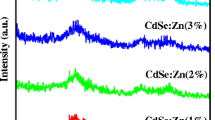
Abstract
For CdSe quantum dots (QDs) produced via high temperature pyrolysis in trioctylphosphine oxide (TOPO), a MnSe precursor such as Mn2(μ-SeMe)2(CO)8appears to be necessary to successfully incorporate low levels of Mn. A simple etching experiment and electron paramagnetic resonance (EPR) measurements reveal that most of the dopant atoms reside in the surface layers of the inorganic lattice. The dopant dramatically affects 113Cd solid state NMR spectra; the observed paramagnetic shift and decreased longitudinal relaxation time reproduce bulk material behavior. Paramagnetic atoms in QDs generate large effective magnetic fields, which implies that magneto-optical experiments can be performed simply by doping. Results from fluorescence line narrowing (FLN) studies on Mn doped CdSe QDs mirror previous findings on undoped QDs in an external magnetic field. Experimental fitting of photoluminescence excitation (PLE) spectra of doped QDs reveals that the effective absorption lineshape contains a new feature which is believed to be a previously unobserved - but theoretically predicted - optically dark fine structure state.
Similar content being viewed by others
References
Furdyna, J. K. J. Appl. Phys. 1988, 64, R29–R64.
Alivisatos, A. P. J. Phys. Chem. 1996, 100, 13226–13239.
Wang, Y.; Herron, N.; Moller, K.; Bein, T.Solid State Commun. 1991, 77, 33–38.
(b) Dhingra, S.; Kim, K.-W.; Kanatzidis, M. G. Mat. Res. Soc. Symp. Proc. 1991, 204, 163–168.
Kim, K.-W.; Cowen, J. A.; Dhingra, S.; Kanatzidis, M. G. Mat. Res. Soc. Symp. Proc. 1992, 272, 27–33.
Yanata, K.; Suzuki, K.; Oka, Y. J. Appl. Phys. 1993, 73, 4595–4598.
Bhargava, R. N.; Gallagher, D.; Hong, X.; Nurmikko, A. Phys. Rev. Lett. 1994, 72, 416–419.
Yanata, K.; Oka, Y. Jpn. J. Appl. Phys. 1995, Suppl 34-1, 164–166.
Bandaranayake, R. J.; Smith, M.; Lin, J. Y.; Jiang, H. X.; Sorensen, C. M. IEEE Trans. Magnetics 1994, 30, 4930–4932.
Sooklal, K.; Cullum, B. S.; Angel, S. M.; Murphy, C. J. J. Phys. Chem. 1996, 100, 4551–4555.
Levy, L.; Hochepied, J. F.; Pileni, M. P. J. Phys. Chem. 1996, 100, 18322–18326.
Counio, G.; Esnouf, S.; Gacoin, T.; Boilot, J.-P. J. Phys. Chem. 1996, 100, 20021–20026.
Levy, L.; Feltin, N.; Ingert, D.; Pileni, M. P. J. Phys. Chem. B 1997, 101, 9153–9160.
Ladizhansky, V.; Hodes, G.; Vega, S. J. Phys. Chem. B 1998, 102, 8505–8509.
Coleman, A. P.; Dickson, R. S.; Deacon, G. B.; Fallon, G. D.; Ke, M.; McGregor, K.; West, B. O. Polyhedron 1994, 13, 1277–1290.
Welcman, N.; Rot, I. J. Chem. Soc. 1965, 7515–7516.
Murray, C. B.; Norris, D. J.; Bawendi, M. G. J. Am. Chem. Soc. 1993, 115, 8706–8715.
Kuno, M.; Lee, J. K.; Dabbousi, B. O.; Mikulec, F. V.; Bawendi, M. G. J. Chem. Phys. 1997, 106, 9869–9882.
Ludwig, G. W.; Woodbury, H. H. in Solid State Physics, vol. 13, edited by Seitz, F.; Turnbull, D.; Academic Press: New York, 1962; p 297.
Kennedy, T. A.; Glasser, E. R.; Klein, P. B.; Bhargava, R. N. Phys. Rev. B 1995, 52, R14356-R14359.
Gavish, M.; Vega, S.; Zamir, D.Phys. Rev. B 1993, 48, 2191–2199.
Nirmal, M.; Norris, D. J.; Kuno, M.; Bawendi, M. G.; Efros, A. L.; Rosen, M.Phys. Rev. Lett. 1995, 75, 3728–3731.
Norris, D. J.; Efros, A. L.; Rosen, M.; Bawendi, M. G.Phys. Rev. B 1996, 53, 16347–16354.
Kuno, M. Ph.D. Thesis, Massachusetts Institute of Technology, 1998, pp 229–246.
Nirmal, M.; Dabbousi, B. O.; Bawendi, M. G.; Macklin, J. J.; Trautman, J. K.; Harris, T. D.; Brus, L. E. Nature 1996, 383, 802–804.
Empedocles, S. A.; Norris, D. J.; Bawendi, M. G. Phys. Rev. Lett. 1996, 77, 3873–3876.
Efros, A. L.; Rosen, M.; Kuno, M.; Nirmal, M.; Norris, D. J.; Bawendi, M. Phys. Rev. B 1996, 54, 4843–4856.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mikulec, F.V., Kuno, M., Bennati, M. et al. Organometallic Synthesis and Spectroscopic Characterization of Manganese Doped CdSe Nanocrystals. MRS Online Proceedings Library 582, 24 (1999). https://doi.org/10.1557/PROC-582-H2.4
Published:
DOI: https://doi.org/10.1557/PROC-582-H2.4