Abstract
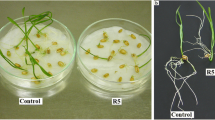
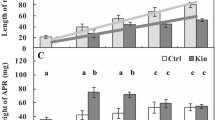
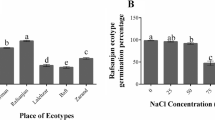
This study compares the histological, cytological and biochemical effects of the cyanobacterial toxins microcystin-LR (MCY-LR) and cylindrospermopsin (CYN) in white mustard (Sinapis alba L.) seedlings, with special regard to the developing root system. Cyanotoxins induced different alterations, indicating their different specific biochemical activities. MCY-LR stimulated mitosis of root tip meristematic cells at lower concentrations (1 μg ml−1) and inhibited it at higher concentrations, while CYN had only inhibitory effects. Low CYN concentrations (0.01 μg ml−1) stimulated lateral root formation, whereas low MCY-LR concentrations increased only the number of lateral root primordia. Both inhibited lateral root development at higher concentrations. They induced lignifications, abnormal cell swelling and inhibited xylem differentiation in roots and shoots. MCY-LR and CYN induced the disruption of metaphase and anaphase spindles, causing altered cell divisions. Similar alterations could be related to decreased protein phosphatase (PP1 and PP2A) activities in shoots and roots. However, in vitro phosphatase assay with purified PP1 catalytic subunit proved that CYN in contrast to MCY-LR, decreased phosphatase activities of mustard in a non-specific way. This study intends to contribute to the understanding of the mechanisms of toxic effects of a protein phosphatase (MCY-LR) and a protein synthesis (CYN) inhibitory cyanotoxin in vascular plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BGST:
-
Blue-Green Sinapis Test
- CYN:
-
cylindrospermopsin
- DAPI:
-
4’,6’-diamidino-2 phenylindole
- GPx:
-
Glutathione-peroxidase enzymes
- GST:
-
Glutathione-S-transferase enzymes
- IC50:
-
fifty percent inhibitory concentration
- MAPs:
-
microtubule associated proteins
- MCY:
-
microcystins
- MCY-LR:
-
microcystin-LR
- PCD:
-
programmed cell death
- PP1:
-
serine/threonine protein phosphatases of type 1
- PP1c:
-
catalytic subunit of protein phosphatase 1
- PP2A:
-
serine/threonine protein phosphatases of type 2A
- ROS:
-
reactive oxygen species
References
Abe, T., Lawson, T., Weyers, J. D. B., Codd, G. A. (1996) Microcystin-LR inhibits photosynthesis of Phaseolus vulgaris primary leaves: Implications for current spray irrigation practice. New Phytol. 133, 651–658.
Allen, M. M. (1968) Simple conditions for the growth of unicellular blue-green algae on plates. J. Phycol. 4, 1–4.
Armstrong, J., Armstrong, W. (2001) An overview of the effects of phytotoxins on Phragmites aus-tralis in relation to die-back. Aquatic Bot. 69, 251–268.
Banker, R., Carmeli, S., Hadas, O., Teltsch, B., Porat, R., Sukenik, A. (1997) Identification of cylin-drospermopsin in Aphanizomenon ovalisporum (Cyanophyceae) isolated from lake Kinneret, Israel. J. Phycol. 33, 613–616.
Beyer, D., Surányi, G., Vasas, G., Roszik, J., Erdődi, F., M-Hamvas, M., Bácsi, I., Bátori, R., Serfőző, Z., Szigeti, Z. M., Vereb, G., Demeter, Z., Gonda, S., Mathe, C. (2009) Cylindrospermopsin induces alterations of root histology and microtubule organization in common reed (Phragmites australis) plantlets cultured in vitro. Toxicon 54, 440–449.
Campos, A., Vasconcelos, V. (2010) Molecular mechanisms of microcystin toxicity in animal cells. Review. Int. J. Mol. Sci. 11, 268–287.
Carmichael, W. W., Azevedo, S. M. F. O., An, J. S., Molica, R. J. R., Jochimsen, E. M., Lau, S., Rinehart, K. L., Shaw, G. R., Eaglesham, G. K. (2001) Human fatalities from cyanobacteria: chemical and biological evidence for cyanotoxins. Environ. Health Persp. 109, 663–668.
Chaffey, N., Cholewa, E., Regan, S., Sundberg, B. (2002) Secondary xylem development in Arabidopsis: a model for wood formation. Physiol. Plan. 114, 594–600.
Chen, J., Han, F. X., Wang, F., Zhang, H., Shi, Z. (2012) Accumulation and phytotoxicity of micro-cystin-LR in rice (Oryza sativa). Ecotoxicol. Environ. Saf. 76, 193–199.
Chen, J., Song, L., Dai, J., Gan, N., Liu, Z. (2004) Effects of microcystins on the growth and the activity of superoxide dismutase and peroxidase of rape (Brassica napus L.) and rice (Oryza sativa L.). Toxico. 43, 393–400.
