Abstract
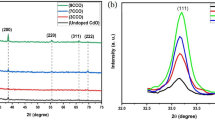
Boron-doped diamond films have been grown by the hot filament chemical vapor deposition process. The feed gas was a mixture of argon, bubbled through a solution of B2O3 in ethanol, and hydrogen. The highest growth rate was 0.7 μm/h. The boron concentration in the films depended on the concentration of B2O3 in the ethanol. The highest boron doping level, as measured by secondary ion mass spectroscopy, was 6300 atomic ppm. Raman spectroscopy and x-ray diffraction both confirmed the presence of crystalline diamond in the films. The frequency of the diamond Raman line decreased with increasing boron concentration. This shift may arise from an interaction of the charged carriers (holes) produced by the boron doping and the Raman-active optic phonon. The oxidation rates of doped and undoped films were measured by thermogravimetric analysis at 700 °C in flowing high purity oxygen. Films with a boron concentration of 6300 ppm oxidized at one-tenth the rate of undoped diamond. A layer of B2O3, detected on the surface of an oxidized B-doped film, is believed to act as a protective barrier that decreases the oxidation rate.
Similar content being viewed by others
References
The Properties of Diamond, edited by J. E. Field (Academic Press, London, 1979).
E.L. Simons and P. Cannon, Nature (London) 210, 90 (1966),
C. E. Johnson, M. A. S. Hasting, and W. A. Weimer, J. Mater. Res. 5, 2320 (1990).
M. Alam and Q. Sun, J. Mater. Res. 8, 2870 (1993).
A. Joshi, R. Nimmagadda, and J. Herrington, J. Vac. Sci. Technol. A 8, 2137 (1990).
K. Tankala, T. DebRoy, and M. Alam, J. Mater. Res. 5, 2483 (1990).
M. Alam and Q. Sun, J. Mater. Sci. Lett. 12, 1389 (1993).
J. Raun, K. Kobashi, and W.J. Choyke, Appl. Phys. Lett. 60, 1884 (1992).
V. Venkatesan, K. Das, D.L. Dreifus, G.G. Fountain, R.A. Rudder, J.B. Posthill, and R.J. Markunas, in Proc. 2nd Symp. on Diamond Materials (Proceedings Volumes 91-98), edited by A. J. Purdes, B. M. Meyerson, J. C. Angus, K. E. Spear, R. F. Davis, and M. Yoder (The Electrochemical Society, Pennington, NJ, 1991), pp. 558-565.
K. Nishimura, K. Das, and J.T. Glass, J. Appl. Phys. 69, 3142 (1991).
H. Kawarada, Y. Yokata, H. Matsuyama, T. Sogi, and A. Hiraki, in Proc. 2nd Symp. on Diamond Materials (Proceedings Volumes 91-98), edited by A.J. Purdes, B.M. Meyerson, J.C. Angus, K. E. Spear, R. F. Davis, and M. Yoder (The Electrochemical Society, Pennington, NJ, 1991), pp. 420-426.
K. Miyata, D.L. Dreifus, K. Daas, J.T. Glass, and K. Kobashi, in Proc. 2nd Symp. on Diamond Materials (Proceedings Volumes 91-98), edited by A.J. Purdes, B.M. Meyerson, J.C. Angus, K. E. Spear, R. F. Davis, and M. Yoder (The Electrochemical Society, Pennington, NJ, 1991), pp. 543-550.
S.A. Grot, C.W. Hatfield, G.Sh. Gildenblat, A.R. Badzian, T. Badzian, and R. Messier, Appl. Phys. Lett. 58, 1542 (1991).
G. Sh. Gildenblat, S. A. Grot, C. W. Hatfield, C. R. Wronski, A. R. Badzian, T. Badzian, and R. Messier, Mater. Res. Bull. 25, 129 (1990).
R. Ramesham, T. Roppel, and C. Ellis, J. Electrochem. Soc. 138, 2981 (1991).
J. Mort, D. Kuhman, M. Machonkin, M. Morgan, F. Jansen, and K. Okumura, Appl. Phys. Lett. 55, 1121 (1989).
J. Mort, M. A. Machonkin, and K. Okumura, Appl. Phys. Lett. 58, 1908 (1991).
K. Okano, H. Naruki, Y. Akiba, T. Kurosu, M. Iida, Y. Hirose, Jpn. J. Appl. Phys. 27, L173 (1988).
K. Okano, H. Naruki, Y. Akiba, T. Kurosu, M. Iida, Y. Hirose, and T. Nakamura, Jpn. J. Appl. Phys. 28, 1066 (1989).
K. Okano, H. Kiyota, T. Iwasaki, T. Kurosu, M. Iida, and T. Nakamura, Appl. Phys. Lett. 58, 840 (1991).
A. Masood, M. Aslam, M. A. Tamor, and T. J. Potter, Appl. Phys. Lett. 61, 1832 (1992).
G.G. Fountain, R.A. Rudder, D.P. Malta, S. V. Hattangady, R.G. Alley, G.C. Hudson, J.B. Posthill, R.J. Markunas, T.P. Humphreys, R.J. Nemanich, V. Venkatesan, and K. Das, in Proc. 2nd Symp. on Diamond Materials (Proceedings Volumes 91-98), edited by A.J. Purdes, B.M. Meyerson, J.C. Angus, K. E. Spear, R. F. Davis, and M. Yoder (The Electrochemical Society, Pennington, NJ, 1991), pp. 523-527.
G.S. Sandhu, M.L. Swanson, and W.K. Chu, Appl. Phys. Lett. 55, 1397 (1989).
B. V. Spitsyn and A. E. Alexenko, in Proc. 2nd Symp. on Diamond Materials (Proceedings Volumes 91-98), edited by A.J. Purdes, B.M. Meyerson, J.C. Angus, K.E. Spear, R.F. Davis, and M. Yoder (The Electrochemical Society, Pennington, NJ, 1991), pp. 597-604.
J.W. Glesner, A. A. Morrish, and K.A. Snail, J. Appl. Phys. 70, 5144 (1991).
E. N. Farabaugh, A. Feldman, and L. H. Robins, in Diamond Optics, edited by A. Feldman and S. Holley (SPIE–The International Society for Optical Engineering, Bellingham, WA, 1988), Proc. SPIE 969, pp. 24-31.
Secondary Ion Mass Spectrometry, edited by R. G. Wilson, F. A. Stevie and C. W. Magee, App. E, 3 (John Wiley, New York, 1989).
A 95% confidence interval on the boron and tungsten concentrations is estimated to be ±50% of the determined value, based on scientific judgment and experience with this method.
P.G. Lurie and J.W. Wilson, Surf. Sci. 65, 4768 (1977).
G. Hanke and K. Miiller, J. Vac. Sci. Technol A 2, 964 (1984).
D.S. Knight and W.B. White, J. Mater. Res. 4, 385 (1989).
J. Robertson, Adv. Phys. 35, 317 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Farabaugh, E.N., Robins, L., Feldman, A. et al. Growth and oxidation of boron-doped diamond films. Journal of Materials Research 10, 1448–1454 (1995). https://doi.org/10.1557/JMR.1995.1448
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1995.1448