Abstract
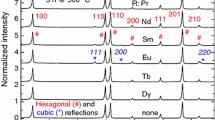
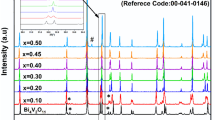
High-quality bulk REBa2Cu3O7−x (RE = Y,Eu,Gd,Nd,La) was synthesized by a solution method. Stoichiometric amounts of yttrium, barium, and copper acetates were dissolved in glacial acetic acid. The acid was boiled away leaving a glassy acetate precursor. This precursor has shown amorphous scattering in XRD, a phenomenon consistent with atomic level mixing of reactants. The glassy precursor was subsequently heat-treated with a 5 °C/min ramp to 900 °C and a 2 h soak at 900 °C in air. The final product was obtained after heat treatment under oxygen at 550 °C with slow cooling to room temperature. Final products were analyzed by XRD, SEM, and four probe de-resistivity measurements. The mechanism of both precursor and product formation was examined through substitution studies and XRD. It was found that a combination of a rare earth acetate, barium acetate, and acetic acid was necessary for the formation of an amorphous precursor.
Similar content being viewed by others
References
R. J. Cava, B. Batlogg, R. B. van Dover, D. W. Murphy, S. Sunshine, T. Siegrist, J. P. Rameika, E. A. Reitman, S. Zahurak, and G. P. Espinosa, Phys. Rev. Lett. 58, 1676 (1987).
C. Wagner, Z. Anorg. Allg. Chem. 236, 320 (1938).
W. E. Brown, D. Dollimore, and A. K. Galwey, Comprehensive Chemical Kinetics, Volume 22, Reactions in the Solid State, edited by C. H. Bamford and C. H. F. Tipper (Elsevier Scientific, New York, 1980).
X. Z. Wang, M. Henrt, J. Livage, and I. Rosenndu, Solid State Commun. 64, 881 (1987).
A. M. Kini, U. Geiser, H. I. Kao, K. D. Carlson, H. H. Wang, M. R. Monaghan, and J. M. Williams, Inorg. Chem. 26, 1836 (1987).
H. S. Horowitz, S. J. McLain, A. W. Sleight, J. D. Druliner, P. L. Gai, M. J. Van Kavelaar, J. L. Wagner, B. D. Biggs, and S. J. Poon, Science 243, 66 (1989).
D. Segal, Chemistry of Solid State Materials 1, Chemical Synthesis of Advanced Materials (Cambridge University Press, Cambridge, 1989).
G. Kordas, J. Non-Cryst. Solids 121, 436 (1990).
S. Katayama and M. Sekine, J. Mater. Res. 5, 683 (1990).
G. L. Messing, S. C. Zhang, and G. V. Jayanthi, J. Am. Ceram. Soc. 76, 2707 (1993).
M. W. Real, Proc. Brit. Ceram. Soc. 38, 59 (1986).
T. L. Ward, T. T. Kodas, A. H. Carim, D. M. Kroeger, and H. Hsu, J. Mater. Res. 7, 827 (1992).
P. J. McGrath and R. M. Laine, J. Am. Ceram. Soc. 75 (5), 1223 (1992).
N. V. Coppa, G. H. Myer, R. E. Salomon, A. Bura, J. W. O’Reilly, J. E. Crow, and P. K. Davies, J. Mater. Res. 7, 2017 (1992).
K. H. Song, H. K. Liu, S. X. Dou, and C. C. Sorrell, J. Am. Ceram. Soc. 73 (6), 1771 (1990).
J. A. Duffy and M. D. Ingram, J. Am. Ceram. Soc. 52, 224 (1969).
R. F. Bartholomew and S. S. Lewek, J. Am. Ceram. Soc. 53, 445 (1970).
J. McKittrick and R. Contreras, Thin Solid Films 206, 146 (1991).
M. L. Kullberg, M. T. Lanagan, W. Wu, and R. B. Poeppel, Supercon. Sci. Technol. 4, 337 (1991).
D. I. dos Santos, U. Balachandran, M. T. Lanagan, D. Shi, M. A. Patel, and R. B. Poeppel, Energy Res. Abstr. 15 (10), No. 23958, ANL/PPRNT-90-204; Order No. DE90008707 (1990).
D. I. dos Santos, U. Balachandran, R. A. Guttschow, and R. B. Poeppel, J. Non-Cryst. Solids 121, 448 (1990).
J. McHale, R. W. Schaeffer, and R. E. Salomon, J. Chem. Ed. 69 (12), 1031 (1992).
S. Peterson and E. K. Dienes, J. Phys. Chem. 55, 1299 (1951).
For a discussion of this topic, see Chap. 7 of J. Kleinberg, W. J. Argersinger, and E. Griswold, Inorganic Chemistry (D. C. Heath and Co., Boston, MA, 1960).
CRC Handbook of Physics and Chemistry, 62nd ed., edited by R.C. Weast (CRC Press Inc., Boca Raton, FL, 1981).
For a discussion of the acetic acid solvent system, see T. C. Waddington, Non-Aqueous Solvent Systems (Academic Press, New York, 1965); or A. K. Holiday and A. G. Massey, Inorganic Chemistry in Non-Aqueous Solvents (Pergamon Press, New York, 1965).
For information on conductivity of solutions of metal acetates in acetic acid, see I. M. Kolthoff and A. William, J. Am. Chem. Soc. 55, 239 (1934).
A. W. Davidson, J. Am. Chem. Soc. 50, 1890 (1928).
Personal communication, D. D. Titus, Department of Chemistry, Temple University, Philadelphia, PA 19122 (1993).
B. M. Nirsha, A. D. Chubinidze, Y. A. Velikodnyi, B. V. Zhadanov, and V. A. Olikova, Zh. Obschch. Khim. USSR 53, 1631 (1983).
A. Davidson and W. McAllister, J. Am. Chem. Soc. 52, 519 (1930).
A. W. Davidson and E. Griswold, J. Am. Chem. Soc. 57, 423 (1935).
E. Griswold and W. van Home, J. Am. Chem. Soc. 67, 763 (1945).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McHale, J., Myer, G.H. & Salomon, R.E. Simplified preparation of REBa2Cu3O7−x via the acetate method. Journal of Materials Research 10, 1–6 (1995). https://doi.org/10.1557/JMR.1995.0001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1995.0001