Abstract
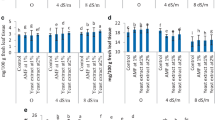
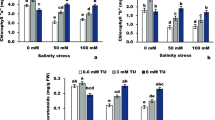
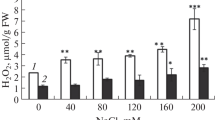
The combined effects of yeast (1 ppm) and salinity on germination, seedling growth, metabolite accumulation and antioxidant defense system of flax (Linum usitatissimum) seeds grown at 100, 200 and 300 mM NaCl were studied. In this investigation, the germination was completely inhibited at 300 mM NaCl. Moreover, salinity induced marked increases in lipid peroxidation product (MDA), soluble carbohydrates as well as the reduced glutathione which were concomitant with sharp decrease in total phenols and ascorbic acid contents in 12-day-old flax seedlings. Furthermore, NaCl treatments increased the activities of some antioxidant enzymes (superoxide dismutase; SOD, peroxidase; POX and polyphenol oxidase; PPO). On the other hand, yeast treatments under salinity stress restored the membrane integrity and improved seedling growth. The results suggested that yeast treatments mitigated salinity stress via accumulation of some osmoprotectants such as free amino acids particularly proline which associated with elevating the defense system in terms of ascorbic acid, glutathione and total phenol contents. Yeast treatments also stimulated the activities of some antioxidant enzymes, preventing membrane peroxidation resulting in high capacity for germination and improved seedling growth under sever salt stress.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ASA:
-
ascorbic acid
- PPO:
-
polyphenol oxidase
- POX:
-
peroxidase
- SOD:
-
superoxide dismutase
- CAT:
-
catalase
- MDA:
-
malondialdehyde
- GSH:
-
glutathione
References
Al-Tawaha, A. R. M. (2011) Effect of soil type and exogenous application of yeast extract on soybean seed isoflavones concentration. Int. J. Agric. Biol. 13, 275–278.
Amer, S. S. A. (2004) Growth, green pods yield and seeds yield of common bean (Phaseolus vulgaris L.) as affected by active dry yeast, salicylic acid and their interaction. J. Agric. Sci. Mansoura Univ. 29, 1407–1422.
Apel, K., Hirt, H. (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 55, 373–399.
Azooz, M. M. (2009) Foliar application with riboflavin (vitamin B2) enhancing the resistance of Hibiscus sabdariffa L. (Deep Red Sepals Variety) to salinity stress. J. Biol. Sci. 9, 109–118.
Badawi, G. H., Yamauchi, Y., Shimada, E., Sasaki, R., Kawano, N., Tanaka, K. (2004) Enhanced tolerance to salt and water deficit by over-expressing superoxide dismutase in tobacco (Nicotiana tabacum) chloroplasts. Plant Sci. 166, 919–928.
Bates, L. S., Waldren, R. P., Tear, I. D. (1973) Rapid determination of free proline for water-stress studies. Plant and Soi. 39, 205–207.
Ben Amor, M., Ben Hamed, K., Debez, A., Grignon, C., Abdelly, C. (2005) Physiological and antioxidant responses of the perennial halophyte Crithmum maritimum to salinity. Plant Sci. 168, 889–899.
Bergmeyer, H. U., Gaulehn, K., Grassl, M. (1974) Methods of Enzymatic Analysis. 2nd edn. Academic Press, New York, pp. 495–496.
Blakeney, A. B., Mutton, L. L. (1980) A simple calorimetric method for the determination of sugars in fruits and vegetables. J. Sci. Food Agric. 31, 889–897.
Boggess, S. F., Aspinall, D., Paleg, L. G. (1976) Stress metabolism. IX. The significance of end product inhibition of proline synthesis and of compartmentation in relation to stress-induced proline accumulation. Aust. J. Plant Physiol. 3, 513–525.
Boscaiu, M., Estrelles, E., Soriano, P., Vicente, O. (2005) Effects of salt stress on the reproductive biology of the halophyte Plantago crassifolia. Biol. Plan. 49, 141–143.
Bradford, M. M. (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72, 248–254.
Bray, E. A., Bailey-Serres, J., Weretilny, K. E. (2000) Responses to abiotic stress: In: Buchanan, B. B., Gruissem, W., Jones, R. L. (eds) Biochemistry and Molecular Biology of Plants. American Society of Plant Biologists, Waldorf, pp. 1158–1203.
Chaparzadeh, N., Amico, M. L. D., Khavari-Najad, R. A., Izzo, R., Navarizzo, F. (2004) Antioxidative responses of Calendula officinalis under salinity conditions. Plant Physiol. Biochem. 42, 695–701.
El-Bassiouny, H. M. S., Gobarah, M. E., Ramadan, A. A. (2005) Effect of antioxidants on growth, yield and Favism Causative Agents in seeds of Viciafaba L. plants grown under reclaimed sandy soil. J. Agron. 4, 281–287.
