Abstract
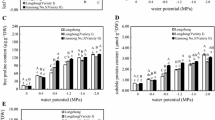
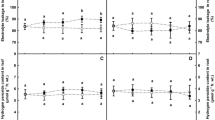
The effect of increasing NaCl concentrations on biomass, hydrogen peroxide (H2O2), ascorbic acid (ASC), proline and total thiol, and the activity of some antioxidant enzymes in alfalfa (Medicago sativa L. cv. Gara-Yonjeh) were investigated. The dry weights of roots and shoots with increasing NaCl concentrations decreased progressively, and the strongest toxicity was detected at NaCl treatment of 200 mM. Superoxide dismutase (SOD) activity in the leaves increased gradually up to NaCl concentrations of 100, while the higher concentration of NaCl reduced SOD activity in both leaves and roots. The maximum levels of ascorbate peroxidase (APX) activity were increased at 150 mM and 100 mM NaCl in leaves and roots of Gara-Yonjeh, respectively. Peroxidase (POD) activity in roots of Gara-Yonjeh increased (82% at 200 mM) by salinity, while it decreased (43% at 200 mM) in leaves. In contrast, catalase (CAT ) activitiy increased (84% at 200 mM) in leaves, and decreased (57% at 200 mM) in the roots of Gara-Yonjeh. Electrophoresis analysis suggested that different patterns in SOD, CAT and POD isoenzymes depend on NaCl concentrations, and the staining intensities of these isoforms are supported the results obtained from the spectrophotometric determinations. In POD and CAT, activity of isoform III was detected at all concentrations, by a “low-high-low” pattern, with the maximum activity at 50 mM of NaCl. Results imply that the function of antioxidant systems in higher NaCl concentration is responsible for the salt tolerance observed in Gara-Yonjeh.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Badawi, G. H., Yamauchi, Y., Shimada, E., Sasaki, R., Kawano, N., Tanaka, K., Tanaka, K. (2004) Enhanced tolerance to salt stress and water deficit by overexpressing superoxide dismutase in tobacco (Nicotiana tabacum) chloroplasts. Plant Sci. 166, 919–928.
Bartels, D., Sunkar, R. (2005) Drought and salt tolerance in plants. Crit. Rev. Plant Sci. 24, 23–58.
Bates, L. S., Waldren, R. P., Teare, I. D. (1973) Rapid determination of free proline for water stress studies. Plant Soil 39, 205–207.
Beauchamp, C., Fridovich, I. (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 44, 276–287.
Boominathan, R., Doran, P. M. (2002) Ni-induced oxidative stress in roots of the Ni hyperaccumulator, Alyssum bertolonii. New Phytol. 156, 205–215.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Annu. Rev. Biochem. 72, 248–254.
Cobbett, C. S. (2000) Phytochelatins biosynthesis and function in heavy-metal detoxification. Curr. Opin. Plant Biol. 3, 211–216.
Ellman, G. L. (1959) Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82, 70–77.
Ghamsari, L., Keyhani, E., Golkhoo, S. (2007) Kinetics properties of guaiacol peroxidase activity in Crocus sativus L. corm during rooting. Iran Biomed. J. 11, 137–146.
Hajiboland, R., Amjad, L. (2007) Does antioxidant capacity of leaves play a role in growth response to selenium at different sulfur nutritional status? Plant Soil Environ. 53, 207–215.
Howieson, J., Ballard, R. (2004) Optimising the legume symbiosis in stressful and competitive environments within southern Australia: some contemporary thoughts. Soil Biol. Biochem. 36, 1261–1273.
Hu, M. L. (1994) Measurement of protein thiol groups and glutathione in plasma. Methods Enzymol. 233, 380–385.
Jebara, S., Jebara, M., Limam, F., Aouani, M. E. (2005) Changes in ascorbate peroxidase, catalase, guaiacol peroxidase and superoxide dismutase activities in common bean (Phaseolus vulgaris) nodules under salt stress. J. Plant. Physiol. 162, 929–936.
Kim, S. Y., Lim, J. H., Park, M. R., Kim, Y. J., Park, T. I. I., Seo, Y. W. (2005) Enhanced antioxidant enzymes are associated with reduced hydrogen peroxide in barley roots under salt stress. J. Biochem. Mol. Biol. 38, 218–224.
Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685.
Lee, D. H., Kim, Y. S., Lee, C. B. (2001) The inductive responses of the antioxidant enzymes by salt stress in the rice (Oryza sativa L.). J. Plant Physiol. 158, 737–745.
