Abstract
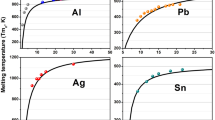
Our phenomenological model without adjustable parameters for the size dependence and dimension dependence of melting point depression and enhancement of nanocrystals is introduced. The predictions of our models are consistent with both of experimental results and other thermodynamic models for metallic nanocrystals while the difference between our model and other theoretical considerations in mesoscopic size range is discussed.
Similar content being viewed by others
References
J.R. Sambles, Proc. R. Soc. A 324, 339 (1971).
Ph. Buffat and J.P. Borel, Phys. Rev. A 13, 2287 (1976).
P.R. Couchman and W.A. Jesser, Nature 269, 481 (1977).
G.L. Allen, W.W. Gile, and W.A. Jesser, Acta. Metall. 28, 1695 (1980).
V.P. Skripov, V.P. Koverda, and V.N. Skokov, Phys. Status Solidi A 66, 109 (1981).
T. Castro, R. Reifenberger, E. Choi, and R.P. Andres, Phys. Rev. B 42, 8548 (1990).
A.N. Goldstein, C.M. Echer, and A.P. Alivistos, Science 256, 1425 (1992).
J. Eckert, J.C. Holzer, C.C. Ahn, Z. Fu, and W.L. Johnson, Nanostruct. Mater. 2, 407 (1993).
F.G. Shi, J. Mater. Res. 9, 1307 (1994).
T. Ben David, Y. Lereah, G. Deutscher, R. Kofmans, and P. Cheyssac, Philos. Mag. A 71, 1135 (1995).
S.L. Lai, J.Y. Guo, V. Petrova, G. Ramanath, and L.H. Allen, Phys. Rev. Lett. 77, 99 (1996).
G.P. Johari, Philos. Mag. A 77, 1367 (1998).
K.F. Peters, J.B. Cohen, and Y-W. Chung, Phys. Rev. B 57, 13430 (1998).
K. Morishige and K. Kawano, J. Phys. Chem. B 103, 7906 (1999).
Q. Jiang, H.X. Shi, and M. Zhao, J. Chem. Phys. 111, 2176 (1999).
Z. Zhang, J.C. Li, and Q. Jiang, J. Phys. D: Appl. Phys. 33, 2653 (2000).
Z. Wen, M. Zhao, and Q. Jiang, J. Phys. Condens. Mater. 12, 8819 (2000).
Z. Zhang, M. Zhao, and Q. Jiang, Semicond. Sci. Technol. 16, L33 (2001).
P. Pawlow, Z. Phys. Chem. 65, 545 (1909).
K.J. Hanszen, Z. Phys. 157, 523 (1960).
D.R.H. Jones . J. Mater. Sci. 9, 1 (1974).
H. Saka, Y. Nishikawa, and T. Imura, Philos. Mag. A 57, 895 (1988).
L. Graback and J. Bohr, Phys. Rev. Lett. 64, 934 (1990).
D.L. Zhang and B. Cantor, Acta Metall. Mater. 39, 1595 (1991).
R. Goswami and K. Chattopadhyay, Philos. Mag. Lett. 68, 215 (1993).
H.W. Sheng, G. Ren, L.M. Peng, Z.Q. Hu, and K. Lu, Philos. Mag. Lett. 73, 179 (1996).
K. Lu, H.W. Sheng, and Z.H. Jin, Chinese J. Mater. Res. 11, 658 (1997, in Chinese).
H.W. Sheng, G. Ren, L.M. Peng, Z.Q. Hu, and K. Lu, J. Mater. Res. 12, 119 (1997).
K. Chattopadhyay and R. Goswami, Prog. Mater. Sci. 42, 287 (1997).
L. Zhang, Z.H. Jin, L.H. Zhang, M.L. Sui, and K. Lu, Phys. Rev. Lett. 85, 1484 (2000).
Q. Jiang, Z. Zhang, and J.C. Li, Chem. Phys. Lett. 322, 549 (2000).
Q. Jiang, Z. Zhang, and J.C. Li, Acta Mater. 48, 4791 (2000).
Q. Jiang, L.H. Liang, and J.C. Li, J. Phys. Condens. Mater. 13, 565 (2001).
H. Reiss and I.B. Wilson, J. Colloid Sci. 3, 551 (1948).
Q. Jiang, H.X. Shi, and M. Zhao, Acta Mater. 47, 2109 (1999).
F.A. Lindemann, Z. Phys. 11, 609 (1910).
N.F. Mott, Proc. R. Soc. A 146, 465 (1934).
A.R. Regel’ and V.M. Glazov, Semiconductors 29, 405 (1995).
H.K. King, in Physical Metallurgy, edited by R.W. Cahn (NorthHolland, Amsterdam, The Netherlands 1970), pp. 59–63.
Table of Periodic Properties of the Elements (Sargent-Welch Scientific, Skokie, IL 1980), p. 1.
A.R. Ubbelohde, Melting and Crystal Structure (Clarendon Press, Oxford, United Kingdom, 1965), p. 171.
J.W.M. Frenken and J.F. van der Veen, Phys. Rev. Lett. 54, 134 (1985).
E.T. Chen, R.N. Barnett, and U. Landman, Phys. Rev. B 41, 439 (1990).
A. Antonelli, S.N. Khanna, and P. Jena, Phys. Rev. B 48, 8263 (1993).
C.M. Cotell and K.S. Granbowski, MRS Bull. 17, 44 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, M., Zhou, X.H. & Jiang, Q. Comparison of different models for melting point change of metallic nanocrystals. Journal of Materials Research 16, 3304–3308 (2001). https://doi.org/10.1557/JMR.2001.0454
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.2001.0454