Abstract
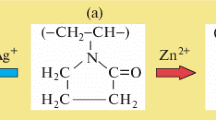
Polyvinylpyrrolidone (PVP) reduction effect on free and complex Ag+ and Au3+ ions was studied from optical measurements by adding a metal precursor (K-30), commonly used as a stabilizer, to PVP. It was found that PVP has a strong reduction effect on free ionic metal, such as Ag+ ion in AgNO3, but much weaker on complex ionic metals, AuCl4- in HAuCl4 and Ag(NH3)2+ in Ag(NH3)2OH. This is explained based on the coordinative field of polar group in PVP molecules.
Similar content being viewed by others
References
M. Faraday: Experimental relations of gold (and other metals) to light, Philos. Trans. R. Soc. London 147, 145 (1857).
M. Giersig and P. Mulvaney: Preparation of ordered colloid monolayers by electrophoretic deposition, Langmuir 9, 3408 (1993).
M. Brust, M. Walker, D. Bethell, D.J. Schiffrin and R. Whyman: Synthesis of thiol derivatised gold nanoparticles in a two phase liquid/liquid system, J. Chem. Soc. 801 (1994).
M.P. Pileni: Nanocrystal self-assemblies: fabrication and collective properties, J. Phys. Chem. B 105, 3358 (2001).
E. Hao and T. Lian: Buildup of polymer/Au nanoparticles multilayer thin films based on hydrogen bonding, Chem. Mater. 12, 3392 (2000).
F.K. Liu, S.Y. Hsieh, T.C. Chu and B.T. Dai: Synthesis of nanometer-sized poly (methyl methacrylate) polymer network by gold nanoparticle template, Jpn. J. Appl. Phys. 42, 4147 (2003).
N. Toshima: Metals—reactions in homogenous solutions, in Fine Particles—Synthesis, Characterization, and Mechanisms of Growth, edited by T. Sugimoto (Institute for Advanced Materials Proc., Marcel Dekker, New York, 2000), p. 439.
Y.G. Sun, B. Gates, B. Mayers and Y.N. Xia: Crystalline Silver nanowires by soft solution processing, Nano Lett. 2, 165 (2002).
P.Y. Silvert, R.H. Urbina and K.T. Elhsissen: Preparation of colloidal silver dispersions by the polyol process. Part 2: Mechanism of particles formation, J. Mater. Chem. 7, 293 (1997).
D.G. Duff, A. Baiker and P. Edwards: New hydrosol of gold clusters. 1. Formation and particle size variation, Langmuir 9, 2301 (1993).
I.P. Santos and L.M. Marzán: Formation of PVP-protected metal nanoparticles in DMF, Langmuir 18, 2888 (2002).
Y.G. Sun, B. Mayers, T. Herricks and Y.N. Xia: Polyol synthesis of uniform silver nanowires: A plausible growth mechanism and the supporting evidence, Nano Lett. 3, 955 (2002).
G. Carotenuto, S. DeNicola and L. Nicolais: Spectroscopic study of the growth mechanism of silver microclusters, J. Nanopart. Res. 3, 469 (2001).
M.Y. Han, C.H. Quek, W. Huang, C.H. Chew and L.M. Gan: A simple and effective chemical route for the preparation of uniform nonaqueous Au colloids, Chem. Mater. 11, 1144 (1999).
U. Krebig and M. Vollmer: Theoretical considerations—single clusters: Intrinsic size effects of the optical properties, in Optical Properties of Metal Clusters, edited by U. Gonser, R.M. Osgood Jr. M.B. Panish, and H. Sakaki (Springer-Verlag, Berlin and Heidelberg, Germany, 1995), p. 83.
C.F. Bohren and D.R. Huffman: Surface modes in small particles, in Absorption and Scattering of Light by Small Particles, edited by C.F. Bohren and D.R. Huffman (J. Wiley & Sons Inc., New York, 1983), p. 373.
K. Esumi, J. Hara, N. Aihara, K. Usui and K. Torigoe: Preparation of aniosotropic gold particles using a Gemini surfactant, J. Colloid Interface Sci. 208, 578 (1998).
G.D. Zhou and L.Y. Duan: Structure and properties of coordination composites, in Structural Chemistry, edited by G.D. Zhou and L.Y. Duan (Beijing University Press, Beijing, China, 1995), p. 288.
Z.T. Zhang, B. Zhao and L.M. Hu: PVP protective mechanism of ultrafine silver powder synthesized by chemical reduction processes, J. Solid State Chem. 121, 105 (1996).
M. Gratzel and A.J. Frank: Interfacial electron-transfer reactions in colloidal semiconductor dispersions. Kinetic analysis, J. Phys. Chem. 86, 2964 (1982).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kan, C., Cai, W., Li, C. et al. Optical studies of polyvinylpyrrolidone reduction effect on free and complex metal ions. Journal of Materials Research 20, 320–324 (2005). https://doi.org/10.1557/JMR.2005.0039
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.2005.0039