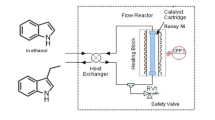
Abstract
The first continuous flow carbonylation reaction using aryl formates as CO precursor is reported. The reaction is practical, scalable and high yielding. The use of a flow protocol safely allows expanding the scope to activated chlorides, nitrogen heterocycles and to the selective introduction of an ester group in dihalo-derivatives. Further selective reduction of the ester formed to an aldehyde in flow is also described.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
(a) Brennführer, A.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2009, 48, 4144
(b) Brennführer, A.; Neumann, H.; Beller, M. ChemCatChem 2009, 1, 28
(c) Kiss, G. Chem. Rev. 2001, 101, 3435
(d) Ali, B. E.; Alper, H. Transition Met. Org. Synth.
(e) Beller, M.; Bolm, C., Ed.; Wiley-VCH: Weinheim; 1998, 49
(f) Colquhoun, H. M.; Thompson, D. J.; In Carbonylation: Direct Synthesis of Carbonyl Compounds; Twigg, M. V., Ed.; Plenum: New York, 1991; 119.
Morimoto, T.; Kakiuchi, K. Angew. Chem., Int. Ed. 2004, 43, 5580.
Wieckowska, A.; Fransson, R.; Odell, L. R.; Larhed, M. J. Org. Chem. 2011, 76, 978.
(a) Morimoto, T.; Fuji, K.; Tsutsumi, K.; Kakiuchi, K. J. Am. Chem. Soc. 2002, 124, 3806
(b) Shibata, T.; Toshida, N.; Takagi, K. Org. Lett. 2002, 4, 1619
(c) Morimoto, T.; Yamasaki, K.; Hirano, A.; Tsutsumi, K.; Kagawa, N.; Kakiuchi, K.; Harada, Y.; Fukumoto, Y.; Chatani, N.; Nishioka, T. Org. Lett. 2009, 11, 1777.
(a) Hosoi, K.; Nozaki, K.; Hiyama, T. Org. Lett. 2002, 4, 2849
(b) Wan, Y.; Alterman, M.; Larhed, M.; Hallberg, A. J. Org. Chem. 2002, 67, 6232
(c) Ko, S.; Lee, C.; Choi, M.-G.; Na, Y.; Chang, S. J. Org. Chem. 2003, 68, 1607.
Cacchi, S.; Fabrizi, G.; Goggiamani, A. J. Comb. Chem. 2004, 6, 692.
(a) Fujihara, T.; Hosoki, T.; Katafuchi, Y.; Iwai, T.; Terao, J. Tsuji, Y. Chem. Commun. 2012, 48, 8012
(b) Ueda, T.; Konishi, H.; Manabe, K. Org. Lett. 2012, 14, 3100
(c) Ueda, T.; Konishi, H.; Manabe, K. Org. Lett. 2012, 14, 5370.
(a) Ueda, T.; Konishi, H.; Manabe, K. Angew. Chem., Int. Ed. 2013, 52, 8611
(b) Ueda, T.; Konishi, H.; Manabe, K. Org. Lett. 2013, 15, 5370.
Hessel, V.; Kralisch, D.; Kockmann, N.; Nöel, T.; Wang, Q. ChemSusChem 2013, 6, 746.
(a) Microreactors: New Technology for Modern Chemistry; Ehrfield, W.; Hessel, V.; Löwe, H., Ed.; Wiley-VCH: Weinheim, 2000
(b) Wiles, C.; Watts, P. Eur. J. Org. Chem. 2008, 1655
(c) Illg, T.; Löb, P.; Hessel, V. Bioorg. Med. Chem. 2010, 18, 3707
(d) Watts, P.; Haswell, S. J. Drug Discovery Today 2003, 8, 586
(e) Microreactors in Organic Synthesis and Catalysis; Wirth T., Ed.; Wiley-VCH: Weinheim, 2008
(f) Mason, B. P.; Price, K. E.; Steinbacher, J. L.; Bogdan, A. R.; McQuade, D. T.; Chem. Rev. 2007, 107, 2300
(g) Sahoo, H. R.; Kralj, J. G.; Jensen, K. F. Angew. Chem., Int. Ed. 2007, 46, 5704
(h) Hartman, R. L.; McMullen, J. P.; Jensen, K. F. Angew. Chem., Int. Ed. 2011, 50, 7502
(i) Baxendale, I. R.; Griffiths-Jones, C. M.; Ley, S. V.; Tranmer, G. K. Synlett 2006, 427.
(a) Wiles, C.; Watts, P. Green Chem. 2012, 14, 38
(b) Ley, S. V. Chem. Rec., 2012, 12, 378.
Csajági, C.; Borcsek, B.; Niesz, K.; Kovács, I.; Székelyhidi, Z.; Bajkó, Z.; Ürge, L.; Darvas, F. Org. Lett. 2008, 10, 1589.
Koos, P.; Gross, U.; Polyzos, A.; O’Brien, M.; Baxendale, I.; Ley, S. V. Org. Biomol. Chem. 2011, 9, 6903.
Brancour, C.; Fukuyama, T.; Mukai, Y.; Skrydstrup, T.; Ryu, I. Org. Lett. 2013, 15, 2794.
For further information about SiliaCat catalysts, visit the web: http://www.silicycle.com. Recent references for the use of this catalyst in flow: (a) Munoz, J. de M.; Alcazar, J.; de la Hoz, A.; Diaz-Ortiz, A. Adv. Synth. Catal. 2012, 354, 3456
(b) Egle, B.; Muñoz, J. de M.; Alonso, N.; De Borggraeve, W.M.; de la Hoz, A.; Díaz-Ortiz, A.; Alcázar, J. J. Flow. Chem. 2014, 4, 22.
The following reference exemplifies translation of microwave conditions to flow: Damm, M.; Glasnov, T. N.; Kappe, C. O. Org. Process Res. Dev. 2010, 14, 215.
For further information about Vapourtec flow reactors, visit the web: http://www.vapourtec.co.uk.
An empty 70-mm CatCart cartridge from Thalesnano was used as filter. For further information aboutCatCart cartridges, visitthe web: http://thalesnano.com/products/CatCart-and-compatible-system.
CAUTION: Care should be taken when preparing stock solutions of trichlorophenyl formate. The quality of DMF used is important as small amounts of base can induce CO formation, even at room temperature. It is recommended to first assess the stability of the precursor in a particular batch of solvent.
Muñoz, J. de M.; Alcázar, J.; de la Hoz, A.; Díaz-Ortiz, A. Eur. J. Org. Chem. 2012, 260.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alonso, N., de Muñoz, J.M., Egle, B. et al. First Example of a Continuous-Flow Carbonylation Reaction Using Aryl Formates as CO Precursors. J Flow Chem 4, 105–109 (2014). https://doi.org/10.1556/JFC-D-14-00005
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/JFC-D-14-00005