Chong, M. W. K., Wong, B. S. E., Lam, P. K. S., Shaw, G. R., Seawright, A. A. (2002) Toxicity and uptake mechanism of cylindrospermopsin and lophyrotomin in primary rat hepatocytes. Toxico. 40, 205–211.
Conrath, U., Pieterse, C. M. J., Mauch-Mani, B. (2002) Priming in plant-pathogen interactions. Trends Plant Sci. 7, 210–216.
Demeter, A., Vámosi, J., Peregovits, L., Topál, G. (1995) An image capture and data collection system for morphometric studies. In: Marcus, L. F., Corti, M., Loy, A., Naylor, G., Slice, D. E. (eds) Advances in Morphometries. Plenum Press, New York, pp. 91–101.
Erdődi, F., Tóth, B., Hirano, K., Hirano, M., Hartshorne, D. J., Gergely, P. (1995) Endothall thioan-hydride inhibits protein phosphatases-1 and -2A in vivo. Am. J. Physiol. 269, C1176–C1184.
Froscio, S. M., Humpage, A. R., Wickramasinghe, W., Shaw, G., Falconer, I. R. (2008) Interaction of the cyanobacterial toxin cylindrospermopsin with the eukaryotic protein synthesis system. Toxico. 51, 191–198.
Fukuda, H. (2004) Signals that control plant vascular cell differentiation. Nat. Rev. Mol. Cell Bio. 5, 379–391.
Gácsi, M., Antal, O., Vasas, G., Máthé, C., Borbély, G., Saker, M. L., Győri, J., Farkas, A., Vehovszky, Á., Bánfalvi, G. (2009) Comparative study of cyanotoxins affecting cytoskeletal and chromatin structures in CHO-K1 cells. Toxicol, in Vitr. 23, 710–718.
Gehringer, M. M. (2004) Microcystin-LR and okadaic acid-induced cellular effects: a dualistic response. FEES Lett. 557, 1–8.
Gergely, P., Erdődi, F., Bot, G. (1984) Heparin inhibits the activity of protein phosphatase-1. FEES Lett. 169, 45–48.
Huang, W., Xing, W., Li, D., Liu, Y. (2008) Microcystin-RR induced apoptosis in tobacco BY-2 suspension cells is mediated by reactive oxygen species and mitochondrial permeability transition pore status. Toxicol, in Vitr. 22, 328–337.
Kinnear, S. H. W., Fabbro, L. D., Duivenvoorden, L. J. (2008) Variable growth responses of water thyme (Hydrilla verticillata) to whole-cell extracts of Cylindrospermopsis raciborskii. Arc. Environ Contain. Toxicol. 54, 187–194.
Kós, R., Gorzó, G., Suranyi, G., Borbély, G. (1995) Simple and efficient method for isolation and measurement of cyanobacterial hepatotoxins by plant tests (Sinapis alba). Anal. Biochem. 225, 49–53.
Kováts, N., Ács, A., Paulovits, G., Vasas, G. (2011) Response of Lemna minor clones to microcystis toxicity. Appl. Ecol. Env. Res. 9, 17–26.
Kurki-Helasmo, K., Meriluoto, J. (1998) Microcystin uptake inhibits growth and protein phosphatase activity in mustard (Sinapis alba L.) seedlings. Toxico. 36, 1921–1926.
Luan, S. (2003) Protein phosphatases in plants. Annu. Rev. Plant Biol. 42, 63–92.
MacKintosh, C., Beattie, K. A., Klumpp, S., Cohen, P., Codd, G. A. (1990) Cyanobacterial microcys-tin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEES Lett. 264, 187–192.
Máthé, C., M-Hamvas, M., Vasas, G., Surányi, G., Bácsi, I., Beyer, D., Tóth, S., Tímár, M., Borbely, G. (2007) Microcystin-LR, a cyanobacterial toxin, induces growth inhibition and histological alterations in common reed (Phragmites australis) plants regenerated from embryonic calli. New Phyto. 176, 824–835.
Máthé, C., Beyer, D., Erdődi, F., Serfőző Z., Székvölgyi, L., Vasas, G., M-Hamvas, M., Jámbrik, K., Gonda, S., Kiss, A., Szigeti Z. M., Suranyi, G. (2009) Microcystin-LR induces abnormal root development by altering its microtubule organization in tissue-cultured common reed (Phragmites australis) plantlets. Aquat. Toxicol. 92, 122–130.
Metcalf J. S., Barakate, A., Codd, G. A. (2004) Inhibition of plant protein synthesis by the cyanobacterial hepatotoxin, cylindrospermopsin. FEMS Microbiol. Lett. 235, 125–129.
Metcalf, J. S., Lindsay, J., Beattie, K. A., Birmingham, S., Saker, M. L., Törökné, A. K., Codd, G. A. (2002) Toxicity of cylindrospermopsin to the brine shrimp Artemia salina: comparisons with protein synthesis inhibitors and microcystins. Toxico. 40, 1115–1120.
