Abstract
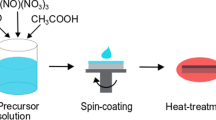
A new soluble synthetic route was developed to fabricate thin films of layered structure transition metal dichalcogendies, MoS2 and WS2. High-quality thin films of the dichalcogenides were prepared using new soluble precursors, (CH3NH3)2MS4 (M = Mo, W). The precursors were dissolved in organic solvents and spun onto substrates via both single- and multistep spin coating procedures. The thin films were formed by the thermal decomposition of the coatings under inert atmosphere. Structural, electrical, optical absorption, thermal, and transport properties of the thin films were characterized. Surface morphology of the films was analyzed by atomic force microscopy and scanning electron microscopy. Highly conductive and textured n-type MoS2 films were obtained. The measured room temperature conductivity ≈50 Ω−1 cm−1 is substantially higher than the previously reported values. The n-type WS2 films were prepared for the first time using solution-processed deposition. WS2 displays a conductivity of ≈6.7 Ω−1 cm−1 at room temperature.
Similar content being viewed by others
References
B.A. Ridley, B. Nivi, and J.M. Jacobson: All-inorganic field effect transistors fabricated by printing. Science 286, 746 (1999).
D.B. Mitzi, M. Copel, and S.J. Chey: Low-voltage transistor employing a high-mobility spin-coated chalcogenide semiconductor. Adv. Mater. 17, 1285 (2005).
C.R. Kagan, D.B. Mitzi, and C.D. Dimitrakopoulos: Organic-inorganic hybrid materials as semiconducting channels in thin-film field-effect transistors. Science 286, 945 (1999).
D.B. Mitzi, L.L. Kosbar, C.E. Murray, M. Copel, and A. Afzall: High-mobility ultrathin semiconducting films prepared by spin coating. Nature 428, 299 (2004).
L. Rapoport, Y. Bilik, Y. Feldman, M. Homyonfer, S.R. Cohen, and R. Tenne: Hollow nanoparticles of WS2 as potential solid-state lubricants. Nature 387, 791 (1997).
B. Hinnemann, P.G. Moses, J. Bonde, K.P. Jørgensen, J.H. Nielsen, S. Horch, I. Chorkendorff, and J.K. Nørskov: Biomimetic hydrogen evolution: MoS2 nanoparticles as catalyst for hydrogen evolution. J. Am. Chem. Soc. 127, 5308 (2005).
R.A. Simon, A.J. Ricco, J. Harrison, and M.S. Wrighton: Improvement of the photoelectrochemical oxidation of halides by platinization of metal dichalcogenide photoanodes. J. Phys. Chem. 87, 4446 (1983).
J. Chen, S.L. Li, F. Gao, and Z.L. Tao: Synthesis and characterization of WS2 nanotubes. Chem. Mater. 15, 1012 (2003).
P. Afanasiev and I. Bezverkhy: Synthesis of MoSx (5 < x < 6) amorphous sulfides and their use for preparation of MoS2 monodispersed microspheres. Chem. Mater. 14, 2826 (2002).
C.M. Zelenski and P.K. Dorhout: Template synthesis of near-monodisperse microscale nanofibers and nanotubules of MoS2. J. Am. Chem. Soc. 120, 734 (1998).
A. Jäger-Waldau, M.Ch. Lux-Steiner, G. Jäger-Waldau, and E. Bucher: WS2 thin films prepared by sulphurization. Appl. Surf. Sci. 71, 731 (1993).
J. Jebarj Devadasan, C. Sanjeeviraja, and M. Jayachandran: Electrodeposition ofp -WS2 thin film and characterization. J. Cryst. Growth 226, 67 (2001).
C. Ballif, M. Regula, F. Lévy, F. Burmeister, C. Schäfle, T. Matthes, P. Leiderer, P. Niedermann, W. Gutmannsbauer, and R. Bucher: Submicron contacts for electrical characterization of semiconducting WS2 thin films. J. Vac. Sci. Technol., A 16, 1239 (1998).
S.J. Li, J.C. Bernède, J. Pouzet, and M. Jamali: WS2 thin films prepared by solid state reaction (induced by annealing) between the constituents in thin film form. J. Phys. Condens. Matter 8, 2291 (1996).
K.C. Mandal and A. Mondal: Chemically deposited semiconducting molybdenum sulfide thin films. J. Solid State Chem. 85, 176 (1990).
D. Tonti, F. Varsano, F. Decker, C. Ballif, M. Regula, and M. Remškar: Preparation and photoelectrochemistry of semiconducting WS2 thin films. J. Phys. Chem. B 101, 2485 (1997).
J. Pütz and M.A. Aegerter: Spin deposition of MoSx thin films. Thin Solid Films 351, 119 (1999).
W.M. Wendlandt and H.G. Hecht: Reflectance Spectroscopy (Wiley Interscience, New York, 1966).
ICDD Card Nos. 37-1492 and 08-0237 International Center for Diffraction Data: Newton Square, PA, 1996.
E. Bucher and A. Aruchamy, Photoelectrochemistry and Photovoltaics of Layered Semiconductors (Kluwer Academic Publishers, Dordrecht, NL, 1992), p. 36.
J.C. Bernède, J. Pouzet, E. Gourmelon, and H. Hadouda: Recent studies on photoconductive thin films of binary compounds. Synth. Met. 99, 45 (1999).
J. Cheon, J.E. Gozum, and G.S. Girolami: Chemical vapor deposition of MoS2 and TiS2 films from the metal-organic precursors Mo(S-t-Bu)4 and Ti(S-t-Bu)4. Chem. Mater. 9, 1847 (1997).
N. Barreau and J.C. Bernède: MoS2 textured films grown on glass substrates through sodium sulfide based compounds. J. Phys. D Appl. Phys. 35, 1197 (2002).
J.A. Wilson and A.D. Yoffe: The transition metal dichalcogenides discussion and interpretation of the observed optical, electrical and structural properties. Adv. Phys. 18, 193 (1969).
G. Alonso, V. Petranovskii, M. Del Valle, J. Cruz-Reyes, A. Licea-Claverie, and S. Fuentes: Preparation of WS2 catalysts by in situ decomposition of tetraalkylammonium thiotungstates. Appl. Catal. G E N 197, 87 (2000).
S.M. Sze: Semiconductor Devices: Physics and Technology2nd ed. (John Wiley & Sons, New York, 1985).
D. Mergel and Z. Qiao: Correlation of lattice distortion with optical and electrical properties of In2O3:Sn films. J. Appl. Phys. 95, 5608 (2004).
J. Ouerfelli, J.C. Bernède, A. Khelil, and J. Pouzet: Photoconductive WS2 thin films obtained from multilayer Ni/W/S … W/S structure. Appl. Surf. Sci. 120, 1 (1997).
R.I. Walton and S.J. Hibble: A combined in situ x-ray absorption spectroscopy and x-ray diffraction study of the thermal decomposition of ammonium tetrathiotungstate. J. Mater. Chem. 9, 1347 (1999).
K. Ellmer, C. Stock, K. Diesner, and I. Sieber: Deposition of c¬-oriented tungsten disulfide (WS2) films by reactive DC magnetron sputtering from a W-target in Ar/H2S. J. Cryst. Growth 182, 389 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ki, W., Huang, X., Li, J. et al. Highly conductive group VI transition metal dichalcogenide films by solution-processed deposition. Journal of Materials Research 22, 1390–1395 (2007). https://doi.org/10.1557/jmr.2007.0179
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2007.0179