Abstract
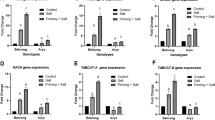
Drought is a worldwide problem, getting more serious with global climate change. Among various strategies, seed priming is the simplest approach for improving drought tolerance in crop plants. Seed priming treatments were applied by soaking seeds in aerated solution of 1% mannose (56 mM) and 10 mM mannitol for 8 h while 100 μM H2 O2 for 5 h. Seeds soaked in aerated water (hydropriming) and non-primed seed were used as controls. Drought stress significantly reduced the seedling fresh weight and leaf relative water content. Pre-sowing seed treatment with mannitol significantly increased the seedling, root and shoot fresh as well as dry weights under non-stress condition. Moreover, H2 O2 increased the root length; seedling and root dry weights while mannose increased the shoot dry weight under drought stress. Leaf relative water content (RWC) improved after mannitol and H2 O2 priming under drought and non-stressed conditions. Hydropriming increased the root and shoot fresh weights, shoot dry weight and RWC under non-stress condition while seedling, root, shoot fresh weights and shoot dry weight along with raised TSP, MDA, reducing sugars under drought stress. Drought stress raised the total soluble protein (TSP), protease, APX and POD activities, MDA and reducing sugars in leaves. Mannitol and H2 O2 confiscate the drought-induced increase in TSP while H2 O2 significantly increase it under non-stress condition. Drought stress reduced the catalase activity in leaves while H2 O2 and mannitol priming brought it back to control level. Drought stress elevated the MDA in leaves and H2 O2 treatment prevented this increase. Only mannose priming rose the reducing sugars in leaves under non-stress condition. Under drought, mannose and mannitol priming raised the reducing sugars in the leaves as a tactic for osmotic adjustment. In conclusion, seed priming treatments ameliorated the drought tolerance in wheat by elevating the level of antioxidants, reducing oxidative damage of biomolecules and accumulating more reducing sugars for osmotic adjustments.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Afzal, I., Munir, F., Ayub, C.M., Basra, S.M.A., Hameed, A., Nawaz, A. 2009. Changes in antioxidant enzymes, germination capacity and vigour of tomato seeds in response of priming with polyamines. Seed Sci. and Technol. 37:765–770.
Ahmad, I., Khaliq, T., Ahmad, A., Basra, S.M.A., Hasnain, Z., Ali, A. 2012. Effect of seed priming with ascorbic acid, salicylic acid and hydrogen peroxide on emergence, vigour and antioxidant activities of maize. African J. of Biotech. 11:1127–1137.
Ainsworth, E.A., Gillespie, K.M. 2007. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nature Protocols 2:875–877.
Bailly, C., Benamar, A., Corbineau, F., Come, D. 1998. Free radical scavenging as affected by accelerated ageing and subsequent priming in sunflower seeds. Physiologia Plantarum 104:646–652.
Bailly, C., Benamar, A., Corbineau, F., Come, D. 2000. Antioxidant systems in sunflower (Helianthus annuus L.) seeds as affected by priming. Seed Sci. Res. 10:35–42.
Basal, H., Smith, C.W., Thaxton, P.S., Hemphill, J.K. 2005. Seedling drought tolerance in upland cotton. Crop Sci. 45:766–771.
Beers, R.F., Sizer, I.W. 1952. A spectrophotometeric method for measuring the breakdown of hydrogen peroxide by catalase. J. Biol. Chem. 195:133.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Chance, B., Maehly, A.C. 1955. Assay of catalase and peroxidase. Method Enzymol. 2:764–775.
Chen, G.X., Asada, K. 1989. Ascorbate peroxidase in tea leaves: Occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant and Cell Physiol. 30:987–998.
Dhanda, S.S., Sethi, G.S., Behl, R.K. 2004. Indices of drought tolerance in wheat genotypes at early stages of plant growth. J. Agron. Crop Sci. 190:6–12.
Di-Fonzo, N., Flagella, Z., Campanile, R.G., Stopelli, M.C., Spano, G., Rascio, A., Russo, M., Trono, D., Padalino, L., Laus, M., De Vita, P., Shewry, P.R., Lawlor, D., Troccoli, A. 2000. Resistance to abiotic stresses in durum wheat: Which ideotype? Options Mediterraneenes, Series A 40:215–225.
Dixit, V., Pandey, V., Shyam, R. 2001. Differential antioxidative response to cadmium in roots and leaves of pea. J. Exp. Bot. 52:1101–1109.
Drapeau, G.R. 1976. Protease from Staphylococcus aureus. Methods in Enzymology 45:469–475.
El-Tayeb, M.A. 2006. Differential response of two Vicia faba cultivars to drought: Growth, pigments, lipid peroxidation, organic solutes, catalase and peroxidase activity. Acta Agron. Hung. 54:25–37.
Farooq, M., Barsa, S.M.A., Wahid, A. 2006. Priming of field-sown rice seed enhances germination, seedling establishment, allometry and yield. Plant Growth Regul. 49:285–294.
