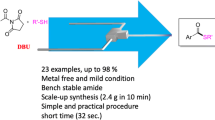
Abstract
The synthesis and utilisation of acyl azides in a flow apparatus combined with an automated extraction unit is described. This process safely provides multi-100 g quantities of a labile diacyl azide (3) as an intermediate that could not be generated safely by classic batch methods. Its subsequent conversion to the desired amine (4) represents an example for process intensification. The same set-up with an output capacity of >30 g/h was used for the unattended synthesis of benzoyl azide as the final product in solution (tert-butyl methyl ether (TBME), 0.5 M).
Article PDF
Similar content being viewed by others
References
Curtius, T. J. Prakt. Chem. 1922, 52, 225–227
reviews on azide chemistry: (b) Scriven, E. F. V.; Turnbull, K. Chem. Rev. 1988, 88, 297–369
Braese, S.; Gil, C.; Knepper, K.; Zimmermann, V. Angew. Chem. Int. Ed. 2005, 44, 5188–5242.
Staudinger, H.; Hauser, E. Helv. Chim. Acta 1921, 4, 861–886
Katritzky, A.; Khashab, N.; Bobrov, S. Helv. Chim. Acta 2005, 88, 1664–1675.
Keicher, T.; Loebbecke, S. In Organic Azides: Syntheses and Applications; Braese, S., Banert, K., Eds.; Wiley: New York, 2009.
Reviews on microreaction technology: (a) Pennemann, H.; Watts, P.; Haswell, S. J.; Hessel, V.; Loewe, H. Org. Process Res. Dev. 2004, 8, 422–439
Jaehnisch, K.; Hessel, V.; Loewe, H.; Baerns, M. Angew. Chem. Int. Ed. 2004, 43, 406–446 and literature quoted therein.
Mueller, G.; Gaupp, T.; Wahl, T.; Wille, G. Chimia 2006, 60, 618–622
Schwalbe, T.; Autze, V.; Hohmann, M.; Stirner, W.; Org. Process Res. Dev. 2004, 8, 440–454.
Membrane phase separation on acylic azides: Hemantkumar R.; Sahoo, H.; Kralj, J.; Jensen, K. F. Angew. Chem. Int. Ed. 2007, 46, 5704–5708.
Rumi, L.; Pfleger, C.; Spurr, P.; Klinkhammer, U.; Bannwarth W. Org. Process Res. Dev. 2009, 13, 747–750.
Alternative methods in flow systems: (a) Baumann, M.; Baxendale, I. R.; Ley, S. V.; Nikbin N.; Smith C. D. Org. Biomol. Chem., 2008, 6, 1587–1593
Brandt J. C.; Wirth T. Beilstein J. Org. Chem. 2009, 5(30).
Burgos, A.; Ellames, G. J. J. Labelled Comp. Radiopharm. 1998, XLI, 443–449.
The glass microreactor type S02 is available with the Sigma-Aldrich Microreaction Explorer Kit, article number 19979. Also see www.sigmaaldrich.com/mrt/mrt.
Impedance glass probe SGK 180/40 and control unit MIL 8130 were purchased from Aquasant Messtechnik AG (Bubendorf, Switzerland).
For United Nation criteria on chemical products, see http://www.unece.org/trans/danger/publi/manual/manual_e.html
Sigma-Aldrich product numbers: 4, 726184; optical pure isomers 726311 and 726338; 6, 778486. For further information and other azides, see www.sigmaaldrich.com.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sprecher, H., Payán, M.N.P., Weber, M. et al. Acyl Azide Synthesis and Curtius Rearrangements in Microstructured Flow Chemistry Systems. J Flow Chem 2, 20–23 (2012). https://doi.org/10.1556/jfchem.2011.00017
Published:
Issue Date:
DOI: https://doi.org/10.1556/jfchem.2011.00017