El-Tohamy, W. A., El-Gready, N. H. M. (2007) Physiological responses, growth, yield and quality of snap beans in response to foliar application of yeast, vitamin E and zinc under sandy soil conditions. Aust. J. Basic Appl. Sci. 1, 294–299.
Fathy, E. S. L., Farid, S. (1996) The possibility of using vitamin Bs and yeast to delay senescence and improve growth and yield of common beans (Phaseolus vulgaris L.). J. Agric. Sci. Mansoura Univ. 21, 1415–1423.
Foyer, C. H. (1993) Ascorbic acid. In: Alscher, R. G., Hess, J. L. (eds) Antioxidants in Higher Plants. CRC Press, Boca Raton, pp. 31–58.
Frechill, S., Lasa, B., Ibarretxe, L., Lamsfus, C., Aparicio Trejo, P. (2001) Pea responses to saline stress is affected by the source of nitrogen nutrition (ammonium or nitrate). Plant Growth Regul. 35, 171–179.
Gaballah, M. S., Ouda, S. A., Mandour, M. S., Rady, M. M. (2006) Predicting the role of antioxidants and irrigation on sunflower yield grown under saline conditions. In: Proceedings of the IASTED International Conference on Environmentally Sound Technology in Water Resources Management, 515–043.
Gaber, A. M. (1981) Studies on nitrogen metabolism in certain plant tissues. M. Sc. Thesis, Mansoura University, Mansoura, Egypt.
Hauschild, R., Von Schaewen, A. (2003) Differential regulation of glucose-6-phosphate dehydrogenase isoenzyme activities in potato. Plant Physiol. 133, 47–62.
Heath, R. L., Packer, L. (1968) Photo-peroxidation in isolated chloroplasts. I. Kinetics and stoichiom-etry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 189–198.
Homme, P. M., Gonalez, B., Billard, J. (1992) Carbohydrate content, fructane and sucrose enzyme activities in roots, stubble and leaves of rye grass (Lolium perenne L.) as affected by source/sink modification after cutting. J. Plant Physiol. 140, 282–291.
Hoque, M. D. A., Okuma, E., Banu, M. N. A., Nakamura, Y., Shimoishi, Y., Murata, Y. (2007) Exogenous proline mitigates the detrimental effects of salt stress more than exogenous betaine by increasing antioxidant enzyme activities. J. Plant Physiol. 164, 553–561.
Jain, M., Mathur, G., Koul, S., Sarin, N. B. (2001) Ameliorating effects of proline on salt stress lipid peroxidation in cell lines of groundnut (Arachis hypogea L.). Plant Cell Report 20, 463–468.
Jaleel, C. A., Sankar, B., Sridharan, R., Panneerselvam, R. (2008) Soil salinity alters growth, chlorophyll content, and secondary metabolite accumulation in Catharanthus roseus. Turk. J. Biol. 32, 79–83.
Kampfenkel, K., Van Montagu, M., De Inzé, D. (1995) Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal. Biochem. 225, 165–167.
Kar, M., Mishra, D. (1976) Catalase, peroxidase and polyphenol oxidase activities during rice leaf senescence. Plant Physiol. 57, 315–319.
Koch, K. E. (1996) Carbohydrate-modulated gene expression in plants. Ann. Review Plant Physiol. Plant Mol. Biol. 47, 509–540.
Kocsy, G., Galiba, G., Brunold, C. (2001) Role of glutathione in adaptation and signaling during chilling and acclimation in plants. Physiol. Plant. 113, 158–164.
Ksouri, R., Megdiche, W., Debez, A., Falleh, H., Grignon, C., Abdelly, C. (2007) Salinity effects on polyphenol content and antioxidant activities in leaves of the halophyte Cakile maritima. Plant Physiol. Biochem. 45, 244–249.
Lin, C. C., Kao, C. H. (2002) Osmotic stress-induced changes in cell wall peroxidase activity and hydrogen peroxide level in roots of rice seedlings. Plant Growth Regulatio. 37, 177–184.
Lopez, M., Tejera, N. A., Liuch, C. (2008) Differential strategies of the model legumes Lotus japoni-cus and Medicago truncatula in the adaptation to salt stress: Photosynthetic and nutritional responses. Am. J. Plant Physiol. 3, 121–130.
Malik, C. P., Singh, M. B. (1980) Plant Enzymology of Histoenzymology. Kalyani Publishers, New Delhi.
Marklund, S., Marklund, G. (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47, 469–474.
May, M. J., Vernoux, T., Leaver, C., Van Montagu, M., Inze, D. (1998) Glutathione homeostasis in plants: implications for environmental sensing and development. J. Exp. Bot. 49, 649–667.
