Abstract
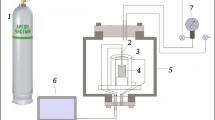
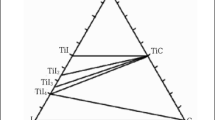
Titanium carbide may be readily produced in high purity via direct reaction between the solid phases of titanium and carbon in the process of combustion synthesis, also known as self-propagating high-temperature synthesis (SHS). The high temperatures generated by this exothermic reaction (<3000 °C) melt the titanium phase which subsequently flows to and reacts with the solid carbon. As a result, with lightly or uncompressed green bodies a product is obtained which retains most of the carbon precursor powder morphology down to the μm scale. The effect of three distinctly different carbon precursor powders on the final titanium carbide morphology has been examined. Subsequent effects of particle size and microstructure on the quality of the titanium carbide product are also noted.
Similar content being viewed by others
References
A. P. Hardt and P. V. Phung, Combust. Flame 21 (1), 77 (1973).
J. B. Holt, MRS Bulletin 12 (7), 60 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mullins, M.E., Riley, E. The effect of carbon morphology on the combustion synthesis of titanium carbide. Journal of Materials Research 4, 408–411 (1989). https://doi.org/10.1557/JMR.1989.0408
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1989.0408