M-Hamvas, M., Máthé, Cs., Molnár, E., Vasas, G., Grigorszky, I., Borbely, G. (2003) Microcystin-LR alters the growth, anthocyanin content and single-stranded DNase enzyme activities in Sinapis alba L. seedlings. Aquat. Toxicol. 62, 1–9.
M-Hamvas, M., Máthé, C., Vasas, G., Jámbrik, K., Papp, M., Beyer, D., Mészaros, I., Borbély, G. (2010) Cylindrospermopsin and microcystin-LR alter the growth, development and peroxidase enzyme activity of white mustard (Sinapis alba L.) seedlings, a comparative analysis. Acta Biol Hung. 61 (Suppl), 35–48.
Padisak, J. (1997) Cylindrospermopsis raciborkii (Woloszynska) Seenaya et Subba Raju, an expanding, highly adaptive cyanobacterium: worldwide distribution and review of its ecology. Arch. Hidrobiol. Suppl. 4, 563–593.
Peuthert, A., Chakrabarti, S., Pflugmacher, S. (2007) Uptake of microcystin-LR and -LF (cyanobacterial toxins) in seedlings of several important agricultural plant species and the correlation with cellular damage (lipid peroxidation). Environ. Toxicol. 22, 436–442.
Pflugmacher, S. (2004) Promotion of oxidative stress in aquatic macrophyte Ceratophyllum demer-sum during biotransformation of the cyanobacterial toxin microcystin-LR. Aquat. Toxicol. 70, 169–178.
Prieto, A., Campos, A., Camean, A., Vasconcelos, V. (2011) Effects on growth and oxidative stress status of rice plants (Oryza sativa) exposed to two extracts of toxin-producing cyanobacteria (Aphanizomenon ovalisporum and Microcystis aeruginosa). Ecotox. Environ. Safe. 74, 1973–1980.
Puerto, M., Campos, A., Prieto, A., Cameán, A., de Almeida, A. M., Coelho, A. V., Vasconcelos, V. (2011) Differential protein expression in two bivalve species: Mytilus galloprovincialis and Corbicula fluminea; exposed to Cylindrospermopsis raciborskii cells. Aquat. Toxicol. 101, 109–116.
Ros Barceló, A. (1998) The generation of H2O2 in the xylem of Zinnia elegans is mediated by an NADPH-oxidase-like enzyme. Plant. 207, 207–216.
Runnegar, M. T., Kong, S. M., Zhong, Y. Z., Lu, S. C. (1995) Inhibition of reduced glutathion synthesis by cyanobacterial alkaloid cylindrospermopsin in cultured rat hepatocytes. Biochem. Pharmacol. 49, 219–225.
Smertenko, A. P., Chang, H. Y., Sonobe, S., Fenyk, S. I., Weingartner, M., Bogre, L., Hussey, P. J. (2006) Control of the AtMAP65-l interaction with microtubules through the cell cycle. J. Cell Sci. 119, 3227–3237.
Törökné, A. K., László, E., Chorus, I., Fastner, I., Heinze, R., Padisák, J., Barbosa, F. A. R. (2000) Water quality monitoring by Thamnotoxkit F(™) including cyanobacterial blooms. Water Sci. Techno. 42, 381–385.
Vasas, G., Cáspár, A., Páger, C., Surányi, G., Máthé, C., M-Hamvas, M., Borbely, G. (2004) Analysis of cyanobacterials toxins (anatoxin-a, cylindrospermopsin, microcystin-LR) by capillary electrophoresis. Electrophoresi. 25, 108–115.
Vasas, G., Gáspár, A., Surányi, G., Batta, G., Gyémánt, G., M-Hamvas, M., Máthé, C., Grigorszky, I., Molnár, E., Borbély, G. (2002) Capillary electrophoretic assay and purification of cylindrospermopsin, a cyanobacterial toxin from Aphanizomenon ovalisporum by plant test (Blue-Green Sinapis Test). Anal. Biochem. 302, 95–103.
Žegura, B., Štraser, A., Filipič, M. (2011) Genotoxicity and potential carcinogenicity of cyanobacterial toxins - a review. Mutat. Res.-Rev Mutat. 727, 16–41.
Acknowledgements
We acknowledge Dr. Ola Hadas for kindly helping with Aphanizomenon ovalisporum ILC164. We thank Dr. István Grigorszky for his help in taking microscopical photos. The work was supported by the Hungarian National Research Foundation Grants OTKA F46493, K68416, K81370, GVOP-3.2.1.-2004-04-0110/3.0. M. M-Hamvas, C. Máthé, G. Surányi and G. Vasas greatly acknowledge the personal support from Bolyai Janos Scholarship of the Hungarian Academy of Sciences. Mrs. Ágnes Németh, Éva Barabás and Katalin Havelant are acknowledged for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Máthé, C., Vasas, G., Borbély, G. et al. Histological, Cytological and Biochemical Alterations Induced by Microcystin-LR and Cylindrospermopsin in White Mustard (Sinapis alba L.) Seedlings. BIOLOGIA FUTURA 64, 71–85 (2013). https://doi.org/10.1556/ABiol.64.2013.1.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.64.2013.1.7