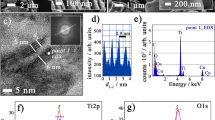
Abstract
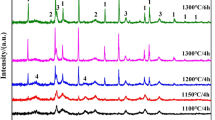
Three kinds of inorganic–organic hybrid gel sheets were prepared from the liquid mixtures of ethyl silicate and water–soluble phenol resin. The prepared transparent gel sheets were fired at various temperatures. The density of the fired sheets jumped up at 773–1023 K with disappearance of organic groups. The sheets kept the prepared shape after the 1273 K firing, and their density increased with the silica content. After the firing at 1873 K, the sheets with high carbon content (C/SiO2: 5.60, 3.52) were converted into the sheets composed of silicon carbide aggregate with excess carbon, while the sheet with low carbon content (C/SiO2: 1.43) was converted into the fragile powders.
Similar content being viewed by others
References
J.E. Mark, Heterogeneous Chem. Rev. 3, 307 (1996).
J. Premachandra, C. Kumudinie, W. Zhao, J.E. Mark, T.D. Dang, J.P. Chen, and F.E. Arnold, J. Sol-Gel Sci. Technol. 7, 163 (1996).
C.L. Jackson, B.J. Bouer, A.I. Nakatani, and J.D. Barnes, Chem. Mater. 8, 727 (1996).
G.C. Wei, C.R. Kennedy, and L.A. Harris, Ceramic Bulletin 63, 1054 (1984).
H. Tanaka and Y. Kurachi, Ceramic International 14, 109 (1988).
K. Ono and Y. Kurachi, J. Mater. Sci. 24, 388 (1991).
T. Shimoo, M. Takemura, K. Okamura, Y. Kurachi, and M. Kajiwara, J. Ceram. Soc. Jpn. 102, 880 (1994).
I. Hasegawa, T. Nakamura, S. Motojima, and M. Kajiwara, J. Sol-Gel Sci. Technol. 8, 577 (1997).
S. Ueno, K. Kameda, J. Yu, K. Hiragushi, and Y. Miura, J. Ceram. Soc. Jpn. 106, 688 (1998).
M. Narisawa, Y. Okabe, M. Iguchi, K. Okamura, and Y. Kurachi, J. Sol-Gel Sci. Technol. 12, 143 (1998).
D. Huang, Y. Ikuhara, M. Narisawa, and K. Okamura, J. Am. Ceram. Soc. 81, 3173 (1998).
M. Narisawa, Y. Okabe, K. Okamura, and Y. Kurachi, J. Ceram. Soc. Jpn. 107, 285 (1999).
A. Morikawa, Y. Iyoku, M. Kakimoto, and Y. Imai, Polym. J. 24, 107 (1992).
K. Haraguchi and Y. Usami, Chem. Lett. 51 (1997).
K. Haraguchi, Y. Usami, and Y. Ono, J. Mater. Sci. 33, 3337 (1998).
E. Fitzer, W. Schafer, and S. Yamada, Carbon 7, 643 (1969).
E. Fitzer and W. Schafer, Carbon 8, 353 (1970).
F.H.V. Cauwelaert, P.A. Jacobs, and J.B. Uytterhoeven, J. Phys. Chem. 76, 1434 (1972).
B.A. Morrow and I.A. Cody, J. Phys. Chem. 77, 1465 (1973).
K.M. Davis, A. Agarwal, M. Tomozawa, and K. Hirao, J. Non-Cryst. Solids 203, 27 (1996).
K. Tanaka, K. Ohzeki, T. Yamabe, and S. Yata, Synth. Met. 9, 41 (1984).
K.A. Trick and T.E. Saliba, Carbon 33, 1509 (1995).
J.G. Lee and I.B. Cutler, Am. Ceram. Soc. Bull. 54, 195 (1975).
T. Shimoo, F. Mizutaki, S. Ando, and H. Kimura, J. Jpn. Inst. Met. 52, 279 (1988).
V.M. Kevorkijan, M. Komac, and D. Kolar, J. Mater. Sci. 27, 2705 (1992).
W.S. Seo and K. Koumoto, J. Am. Ceram. Soc. 79, 1777 (1996).
W.S. Seo, K. Koumoto, and S. Arai, J. Am. Ceram. Soc. 81, 1255 (1998).
H. Tateyama, H. Noma, Y. Adachi, and M. Komatsu, Chem. Mater. 9, 766 (1997).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Narisawa, M., Yamane, K., Okabe, Y. et al. Carbon–silica alloy material as silicon carbide precursor prepared from phenol resin and ethyl silicate. Journal of Materials Research 14, 4587–4593 (1999). https://doi.org/10.1557/JMR.1999.0621
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1999.0621