Abstract
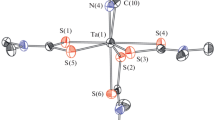
The reactions of (Me3Si)3As with group III halides have been utilized to prepare AlAs, GaAs and InAs. The adduct (Me3Si)3AsAlCl3 has been isolated as an intermediate in the formation of AlAs from (Me3Si)3As and AICI3. The crystal structure of its toluene solvate has been determined.
Similar content being viewed by others
References
G.B. Stringfellow, J. Electron. Mater. 17, 327 (1988).
C.G. Pit, A.P. Purdy, K.T. Higa, and R.L. Wells, Organometallics 5, 1266 (1986); A.P. Purdy, R.L. Wells, A.T. McPhail, and C.G. Pitt, ibid. 6, 2099 (1987); R.L. Wells, S. Shafieezad, A.T. McPhail, and C.G. Pitt, J. Chem. Soc., Chem. Commun. 1987, 1823; R.L. Wells, A.P. Purdy, A.T. McPhail, and C.G. Pitt, J. Organomet. Chem. 354, 287 (1988).
The reactions of (Me3Si)3As with GaCl3, GaBr3 and InCl3 are described in more detail in the following: R.L. Wells, C.G. Pitt, A.T. McPhail, A.P. Purdy, S. Shafieezad, and R.B. Hallock, Chemistry of Materials (in press).
All manipulations were carried out on a vacuum line or in a nitrogen-filled glovebox. The quanitities of Me3SiCl or Me3SiBr formed in the reactions were determined as HCl or HBr by vacuum distillation of all volatiles from the reaction mixture followed by hydrolysis and titration with standard NaOH solution.
V.G. Becker, G. Gutekunst, and H.J. Wessely, Z. Anorg. Allg. Chem. 462, 113(1980).
Crystallographic calculations were performed on PDP11/44 and Micro VAX computers by use of the Enraf-Nonius Structure Determination Package incorporating the direct methods program MULTAN11/82.
R=Σ | |Fo| – |Fc| | / Σ |Fo|; Research-articlew =[Σw(|Fo| – |Fc|)2/Σw(|Fo|)2]1/2; w =1/σ 2(|Fo|); Σw(|Fo- |Fc |)2 minimized.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wells, R.L., Pitt, C.G., Mcphail, A.T. et al. The Use of Tris(Trimethylsilyl)Arsine to Prepare AlAs, GaAs and InAs. The X-Ray Crystal Structure of (Me3Si)3AsAlCl3.C7H8.. MRS Online Proceedings Library 131, 45 (1988). https://doi.org/10.1557/PROC-131-45
Published:
DOI: https://doi.org/10.1557/PROC-131-45