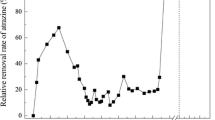
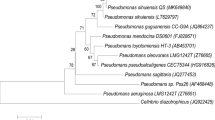
Abstract
Six isolates with phenol degrading ability were obtained from marine sediments by enrichment procedures and an isolate, AM4, was identified as Alcaligenes sp. by 16S rDNA sequencing. The Plackett-Burman design was applied to estimate the significance of culture medium components and conditions for phenol degradation by Alcaligenes sp. AM4. The resulting medium formula which was predicted to be near optimal was: phenol conc. (240 μg/ml), culture volume (37.5 ml), inoculum’s size (0.15 ml), NH4SO4(0.5 g/l), K2HPO4(0.75 g/l), KH2PO4(0.75 g/l), MgSO4(0.3 g/l) and NaCl (0.25 g/l). Scanning electron microscopy was applied to cells exposed to phenol, and a larger cell size was detected, resulting in a reduced cell surface. This relative reduction of the cell surface represents a cellular mechanism to reduce the toxic effect of this environmental stress factor.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agarry, S. E., Solomon, B. O., Layokun, S. K. (2008) Substrate inhibition kinetics of phenol degradation by binary mixed culture of Pseudomonas aeruginosa and Pseudomonas fluorescence from steady state and wash-out data. Afri. J. Biotechnol. 7, 3927–3933.
Aksu, S., Yener, J. (1998) Investigation of biosorption of phenol and monochlorinated phenols on the dried activated sludge. Process Biochem. 33, 649–655.
Allsop, P. J., Chisti, Y., Moo-Young, M., Sullivan, G. R. (1993) Dynamics of phenol degradation by Pseudomonas putida. Biotechnol. Bioeng. 41, 572–580.
Annadurai, G., Ling, L. Y., Lee, J. F. (2008) Statistical optimization of medium components and growth conditions by response surface methodology to enhance phenol degradation by Pseudomonas putida. J. Hazard Mater. 151, 171–178.
Bulbul, G., Aksu, Z. (1997) Investigation of wastewater treatment containing phenol using free and Ca-alginated gel immobilized Pseudomonas putida in a batch stirred reactor. Turkish J. Eng. Environ. Sci. 21, 175–181.
Chen, Q., Janssen, D. B., Witholt, B. (1996) Physiological changes and alk gene instability in P seudomonas oleovorans during induction and expression of alk genes. J. Bacteriol. 178, 5508–5512.
Daniel, R. A., Errington, J. (2003) Control of cell morphogenesis in bacteria: two distinct ways to make a rod-shaped cell. Cel. 113, 767–776.
Emerson, E. (1943) The condensation of aminoantipyrine. II. A new color test for phenolic compounds. J. Org. Chem. 8, 417–428.
Hannaford, A. M., Kuek, C. (1999) Aerobic batch degradation of phenol using immobilized Pseudomonas putida. J. Ind. Microbiol. Biotechnol. 22, 121–126.
Hill, G. A., Robinson, C. W. (1975) Substrate inhibition kinetics: phenol degradation by Pseudomonas putida. Biotechnol. Bioeng. 17, 599–615.
Imandi, S. B., Bandaru, V. V., Somalanka, S. R., Bandaru, S. R., Garapati, H. R. (2008) Application of statistical experimental designs for the optimization of medium constituents for the production of citric acid from pineapple waste. Bioresour Technol. 99, 4445–4450.
Jiang, Y., Jianping Wen, J., Bai, J., Jia, X., Zongding Hu, Z. (2007) Biodegradation of phenol at high initial concentration by Alcaligenes faecalis. J. Hazard Mat. 147, 672–676.
Kapoor, A., Kumar, R., Kumar, A., Sharma, A., Prasad, S. (1998) Application of immobilized mixed bacterial culture for the degradation of phenol present in oil refinery effluent. J. Environ. Sci. Health A. 33, 1009–1021.
Karigar, C., Mahesh, A., Nagennahalli, M., Yun, D. J. (2006) Phenol degradation by immobilized cells of Arthrobacter citreus. Biodegradatio. 17, 47–55.
Lin, S. H., Chuang, T.-S. (1994) Combined treatment of phenolic wastewater by wet air oxidation and activated sludge. Technol. Environ. Chem. 44, 243–258.
Marrot, B., Barrios-Martinez, A., Moulin, P., Roc, N. (2006) Biodegradation of high phenol concentration by activated sludge in an immersed membrane bioreactor. J. Biochem. Eng. 30, 174–183.
McDowell, E. M., Trump, B. F. (1976) Histologic fixative suitable for diagnostic light and electron microscopy. Arch. Pathol. Lab. 10, 405–413.
Mohana, S., Shah, A., Divecha, J., Madamwar, D. (2008) Xylanase production by Burkholderia sp. DMAX strain under solid state fermentation using distillery spent wash. Bioresour. Technol. 99, 7553–7564.
Neumann, G. Veeranagouda, Y., T. B., Karegoudar, Sahin, O., Mausezahl, I., Kabelitz, N., Kappelmeyer, U., Heipieper, H. J. (2005) Cells of Pseudomonas putida and Enterobacter sp. Adapt to toxic organic compounds by increasing their size. Extremophile. 9, 163–168.
Plackett, R. L., Burman, J. P. (1946) The design of optimum multi-factorial experiments. Biometrik. 33, 305–325.
Rasanen, L. A., Elvang, A. M., Jansson, J., Lindstrom, K. (2001) Effect of heat stress on cell activity and cell morphology of the tropical Rhizobium, Sinorhizobium arboris. FEMS Microbiol. Ecol. 34, 267–278
Sung, R. H., Soydoa, V., Hiroaki, O. (2000) Biodegradation by mixed microorganism of granular activated carbon loaded with a mixture of phenols. Biotechnol. Lett. 22, 1093–1096.
Watanabe, K., Hino, S., Onodera, K., Kajie, S., Takahashi, N. (1996) Diversity in kinetics of bacterial phenoloxygenating activity. J. Ferment. Bioeng. 81, 562–565.
Yus Azila, Y., Mashitah, M. D., Bhatia, S. (2008) Process optimization studies of lead (Pb(II)) biosorption onto immobilized cells of Pycnoporus sanguineus using response surface methodology. Bioresour. Technol. 99, 8549–8552.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Elahwany, A.M.D., Mabrouk, M.E.M. Isolation and Characterization of A Phenol-Degrading Strain of Alcaligenes sp. AM4. BIOLOGIA FUTURA 64, 106–117 (2013). https://doi.org/10.1556/ABiol.64.2013.1.10
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.64.2013.1.10