Zeng: Self-generated etchant for synthetic sculpturing of Cu2O-Au, Cu2O@Au, Au/Cu2O, and 3D-Au nanostructures. Chem. Eur. J. 18, 14605 (2012).
R.M. Petoral, F. Soderlind, A. Klasson, A. Suska, M.A. Fortin, N. Abrikossova, L. Selegard, P.O. Kall, M. Engstrom, and K. Uvdal: Synthesis and characterization of Tb3+-doped Gd2O3 nanocrystals: A bifunctional material with fluorescent labeling and MRI contrast agent properties. J. Phys. Chem. C 113, 6913 (2009).
A.P. Milanov, T. Toader, H. Parala, D. Barreca, A. Gasparotto, C. Bock, H.W. Becker, D.K. Ngwashi, R. Cross, S. Paul, U. Kunze, R. Fishcher, and A. Devi: Lanthanide oxide thin films by metalorganic chemical vapor deposition employing volatile guanidinate precursors. Chem. Mater. 21, 5443 (2009).
F. Vetrone, J.C. Boyer, A.A. Capobianco, A. Speghini, and M. Bettinelli: Concentration-dependent near-infrared to visible upconversion in nanocrystalline and bulk Y2O3: Er3+. Chem. Mater. 15, 2737 (2003).
F. Pandozzi, F. Vetrone, J.C. Boyer, R. Naccache, J.A. Capobianco, A. Speghini, and M. Bettinelli: A spectroscopic analysis of blue and ultraviolet upconverted emissions from Gd3Ga5O12:Tm3+, Yb3+ nanocrystals. J. Phys. Chem. B 109, 17400 (2005).
X. Bai, H.W. Song, G.H. Pan, Y.Q. Lei, T. Wang, X.G. Ren, S.Z. Lu, B. Dong, Q.L. Dai, and L.B. Fan: Size-dependent upconversion luminescence in Er3+/Yb3+-codoped nanocrystalline yttria: Saturation and thermal effects. J. Phys. Chem. C 111, 13611 (2007).
Acknowledgments
The authors are thankful to the National Natural Science Foundation of China (Grant Nos. 20971051 and 10704073). We are very grateful to Prof. Jihong Yu for her helpful discussion and structural characterization. We are also grateful to Prof. Junqi Sun for his helpful discussion.
Author information
Authors and Affiliations
Corresponding author
Supplementary Material
Supplementary Material
Supplementary material can be viewed in this issue of the Journal of Materials Research by visiting http://journals.cambridge.org/jmr.
Rights and permissions
About this article
Cite this article
Wang, Y., Liu, T., Wang, X. et al. Controlled synthesis of ytterbium ion and erbium ion codoped gadolinium oxyfluoride hollow nanosphere with upconversion luminescence property. Journal of Materials Research 28, 848–855 (2013). https://doi.org/10.1557/jmr.2013.12
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2013.12