Mckersie, B. D., Bowley, S. R., Jones, K. S. (1999) Winter survival of transgenic alfalfa over-expressing superoxide dismutase. Plant Physiol. 119, 839–847.
Mutting, D., Kaiser, E. (1963) Spectrophotometric method of determing amino-N in biological materials by means of the ninhydrin reaction. Seyler’s Zeitsch. Physiol. Chem. 232, 276–281.
Naguib, M. I. (1963) Colorimetric estimation of plant polysaccharides. Zeitsch. Zucker. 16, 15–26.
Navarro, J. M., Flores, P., Garrido, C., Martinez, V. (2006) Changes in the contents of antioxidant compounds in pepper fruits at ripening stages, as affected by salinity. Food Chem. 96, 66–73.
Noctor, G., Foyer, C. H. (1998) Ascorbate and glutathione: Keeping active oxygen under control. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 249–279.
Nuran, X., Hüsnü, Ç. (2002) The effect of salinity on some physiological parameters in two maize cultivars. Bulg. J. Plant Physiol. 28, 66–74.
Rice-Evans, C., Miller, N. J., Paganga, G. (1997) Antioxidant properties of phenolic compounds. Trends in Plant Sci. 2, 152–159.
Rontein, D., Basset, G., Hanson, A. D. (2002) Metabolic engineering of osmoprotectants accumulation in plants. Metab. Eng. 4, 49–56.
Rout, N. P., Shaw, B. P. (2001) Salt tolerance in aquatic macrophytes: Possible involvement of the antioxidative enzymes. Plant Sci. 160, 415–423.
Ryu, J. Y., Song, J. Y., Lee, J. M., Jeong, S. W., Chow, W. S., Choi, S. B., Pogson, B. J., Park, Y. I. (2004) Glucose-induced expression of carotenoid biosynthesis genes in the dark is mediated by cyto-solic pH in the cyanobacterium Synechocystis sp. PCC 6803. J. Biol. Chem. 279, 25320–25325.
Sairam, R. K., Tyagi, A. (2004) Physiology and molecular biology of salinity stress tolerance in plants. Curr. Sci. 86, 407–412.
Salter, J., Morris, K., Bailey, P. C. E., Boon, P. I. (2007) Interactive effects of salinity and water depth on the growth of Melaleuca ericifolia Sm. (Swamp paperbark) seedlings. Aquatic Bot. 86, 213–222.
Schupp, R., Rennenperg, H. (1988) Diurnal changes in the glutathione content of spurce needle (Picea abiec L.). Plant Sci. 27, 113–117.
Shao, H. B., Chu, L. Y., Lu, Z. H., Kang, C. M. (2008) Primary antioxidant free radical scavenging and redox signaling pathway in higher plant cells. Int. J. Biol. Sci. 4, 8–14.
Smirnoff, N., Conklin, P., Loewus, F. A. (2001) Biosynthesis of ascorbic acid in plants: A Renaissance. Ann. Rev. Plant Physiol. Plant Mol. Biol. 52, 437–467.
Smith, A. G., Croft, M. T., Moulin, M., Webb, M. E. (2007) Plants need their vitamins too. Physiol. Metab. 10, 266–275.
Tripathi, S. B., Gurumurthi, K., Panigrahi, A. K., Shaw, B. P. (2007) Salinity induced changes in proline and betaine contents and synthesis in two aquatic macrophytes differing in salt tolerance. Biol. Plan. 51, 110–115.
Vartanian, N., Hervochon, P., Marcotte, L., Larher, F. (1992) Proline accumulation during drought rhizogenesis in Brassica napus var. Oleifera. Plant Physiol. 140, 623–638.
Westscott, N. D., Muir, A. D. (2003) Chemical studies on the constituents of Linum spp. In: Muir, A. D., Westscott, N. D. (eds) Flax, the Genus Linum. Taylor and Francis, London, pp. 55–73.
Xu, J. B., Yuan, X. F., Lang, P. Z. (1997) Determination of catalase inhibition by ultraviolet spectrophotometry. Chinese Environ. Chem. 16, 73–76.
Younis, M. E., Hasaneen, M. N. A., Nmet-Alla, M. M. (1987) Plant growth, metabolism and adaptation in relation to stress conditions IV. Effects of salinity on certain factors associated with the germination of three different seeds high in fats. Ann. Bot. 60, 337–344.
Acknowledgement
I sincerely express my gratitude to Prof. Hemmat Khattab (prof. of plant physiology, Faculty of science, Ain Shams University) for her valuable guidance and encouragement during this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Emam, M.M. Efficiency of Yeast in Enhancement of the Oxidative Defense System in Salt-Stressed Flax Seedlings. BIOLOGIA FUTURA 64, 118–130 (2013). https://doi.org/10.1556/ABiol.64.2013.1.11
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.64.2013.1.11