Farooq, M., Wahid, A., Kobayashi, N., Fujita, D., Basra, S.M.A. 2009. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 29:185–212.
Gamboa-de Buen, A., Cruz-Ortega, R., Martinez-Barajas, E., Sanchez-Coronado, M.E., Orozco-Segovia, A. 2006. Natural priming as an important metabolic event in the life history of Wigandia urens (Hydrophyllaceae) seeds. Physiol. Plant. 128:520–530.
Giannopolitis, C.N., Ries, S.K. 1977. Superoxide dismutases occurrence in higher plants. Plant Physiol. 59:309–314.
Grudkowska, M., Zagdanska, B. 2004. Multifunctional role of plant cysteine proteinases. Acta Biochem Pol. 51:609–624.
Hameed, A., Afzal, I., Iqbal, N. 2010. Seed priming and salinity induced variations in wheat (Triticum aestivum L.) leaf protein profiles. Seed Sci. and Technol. 38:236–241.
Hameed, A., Bibi, N., Akhter, J., Iqbal, N. 2009. Mannose-induced modulations in antioxidants, protease activity, lipid peroxidation, and total phenolics in etiolated wheat leaves. J. Plant Growth Regul. 28:58–65.
Hameed, A., Bibi, N., Akhter, J., Iqbal, N. 2011. Differential changes in antioxidants, proteases, and lipid peroxidation in flag leaves of wheat genotypes under different levels of water deficit conditions. Plant Physiol. and Biochem. 49:178–185.
Hameed, A., Goher, M., Iqbal, N. 2012. Heat stress-induced cell death, changes in antioxidants, lipid peroxidation, and protease activity in wheat leaves. J. Plant Growth Regul. 31:283–291.
Heath, R.L., Packer, L. 1968. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125:189–198.
Hieng, B., Ugrinovic, K., Sustar, V.J., Kidric, M. 2004. Different classes of proteases are involved in the response to drought of Phaseolus vulgaris L. cultivars differing in sensitivity. J. Plant Physiol. 161:519–530.
Leishman, M.R., Westoby, M. 1994. The role of seed size in seedling establishment in dry soil conditions–Experimental evidence from semi-arid species. J. Ecol. 82:249–258.
Matsui, T., Singh, B.B. 2003. Root characteristics in cowpea related to drought tolerance at the seedling stage. Experimental Agriculture 39:29–38.
McCord, J.M. 2000. The evolution of free radicals and oxidative stress. Am. J. Med. 108:652–659.
McDonald, M.B. 2000. Seed priming. In: Black, M., Bewley, J.D. (eds), Seed Technology and Its Biological Basis. Sheffield Academic Press, Sheffield, UK, pp. 287–325.
Miller, G.L. 1972. Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal. Chem. 31:426–428.
Nguyen, G.N., Hailstones, D.L., Wilkes, M., Sutton, B.G. 2009. Drought induced oxidative conditions in rice anthers leading to a programmed cell death and pollen abortion. J. Agron. Crop Sci. 195:157–164.
Orvar, B.L., Ellis, B.E. 1997. Transgenic tobacco plants expressing antisense RNA for cytosolic ascorbate peroxidase show increased susceptibility to ozone injury. Plant J. 11:1297–1305.
Soltani, A., Gholipoor, M., Zeinali, E. 2006. Seed reserve utilization and seedling growth of wheat as affected by drought and salinity. Environ. Exp. Bot. 55:195–200.
Wahid, A., Perveen, M., Gelani, S., Basra, S.M.A. 2007. Pretreatment of seed with H2 O2 improves salt tolerance of wheat seedlings by alleviation of oxidative damage and expression of stress proteins. J. of Plant Physiol. 164:283–294.
Wahid, A., Noreen, A., Basra, S.M.A., Gelani, S., Farooq, M. 2008. Priming-induced metabolic changes in sunflower (Helianthus annuus) achenes improve germination and seedling growth. Botanical Studies 49:343–350.
Walters, C., Landre, P., Hill, L., Corbineau, F., Bailly, C. 2005. Organization of lipid reserves in cotyledons of primed and aged sunflower seeds. Planta 222:397–407.
Wang, H.Y., Chen, C.L., Sung, J.M. 2003. Both warm water soaking and solid priming treatments enhance anti-oxidation of bitter gourd seeds germinated at sub-optimal temperature. Seed Sci. Technol. 31:47–56.
Yagmur, M., Kaydan, D. 2008. Alleviation of osmotic stress of water and salt in germination and seedling growth of triticale with seed priming treatments. African J. of Biotechnol. 7:2156–2162.
Yang, R.C., Jana, S., Clarke, J.M. 1991. Phenotypic diversity and association of some potentially drought-responsive characters in durum wheat. Crop Sci. 31:1484–1491.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Pécsváradi
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hameed, A., Iqbal, N. Chemo-priming with Mannose, Mannitol and H2O2 Mitigate Drought Stress in Wheat. CEREAL RESEARCH COMMUNICATIONS 42, 450–462 (2014). https://doi.org/10.1556/CRC.2013.0066
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/CRC.2013.0066