Lin, C. C., Kao, C. H. (1996) Proline accumulation is associated with inhibition of rice seedling root growth caused by NaCl. Plant Sci. 114, 121–128.
Marschner, H. (1995) Mineral Nutrition of Higher Plants. London, Academic Press.
Martía, M. C., Camejoa, D., Fernández-García, N., Rellán-Álvarezb, R., Marquesc, S., Sevilla, F., Jiménez, A. (2009) Effect of oil refinery sludges on the growth and antioxidant system of alfalfa plants. J. Hazard. Mater. 171, 879–885.
Moran, J. F., Becana, M., Iturbe-Ormaetxe, I., Frechilla, S., Klucas, R. V., Aparicio-Tejo, P. (1994) Drought induces oxidative stress in pea plants. Planta 194, 346–325.
Naya, L., Ladrera, R., Ramos, J., Gonzalez, E. M., Arrese-Igor, C., Minchin, F. R., Becana, M. (2007) The response of carbon metabolism and antioxidant defenses of alfalfa nodules to drought stress and to the subsequent recovery of plants. Plant Physiol. 144, 1104–1114.
Noctor, G., Foyer, C. H. (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 249–279.
Obinger, C., Maj, M., Nicholls, P., Loewen, P. (1997) Activity, peroxide compound formation, and heme d synthesis in Escherichia coli HPII catalase. Arch. Biochem. Biophys. 342, 58–67.
Okeri, H. A., Alonge, P. O. (2006) Determination of the ascorbic acid content of two medicinal plants in Nigeria. Pak. J. Pharm. Sci. 19, 39–44.
Platiša, J., Veljovic-Jovanovic, S., Kukavica, B., Vinterhalter, B., Smigockic, A., Ninkovic, S. (2008) Induction of peroxidases and superoxide dismutases in transformed embryogenic calli of alfalfa (Medicago sativa L.). J. Plant Physiol. 165, 895–900.
Poustini, K., Siosemardeh, E. A., Ranjbar, E. M. (2007) Proline accumulation as a response to salt stress in 30 wheat (Triticum aestivum L.) cultivars differing in salt tolerance. Genet. Resour. Crop Evol. 54, 925–934.
Tambussi, E. A., Bartoli, C. G., Belrano, J., Guiamet, J. J., Arans, J. L. (2000) Oxidative damage to thylakoid protein in water stressed leaves of wheat (Triticum aestivum). Physiol. Plant 108, 398–404.
Tejera Garcí, N. A., Iribarne, C., Palma, F., Lluch, C. (2007) Inhibition of the catalase activity from Phaseolus vulgaris and Medicago sativa by sodium chloride. Plant Physiol. Biochem. 45, 535–541.
Tennant, D. (1975) A test of modified line intersect method of estimating root length. J. Ecol. 63, 995–1001.
Vaidyanathan, H., Sivakumar, P., Chakrabarty, R., Thomas, G. (2003) Scavenging of reactive oxygen species in NaCl-stressed rice (Oryza sativa L.)-differential response in salt-tolerant and sensitive varieties. Plant Sci. 165, 1411–1418.
Velikova, V., Yordanov, I., Edreva, A. (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Protective role of exogenous polyamines. Plant Sci. 151, 59–66.
Wang, W. B., Kim, Y. H., Lee, H. S., Kim, K. Y., Deng, X. P., Kwak, S. S. (2009) Analysis of antioxidant enzyme activity during germination of alfalfa under salt and drought stresses. Plant Physiol. Biochem. 47, 570–577.
Winterbourn, C. C., McGrath, B. W., Carrell, R. W. (1976) Reactions involving superoxide and normal and unstable haemoglobins. Biochem. J. 155, 493–502.
Woodbury, W., Spencer, A. K., Stahman, M. A. (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal. Biochem. 44, 301–305.
Zhou, Z. S., Huang, S. Q., Guo, K., Kant Mehta, S., Zhang, P. C., Yang, Z. M. (2007) Metabolical adaptations to mercury-induced oxidative stress in roots of Medicago sativa L. J. Inorg. Biochem. 101, 1–9.
Zhu, J. K. (2002) Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 53, 247–273.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Dehghan, G., Amjad, L. & Nosrati, H. Enzymatic and Non-enzymatic Antioxidant Responses of Alfalfa Leaves and Roots Under Different Salinity Levels. BIOLOGIA FUTURA 64, 207–217 (2013). https://doi.org/10.1556/ABiol.64.2013.2.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.64.2013.2.7