Abstract
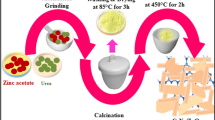
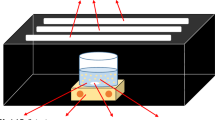
A simple and convenient method for preparing visible light response photocatalyst polypyrrole/TiO2 (PPy/TiO2) nanocomposite was developed. The products were characterized by x-ray diffraction, transmission electron microscopy, atomic force microscopy, ultraviolet-visible, and Fourier transform infrared techniques. The results indicated that the nanohybrid was composed of anatase TiO2 and PPy and exhibited an enhanced visible light-capturing ability. Average diameters of TiO2 and PPy/TiO2 were 18 and 35 nm, respectively. The photocatalytic activity of the nanocomposite was evaluated by the degradation of methyl orange under visible light irradiation. In the presence of PPy/TiO2 nanocomposite, the degradation efficiency of methyl orange of 95.54% could be obtained under visible light irradiation within 120 min. The apparent rate constant was 2.19 × 10−2, which was better than that Degussa P25 nano-TiO2. The sensitization mechanism of PPy/TiO2 photocatalyst was discussed briefly.
Similar content being viewed by others
References
N. Phonthammachai, J. Kim, and T.J. White: Synthesis and performance of a photocatalytic titania-hydroxyapatite composite. J. Mater. Res. 23, 2398 (2008).
J.G. Yu: TiO2 thin film photocatalyst. Rare Met. 23, 289 (2004).
X.L. Yan, J. He, D.G. Evans, X. Duan, and Y.X. Zhu: Preparation, characterization and photocatalytic activity of Si-doped and rare earth-doped TiO2 from mesoporous precursors. Appl. Catal. B. 55, 243 (2005).
O. Yavuz, M.K. Ram, M. Aldissi, P. Poddar, and H. Srikanth: Polypyrrole composites for shielding applications. Synth. Met. 151, 211 (2005).
A.H. Chen, H.Q. Wang, B. Zhao, and X.Y. Li: The preparation of polypyrrole–Fe3O4 nanocomposites by the use of common ion effect. Synth. Met. 139, 411 (2003).
J. Wang and X.Y. Ni: Photoresponsive polypyrrole-TiO2 nanoparticles film fabricated by a novel surface initiated polymerization. Solid State Commun. 146, 239 (2008).
Q.Z. Yan, X.T. Su, Y.P. Zhou, and C.C. Ge: Influence of cerium ions on the anatase-rutile phase transition of TiO2 prepared by sol-gel auto-igniting synthesis. Rare Met. 24, 125 (2005).
A. Sclafani and J.M. Herrmann: Comparison of the photelectronic and photocatalytic activities of various anantase and rutile forms of titania in pure liquid organic phase and in aqueous solution. J. Phys. Chem. 100, 13655 (1996).
J.A. Langford and A.J.C. Wilson: Scherrer after sixty years: A survey and some new results in the determination of crystallite size. J. Appl. Crystallogr. 11, 102 (1978).
H.P. Klong and L.E. Alexander: X-ray Diffraction Procedures for Crystalline and Amorphous Solids (Wiley Press, New York, 1954), p. 491.
J.Y. Ouyang and Y.F. Li: Great improvement of polypyrrole films prepared electrochemically from aqueous solutions by adding nonaphenol polyethyleneoxy (10) ether. Polymer (Guildf.) 38, 3997 (1997).
H.L. Wang and J.E. Fernandez: Blends of polypyrrole and poly (vinyl alcohol). Macromolecules 26, 3336 (1993).
A. Fujishima, T.N. Rao, and D.A. Tryk: Titanium dioxide photocatalysis. J. Photochem. Photobiol., C 1, 1 (2000).
M.E. Vaschetto, A.P. Monkman, and M. Springborg: First-principles studies of some conducting polymers: PPP, PPy, PPV, PPyV, and Pani. J. Mol. Struct. Theochem 468, 181 (1999).
K. Huang, M.X. Wan, Y.Z. Long, Z.J. Chen, and Y. Wei: Multifunctional polypyrrole nanofibers via a functional dopant-introduced process. Synth. Met. 155, 495 (2005).
G.B. Street and T.A. Skotheim: Handbook of Conducting Polymers: Polypyrrole from Powders to Plastics (Marcel Dekker Inc., New York, 1986), pp. 265–292.
W.J. Bae, K.H. Kim, W.H. Jo, and Y.H. Park: A water-soluble and self-doped conducting polypyrrole graft copolymer. Macromolecules 38, 1044 (2005).
R. Kostić, D. Raković, S.A. Stepanyan, I.E. Davidova, and L.A. Gribov: Vibrational spectroscopy of polypyrrole, theoretical study. J. Chem. Phys. 102, 3104 (1995).
R.L. Qiu, D.D. Zhang, Y.Q. Mo, L. Song, E. Brewer, X.F. Huang, and Y. Xiong: Photocatalytic activity of polymer-modified ZnO under visible light irradiation. J. Hazard. Mater. 156, 80 (2008).
J.G. Yu and X.J. Zhao: Effect of surface microstructure of porous TiO2 thin films on photocatalytic decolorization of methyl orange. Chin. J. Catal. 21, 213 (2000).
R.S. Sonawane, B.B. Kale, and M.K. Dongare: Preparation and photo-catalytic activity of Fe-TiO2 thin films prepared by sol–gel dip coating. Mater. Chem. Phys. 85, 52 (2004).
A. Piscopo, D. Robert, and J.V. Weber: Comparison between the reactivity of commercial and synthetic TiO2 photocatalysts. J. Photochem. Photobiol. A. 139, 153 (2001).
H. ěšMt’ánková, G. Mailhot, J. Jirkovský, J. Krýsa, and M. Bolte: Mechanistic approach of the combined (iron–TiO2) photocatalytic system for the degradation of pollutants in aqueous solution: An attempt of rationalization. Appl. Catal. B. 57, 257 (2005).
M.K. Nazeeruddin, A. Kay, I. Rodicio, R.H. Baker, E. Muller, P. Liska, N. Vlachopoulos, and M. Grätzel: Conversion of light to electricity by cis-X2 bis(2,2-bipyridyl-4,4-dicarboxylate) ruthenium(II) charge-transfer sensitizers (X = Cl-, Br-, I-, CN- and SCN-) on nanocrystalline TiO2 electrodes. J. Am. Chem. Soc. 115, 6382 (1993).
Y.F. Li and R.Y. Qian: On the nature of redox processes in the cyclic voltammetry of polypyrrole nitrate in aqueous solutions. J. Electroanal. Chem. 362, 267 (1993).
D.R. Park, J.L. Zhang, K. Ikeue, H. Yamashita, and M. Anpo: Photocatalytic oxidation of ethylene to CO2 and H2O on ultrafine powdered TiO2 photocatalysts in the presence of O2 and H2O. J. Catal. 185, 114 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, S., Chen, M., He, L. et al. Preparation and characterization of polypyrrole/TiO2 nanocomposite and its photocatalytic activity under visible light irradiation. Journal of Materials Research 24, 2547–2554 (2009). https://doi.org/10.1557/jmr.2009.0316
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2009.0316